The 1D TOCSY Experiment
- Posted at June 22, 2016
In my last post I showed how the 2D TOCSY experiment can be used to identify individual “spin systems” or subunits within a molecule, or even within different molecules. Well, there is a very closely related, one-dimensional version of the TOCSY experiment that uses something called “selective excitation” in place of a full 2D experiment. This has the advantage that it is both quicker to run and easier to understand. The basic idea behind this so-called 1D TOCSY experiment is quite straightforward: first, a combination of shaped rf pulses and magnetic field gradients is used to selectively excite one signal in the spectrum; then, a special pulse sequence element called a “spinlock” is used to transfer magnetization from the “excited” spins to the other spins in the spin system, in exactly the same way as is done in 2D TOCSY. The result is a 1D spectrum in which only signals that appear from those that are in the same spin system as the excited signal. This 1D TOCSY experiment can be very useful in a number of situations, particularly when there is considerable overlap in the spectrum. It’s a quick experiment, too, typically taking only a minute or two to run.
Let’s return to our mixture of 2-pentenal and 5-epoxyhexene. As discussed before, the proton spectrum shows a lot of overlap between the signals from the two sample components.
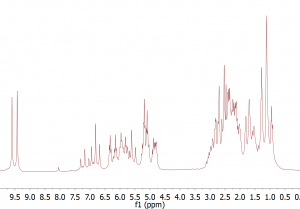
Fig. 1. Proton spectrum of a mixture of 2-pentenal and 5-epoxyhexene. The spectrum shows considerable overlap between the signals from the two components.
However, there is one well resolved resonance at 9.5 ppm, that of the aldehyde in 2-pentenal. With TOCSY 1D we can selectively excite this signal, as shown in the lower spectrum of Fig. 2 below. Then, we can use a TOCSY spinlock to transfer magnetization from the aldehyde proton to the other protons in the 2-pentenal spin system, resulting in the top spectrum of Fig. 2.
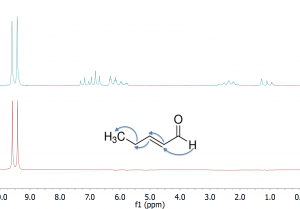
Fig. 2. 1D TOCSY of sample of 2-pentenal and 5-epoxyhexene. In the lower spectrum, selective excitation of the aldehyde resonance was achieved using a double pulse field gradient spin echo (DPFGSE) with a pair of “q3” Gaussian Cascade shaped frequency-selective pulses. In the top spectrum, an additional TOCSY transfer from the aldehyde proton to other protons in 2-pentenal was achieved using a 150 ms DIPSI-2 spinlock. Undesirable zero-quantum coherence responses were suppressed using the method of Thrippleton and Keeler 1 that employs a pair of adiabatic pulses in the presence of field gradients.
Fig. 3 below shows a comparison of the TOCSY 1D spectrum of our sample mixture with the original proton spectrum of the mixture and a proton spectrum of pure 2-pentenal.

Fig. 3. Comparison of 1D TOCSY spectrum of a mixture of 2-pentenal and 5-epoxyhexene with the proton 1D spectrum of the mixture and the proton 1D spectrum of 2-pentenal.
As you can see, in addition to aiding the identification of spin systems, the 1D TOCSY experiment also provides a very powerful way of “editing” the spectrum to remove signal overlap and simplify the spectrum – only signals from 2-pentenal show up in the 1D TOCSY spectrum above.
The Spinsolve benchtop NMR spectrometer is available with software to support pulse programming capabilities, including shaped rf pulses and gradients for coherence selection. If you have any questions or want to discuss how the TOCSY or other NMR methods can help with your application please Contact Us
References “Elimination of Zero-Quantum Interference in Two-Dimensional NMR Spectra” , Michael J. Thrippleton, and James Keeler, J. Angew Chem Int Ed 2003; 42 ; 3938-41.
Acknowledgements Many thanks to Dr. Alan Kenwright of Durham University for the sample used here and for the generous use of their Spinsolve system equipped with gradients.

Questions about our products?
Get in touch with us anytime.
Germany : +49 241 9278 7270 USA: +1 855 667 6835 New Zealand: +64 4 477 7096
Drop an email:
Quick finder, applications.
An Introduction to Total Correlation Spectroscopy (TOCSY)
- Download PDF Copy
- View Supplier Profile
- Request Quote
In NMR experiments, magnetizations that are perpendicular to the static magnetic field B0 will rotate about the B0 field at its typical Larmor frequency. This phenomenon is commonly known as chemical shift precession.
The magnetizations—apart from the chemical shift precession—will also emerge under the effect of the mutual coupling between the spins: the scalar or J-coupling.
While the evolution of the chemical shift accounts for the position of a specific resonance in the spectrum, the J-coupling happens to be the source of the peak splitting patterns. The evolution is controlled by the chemical shift when the J-couplings are small, when compared to the variance between the resonance frequencies of the coupled spins. This might be true because either the B0 field is high or the pair of spins is split in the molecule by many bonds, creating a small coupling.
In certain experiments, it is preferred to suppress the evolution of the chemical shift and enable the spin system to emerge only under the J-coupling. This is usually called isotropic mixing.
The chemical shift evolution can be suppressed by applying an intense RF pulse along a selected direction while making sure that the magnetizations remain aligned with the field. This is known as spin-locking, and the RF field is called the spin-lock field.
There are several techniques that manipulate composite pulses developed to offer a spin-lock. In such cases, imperfections in the RF-pulse can be constantly refocused to offer a clean spin-lock. The DIPSI (Shaka, AJ, Lee, CJ, and Pines, A, Journal of Magnetic Resonance 77(2), 274-293, 1988) and MLEV-17 (Bax, A and Davis, DG, Journal of Magnetic Resonance 65, 355-360, 1985) are the two most common techniques.
Total correlation spectroscopy , or TOCSY in short, can be described as a homonuclear 2D experiment analogous to COSY; in COSY, the J-coupling between two hydrogen nuclei appears as a crosspeak in the spectrum. But unlike COSY, the detection of the coupled spins is not restricted to the closest neighbors. During spin-locking, the TOCSY experiment manipulates the isotropic mixing condition to generate crosspeaks between all hydrogen nuclei forming part of a continuous chain of coupled spins.
A chain of four hydrogens—labeled A, B, C, and D—can be considered. Here, hydrogen A is linked to B that is linked to C, which, in turn, is linked to D. In a COSY experiment, a crosspeak would occur between the hydrogen nuclei in the following pairs of positions—that is, (A,B), (B,C), and (C,D). On the contrary, the hydrogens’ TOCSY spectrum would display crosspeaks between all pairs of hydrogen nuclei, as shown in Figure 1.
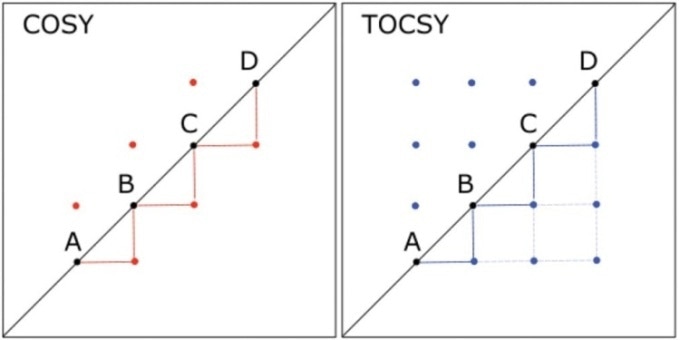
Figure 1. Comparison of 2D COSY and 2D TOCSY spectra for a hypothetical molecule in which hydrogen A is coupled to hydrogen B, which is coupled to C, which in turn, is coupled to D. Lines are drawn to connect the peaks below the diagonal, illustrating the throughbond connectivity shown by each spectrum; heavier lines indicate couplings shown in both spectra, and lighter lines indicate connectivity shown in the TOCSY spectrum but not the COSY.
To further explain this concept, the molecule trans-2-hexenoic acid depicted in Figure 2 (top) is considered. The molecule’s 1D 1 H NMR spectrum shows six resonances (see Figure 2, bottom).
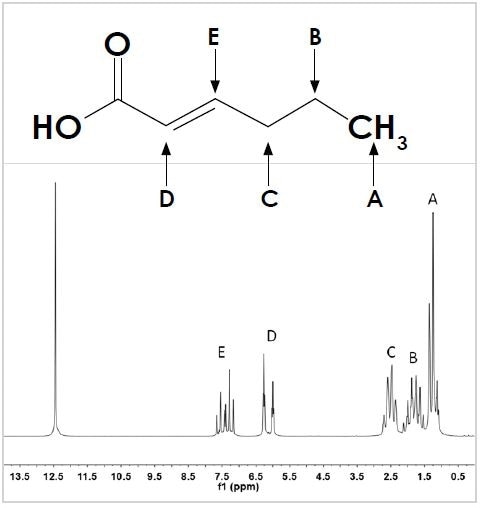
Figure 2. The structure (top) and 1D 1 H spectrum (bottom) of trans-2-hexenoic acid. Hydrogen positions on the carbon backbone are labeled A to E to identify the appropriate resonance in the spectrum. The unlabeled singlet at 12.5 ppm corresponds to the –OH group of the carboxylic acid.
Five of these resonances have been labeled A to E corresponding to the hydrogens coupled to the molecule’s carbon backbone, while the sixth one corresponds to the –OH of the carboxylic acid group. All the resonances A to E display peak splitting that corresponds to the way in which they are attached to the neighboring hydrogens.
As illustrated in Figure 3, the COSY spectrum shows the coupling between (A,B) and (B,C) and between (C,D) and (D,E). In addition, it reveals the coupling between C and E, which may occur as a result of the double bond between D and E; this can lead to a more robust coupling between C and E than between D and B, for example.
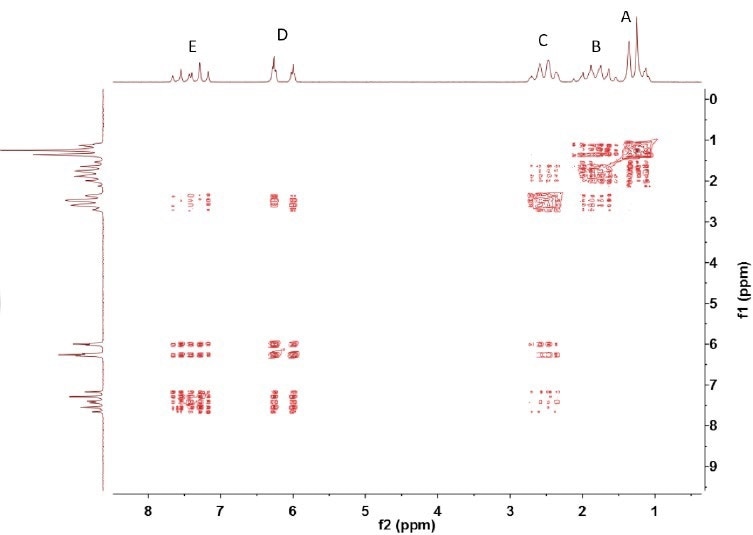
Figure 3. COSY spectrum of trans-2-hexenoic acid
The TOCSY spectrum (see Figure 4) reveals the coupling between all pairs of hydrogen nuclei. This demonstrates the fact that resonances A to E contain a single continuous chain of coupled spins.
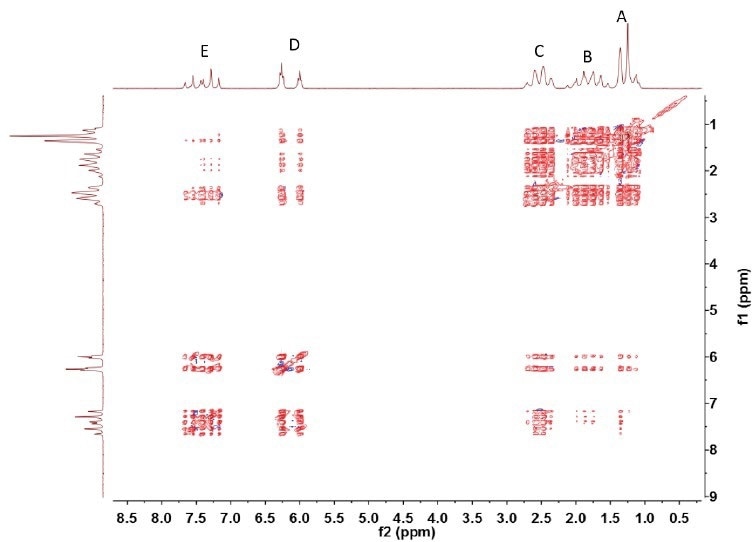
Figure 4. Spin-lock was implemented using MLEV-17
If Figure 3 is compared with the TOCSY spectrum of ethyl crotonate in Figure 5, it can be observed that the ethyl crotonate contains two separate chains of spins—the ethyl group, resonances A and C, and the crotonyl group, resonances B, D, and E.
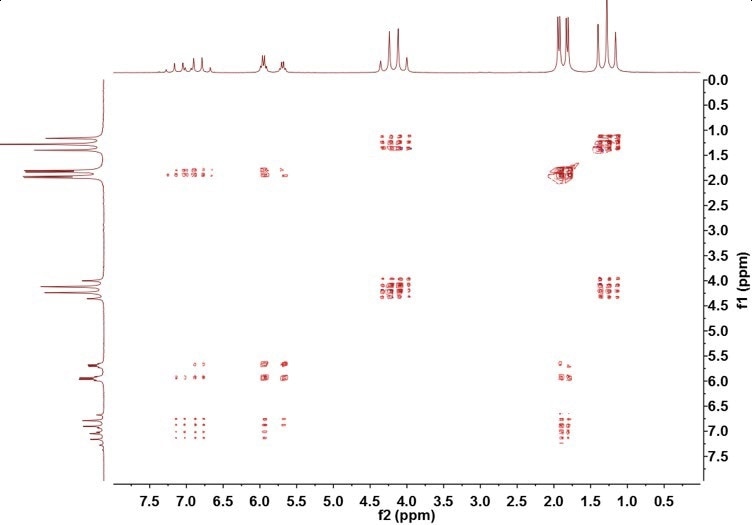
Figure 5. TOCSY spectrum of ethyl crotonate

The absence of crosspeaks between the two sets of spins demonstrates the presence of a nucleus that successfully breaks the J-coupling chain; in this example, the nucleus is the oxygen from the ester coupling.

This information has been sourced, reviewed and adapted from materials provided by Oxford Instruments Magnetic Resonance.
For more information on this source, please visit Oxford Instruments Magnetic Resonance.
Please use one of the following formats to cite this article in your essay, paper or report:
Oxford Instruments Magnetic Resonance. (2023, March 14). An Introduction to Total Correlation Spectroscopy (TOCSY). AZoM. Retrieved on August 30, 2024 from https://www.azom.com/article.aspx?ArticleID=18572.
Oxford Instruments Magnetic Resonance. "An Introduction to Total Correlation Spectroscopy (TOCSY)". AZoM . 30 August 2024. <https://www.azom.com/article.aspx?ArticleID=18572>.
Oxford Instruments Magnetic Resonance. "An Introduction to Total Correlation Spectroscopy (TOCSY)". AZoM. https://www.azom.com/article.aspx?ArticleID=18572. (accessed August 30, 2024).
Oxford Instruments Magnetic Resonance. 2023. An Introduction to Total Correlation Spectroscopy (TOCSY) . AZoM, viewed 30 August 2024, https://www.azom.com/article.aspx?ArticleID=18572.
Ask A Question
Do you have a question you'd like to ask regarding this article?
Cancel reply to comment
Trending Stories
1 new cooling technology offers promise for energy efficiency and sustainability, 2 nano dimension to acquire desktop metal, creating a leader in additive manufacturing, 3 expanding the utility of aromatic ketones through a versatile one-pot transformation, 4 ai accelerates polymer development for energy, filtration, and recycling, 5 surface modification of alloy fibers for concrete with kh-550, 6 polyimide network substrates for electronic waste mitigation, related applications.

Related Equipment

More Content from Oxford Instruments Magnetic Resonance

Introducing Sample Automation and Major New Capabilities for the X-Pulse Benchtop NMR Spectrometer

How to Optimize Battery Electrolytes with Benchtop NMR

NMR Spectroscopy for Chemical Industry
Your AI Powered Scientific Assistant
Hi, I'm Azthena, you can trust me to find commercial scientific answers from AZoNetwork.com.
A few things you need to know before we start. Please read and accept to continue.
- Use of “Azthena” is subject to the terms and conditions of use as set out by OpenAI .
- Content provided on any AZoNetwork sites are subject to the site Terms & Conditions and Privacy Policy .
- Large Language Models can make mistakes. Consider checking important information.
Great. Ask your question.
Azthena may occasionally provide inaccurate responses. Read the full terms .
While we only use edited and approved content for Azthena answers, it may on occasions provide incorrect responses. Please confirm any data provided with the related suppliers or authors. We do not provide medical advice, if you search for medical information you must always consult a medical professional before acting on any information provided.
Your questions, but not your email details will be shared with OpenAI and retained for 30 days in accordance with their privacy principles.
Please do not ask questions that use sensitive or confidential information.
Read the full Terms & Conditions .
Provide Feedback
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- HHS Author Manuscripts

Use of optimized 1D TOCSY NMR for improved quantitation and metabolomic analysis of biofluids
Peter sandusky.
Department of Chemistry, Eckerd College, St. Petersburg, FL 33711, USA
Emmanuel Appiah-Amponsah
Department of Chemistry, Purdue University, West Lafayette, IN 47907, USA
Daniel Raftery
One dimensional selective TOCSY experiments have been shown to be advantageous in providing improved data inputs for principle component analysis (PCA) ( Sandusky and Raftery 2005a , b ). Better subpopulation cluster resolution in the observed scores plots results from the ability to isolate metabolite signals of interest via the TOCSY based filtering approach. This report reexamines the quantitative aspects of this approach, first by optimizing the 1D TOCSY experiment as it relates to the measurement of biofluid constituent concentrations, and second by comparing the integration of 1D TOCSY read peaks to the bucket integration of 1D proton NMR spectra in terms of precision and accuracy. This comparison indicates that, because of the extensive peak overlap that occurs in the 1D proton NMR spectra of biofluid samples, bucket integrals are often far less accurate as measures of individual constituent concentrations than 1D TOCSY read peaks. Even spectral fitting approaches have proven difficult in the analysis of significantly overlapped spectral regions. Measurements of endogenous taurine made over a sample population of human urine demonstrates that, due to background signals from other constituents, bucket integrals of 1D proton spectra routinely overestimate the taurine concentrations and distort its variation over the sample population. As a result, PCA calculations performed using data matrices incorporating 1D TOCSY determined taurine concentrations produce better scores plot subpopulation cluster resolution.
Introduction
The field of metabolomics combines high resolution analytical methods such as NMR and/or mass spectrometry with the multivariate statistical analysis of populations of complex matrix samples such as biofluids, foods, etc. ( Nicholson et al. 1999 ; Fiehn et al. 2000 ; Kell 2004 ; Fernie et al. 2004 ; Van der Greef and Smilde 2005 ; Van Dien and Schilling 2006 ; Serkova and Niemann 2006 ; Pan and Raftery 2007 ; Gowda et al. 2008 ; Zhang et al. 2010 ). The principal objective of the statistical analysis of biofluid populations, as it is currently being applied for forensic and diagnostic purposes, is to detect subpopulations within the parent population in such a way that “unknown” samples may be assigned to one or another of the scores plot subpopulations. Standard metabolomic studies often fail because of inadequate subpopulation cluster resolution, as can often be seen in the scores plots of principal component analysis (PCA) or even supervised multivariate methods (such as partial least squares discriminant analysis, PLSDA). Thus, any technical innovation that routinely increases the subpopulation cluster resolution constitutes a significant advance in the field.
In previous publications the authors have demonstrated that using discrete concentration measurements of selected biofluid constituents, made using the 1D TOCSY experiment, as data inputs for PCA calculations will improve scores plot subpopulation cluster resolution over what can be obtained using bucket integrals of 1D proton NMR spectra ( Sandusky and Raftery 2005a , b ). In a subsequent publication it was demonstrated that using discrete biofluid constituent concentrations made using a spectral library software as PCA data inputs has a very similar effect, in that it will also improve scores plot subpopulation cluster resolution over what can be obtained using bucket integrals of 1D proton NMR spectra as PCA data inputs ( Weljie et al. 2006 ).
Though these studies demonstrated that the use of discrete component measurements as data inputs will improve the PCA scores plot subpopulation cluster resolution for metabolomics studies of biofluid populations, they did not investigate the origins of this effect in a quantitative manner. This work reexamines this phenomenon. After a discussion on optimizing the 1D TOCSY experiment as it applies to the measurement of biofluid chemical constituents, this report compares the precision and accuracy of constituent measurements made in a complex biofluid matrix (human urine) via the 1D TOCSY experiment with those obtained by the bucket integration of 1D proton NMR spectra, and by analysis using Chenomx spectral library software. The utility of adding even limited 1D TOCSY data to multivariate statistical analysis is also demonstrated.
Experimental
Nmr samples.
Taurine, lactate, TMAO, histidine, and TSP (sodium 3-trimethylsilyl (2,2,3,3 2 H 4 ) 1-propionate) were purchased from Sigma–Aldrich (St. Louis, MO) and used without further purification. Metabolite stock solutions were prepared in 100 mM phosphate buffer at pH 7. Human urine samples were collected from healthy volunteers in accordance with the Institutional Review Board at Purdue University. For NMR analysis, urine samples were prepared by the addition of 120 µl of 0.5 M phosphate buffer, pH 7, to 480 µl of neat urine. All NMR samples were run in 5 mm tubes with 10% added D 2 O (Cambridge Isotope Laboratories Inc., Andover, MA) and 100 µM TSP.
NMR spectroscopy
NMR spectra were acquired on a Bruker AVANCE DRX 500 MHz spectrometer (Bruker-Biospin, Fremont, CA), using a 5 mm inverse HCN triple resonance probe equipped with XYZ axis gradient coils. All spectra were acquired at 25°C, and were referenced to the TSP methyl peak at 0.000 ppm. All pulse sequences were performed and spectra were acquired using the Bruker XWINNMR software package, release 3.5. Spectral data were processed and integrated using the Bruker Topspin software package, release 2.0. Chenomx measurements of endogenous taurine concentrations were made in Chenomx Profiler 5.1 (Chenomx, Edmonton, Canada) using the 500 MHz pH 6–8 metabolite spectral library, and the 100 µM TSP peak (0.000 ppm) as a concentration reference.
1D proton NMR spectra
1D proton NMR spectra were acquired using a 1D NOESY pulse sequence incorporating presaturation for water suppression during the relaxation delay and mixing time ( Nicholson et al. 1995 ; Belton et al. 1998 ). The relaxation delay and mixing times were set to 2 s and 300 ms, respectively, and the presaturation power used was the minimum needed to effect complete suppression of the water peak. The sweep width was 10,330 Hz. In order to achieve high signal-to-noise ratios for minor components, sixty-four FID transients (of 64 k points) were averaged, resulting in a total acquisition time of 7 min. The FIDs were zero filled once, and 0.3 Hz line broadening was used in processing the spectra. A “qfil” background correction with a spectral width of 0.2 ppm was used to remove any remaining water peak.
1D TOCSY spectra
The 1D TOCSY pulse sequences described in Fig. 1 were written as modifications of those found in the Bruker XWINNMR pulse program library to match descriptions of the sequences found in ( Kessler et al. 1986 ; Stott et al. 1995 ; and Facke and Berger 1995 ). Gaussian-shaped pulsed z-field gradients were 1 ms in duration. The 100% strength for the gradient pulses was 50 gauss per cm. The Gaussian, Secant and IBURP1 shaped selective pulses used were generated using the XWINNMR “Shape Tool” utility incorporated in the Bruker XWINNMR software package ( Bauer et al. 1984 ; Geen et al. 1989 ; Geen and Freeman 1991 ). The Shape Tool utility was also used to calculate the duration of the chosen shaped pulses corresponding to the desired excitation band width that was typically 10–25 Hz, depending on the multiplet structure of the excited peak ( Sandusky and Raftery 2005a , b ). Two Hz was added to these excitation bandwidths to allow for small variations in chemical shift between samples. Typical shaped pulse lengths were 40–100 ms. The power levels of the shaped pulses effecting selective 90° or 180° rotations were determined and optimized independently for each pulse shape and excitation band width used. Typical pulse power levels for the B1 field were in the range of 58 to 68 dB (Bruker).

Various forms of the Selective TOCSY pulse sequence: a Original form of the experiment as described by Kessler et al. (1986) ; b experiment with pulsed field gradient spin echo module (PFGSE) for improved suppression outside the target excitation band ( Stott et al.1995 ); c experiment using asymmetric field gradients ( Facke and Berger 1995 ); d experiment incorporating both a PFGSE module for selective excitation, and a z filter for read peak rectification ( Sorensen et al. 1984 ). SP shaped RF pulse; Gaussian, Secant, or IBURP 1. GP pulsed z field gradient. SPINLOCK MLEV 17, DIPSI 2, DIPSI 3, or FLOPSY 8. VD z filter variable delay
The MLEV 17 TOCSY spinlock sequence used in the experiment was the same as that for pulse sequences found in the Bruker XWINNMR pulse program library ( Bax and Davis 1985 ). The DIPSI 2, DIPSI 3 and FLOPSY 8 spinlock sequences were written to match the descriptions given in ( Shaka et al. 1988 ; Kadkhodaie et al. 1991 ). The z-filter used in pulse sequence D consisted of two spinlock power level 90° pulses separated by a variable delay (VD) ( Sorensen et al. 1984 ).The z-filter VD list was made using the first ten positive values of a random number list that was generated for a mean value of 10 ms with a standard deviation of 5 ms, as calculated using the Microsoft Excel random number generator. The sweep width used for the 1D TOCSY experiments was 5,000 Hz. Eight 64 k point FID transients were averaged in each 1D TOCSY experiment, resulting in a total acquisition time of 85 s if two dummy scans are included. Line broadening of 0.3 Hz was used in processing the spectra.
Statistical analysis
Populations of 1D proton urine spectra were prepared as data matrices for PCA calculations by bucket integration in AMIX 2.1 (Bruker-Biospin, Fremont, CA). Before bucket integration, the spectra were aligned by setting the TSP peaks to a value of 0.000 ppm. Simple rectangular buckets with widths of 0.04 ppm were employed, and the sum of the integrals for each spectrum was scaled to a value of 1.0. Exclusion regions were employed in those cases, as described in the Results Section below, where integrals arising from the spectral features of ethanol, hippurate and creatinine were excluded. The urea peak appearing between 5.0 and 6.2 ppm was excluded in all cases. The bucket integral tables generated by AMIX were exported into Microsoft Office EXCEL 2003 for analysis (Microsoft Corp., Redmond, WA). Substitutions of the taurine 1D TOCSY read peak integrals for the corresponding taurine 1D proton bucket integrals, and renormalization of the resulting data matrices, were also performed in EXCEL. PCA calculations were performed using both Minitab 13 (Minitab Inc., State College, PA) and MATLAB R2007a (The Mathworks Inc., Natick, MA) with equivalent results. PCA calculations were performed using mean centered data and unit variance scaling. Pearson product moment correlation coefficients were calculated using the “PEARSON” function in Microsoft Office EXCEL 2003. ANOVA p values and F-numbers for the PC1 and PC2 scores were calculated using the “anova” function in MATLAB R2007a.
TOCSY optimization
The 1D TOCSY experiment was originally described in the mid 1980s ( Kessler et al. 1986 ) and various modifications have since then been presented in the literature. These include modifications to the basic pulse sequence ( Fig. 1 ), ( Stott et al. 1995 ; Facke and Berger 1995 ) different types of frequency selective pulses ( Bauer et al. 1984 ; Geen et al. 1989 ; Geen and Freeman 1991 ), and various TOCSY spin lock sequences ( Bax and Davis 1985 ; Shaka et al. 1988 ; Kadkhodaie et al. 1991 ). The effectiveness of these variations, as they relate to the measurement of biofluid constituent concentrations, were examined in experiments using human urine as a representative biofluid matrix. Four common urine constituents, hippurate, histidine, taurine and lactate were used as target species. For each combination of pulse sequence, selective pulse shape and TOCSY spinlock, the experimental parameters were optimized so as to obtain the highest possible target read peak signal-to-noise ratio. The best results were obtained using a sequence incorporating a pulse field gradient spin echo (PFGSE) module for selective band excitation (sequence B in Fig. 1 ). The IBURP shaped pulse used in the PFGSE module for selective inversion provides a more uniform excitation across the target excitation bandwidth, and thus produces a 10–15% improvement in the read peak intensity over that produced using a Gaussian or Secant shaped pulse (data not shown) ( Bauer et al. 1984 ; Geen et al. 1989 ; Geen and Freeman 1991 ). It was also found that FLOPSY 8 performed best as the TOCSY spinlock, except when the target species has smaller J couplings, in which case DIPSI 2 or DIPSI 3 can be used ( Table 1 ) ( Shaka et al. 1988 ; Kadkhodaie et al. 1991 ). A z-filter modification to pulse sequence B is also sometimes useful to remove negative components from the read peaks (sequence D in Fig. 1 ) ( Sorensen et al. 1984 ).
Effect of TOCSY Spinlock sequences on the intensity of read peaks (data obtained using pulse sequence B and the IBURP1 shaped pulse)
| Target | Excitation peak | Read peak | ΔHz | J(Hz) | Mixing time (ms) | Read peak signal to noise ratio | |||
|---|---|---|---|---|---|---|---|---|---|
| MLEV-17 | DIPSI-2 | DIPSI-3 | FLOPSY-8 | ||||||
| Taurine | Taurine 1 | Taurine 2 | 90 | 7 | 80 | 42 | 45 | 45 | 48 |
| Histidine | Taurine 1 | Hist α | 370 | 7 | 40 | 67 | 78 | 74 | 90 |
| Lactate | CH3 | CH | 1,450 | 7 | 35 | 26 | 30 | 30 | 40 |
| Histidine | Hist β | Hist 2(4) | 1,989 | 2–3 | 210 (140) | 8 | 16 | 17 | 10 |
Quantitation
Application of this basic 1D TOCSY experiment to any particular biofluid constituent of interest is very straight forward, and involves three steps. First, the target peak excitation frequency relative to the center of the spectrum, or “offset,” and target peak width are measured from a 1D proton spectrum. For many common biofluid constituents, hippurate, citrate, lactate and creatinine would be examples in the case of urine, this can be done using the endogenous concentrations. In other cases, where peak overlap completely obscures the target peak, it may be necessary to “spike” the constituent of interest into the first sample of the sample population set. Second, for each constituent target peak three 1D TOCSY parameters (selective pulse length, selective pulse power, and TOCSY mixing time) should be adjusted so as to optimize the read peak intensity. The selective pulse length can be calculated from the target excitation peak width using utilities such as the VNMR “PBOX” or XWINNMR “Shape Tool.” However, we strongly recommend the addition of ~2 Hz to the observed target excitation peak width when performing this calculation ( Sandusky and Raftery 2005a , b ). This “loose fit” will avoid the potential problem of small peak shifts that can occur as a result of pH or ion concentration variations in the samples. The selective pulse power is adjusted to give the largest excitation peak with the TOCSY power completely attenuated. The TOCSY is then turned back on, and the optimal TOCSY mixing time is determined. Third, if measurements of absolute concentrations are needed, as opposed to relative concentrations, the response of the 1D TOCSY experiment for each particular constituent of interest should be calibrated using a spiked sample. Of course, in analyzing a set of samples for a metabolomics study of a biofluid population, this parameter optimization and calibration procedure need be done on only one sample.
Figure 2 illustrates the use of the 1D TOCSY experiment applied to the concentration measurement of a single chemical constituent present in a complex biofluid mixture. In this particular example the target species is taurine. It should be noted that when observed as a pure species, taurine gives a classic A 2 X 2 proton NMR spectrum with two triplets of equal intensity appearing at 3.45 and 3.28 ppm. Significantly, in the proton NMR spectrum of the human urine sample shown in Fig. 2 , both of these taurine triplets are largely obscured by the presence of other species. The application of the 1D TOCSY experiment to this sample allows the clean observation of taurine triplets without interference from the other metabolites present. Thus, the use of the lower field triplet at 3.45 ppm as the “target peak” for selective pulse excitation produces a taurine triplet TOCSY “read peak” at 3.28 ppm, while the use of the high field triplet at 3.25 ppm as the “target peak” produces a TOCSY “read peak” for taurine at 3.45 ppm and an additional read peak resulting from a second metabolite species designated here as “U1,” or Unknown 1. The endogenous taurine concentration in this sample was 400 µM. Subsequently, using a urine sample spiked with an aliquot of standard histidine, the major constituent species interfering with the observation of the taurine high field peak were determined to be trimethylamine oxide (TMAO), which produces a singlet at 3.28 ppm, and histidine, which has peaks at 3.17, 3.28 and 4.0 ppm ( Fig. 3 ).

Use of the Selective TOCSY experiment to isolate the peaks of the metabolite taurine from obscuring peaks in a human urine sample. Note that taurine peaks are completely obscured in 1D 1 H NMR spectrum ( top two spectra ), but cleanly isolated in the Selective TOCSY spectra. Selective TOCSY experiments used pulse sequence B shown in Fig. 1 , employing an IBURP1 shaped pulse to effect a 25 Hz selective excitation centered at the frequency positions indicated by an asterisk (*), and a 35 ms DIPSI 3 spinlock. Experiments were performed at an endogenous taurine concentration of 0.4 mM. The unknown species labeled here as “U1” was subsequently identified as histidine (see Fig. 3 )

Selective TOCSY experiment performed on another human urine sample with target excitation at 4.0 ppm. Note that the experiment establishes that one of the histidine beta peaks contributes, along with TMAO, to the peaks obscuring the high field taurine triplet peak at 3.28 ppm. The experimental set up was similar to that used for the experiment shown in Fig. 2 . An IBURP 1 pulse was used to effect a 20 Hz selective excitation centered at 4.0 ppm indicated by the asterisk (*). A DIPSI 2 spinlock was applied for 40 ms. Histidine observed was endogenous in this sample
Figure 4 presents a titration of taurine into a human urine sample. Comparing the plots of the integrals of the taurine TOCSY read peaks and to those of corresponding segments of the 1D proton spectra indicates that, as measurements of biofluid constituent concentrations, the TOCSY read peaks are equivalent to bucket integrals of 1D proton spectra in terms of precision. The slopes of least squares linear fits for the low field and high field taurine 1D TOCSY read peak integration plots are in agreement to within ±1%.

Titration of taurine into a human urine sample: a integral of 1D TOCSY spectrum around 3.28 ppm; b integral of 1D proton spectrum around 3.28 ppm; c integral of 1D TOCSY spectrum around 3.45 ppm; and d integral of 1D proton spectrum around 3.45 ppm. Experimental parameters were similar to those used for Fig. 2 . Note that ordinate offsets in ( c ) and ( d ) are due to contributions from histidine, TMAO and other species
More important however is that, because peak overlap adds an “integral background” to the bucket integrals of the 1D proton spectra, the integrals of the TOCSY read peaks provide a much more accurate, and internally consistent, measurement of constituent concentrations. This effect can readily be seen as the large offsets or intercept values in Fig. 4c and d . Thus, the bucket integral of the 1D proton spectrum at the high field taurine triplet implies an endogenous taurine concentration of 6.6 mM ( Fig. 4c ), while bucket integration of the same 1D proton spectrum at the low field taurine triplet indicates an endogenous taurine concentration of 770 µM ( Fig. 4d ). The actual endogenous taurine concentration in this sample, as measured using 1D TOCSY, is below 100 µM.
Tables 2 and and3 3 presents 1D TOCSY and bucket integral measurements of the endogenous taurine concentration in a population of six human urine samples collected from six different individuals. In all six samples the bucket integration of the 1D proton spectrum significantly overestimates the taurine concentration, and gives internally inconsistent measurements. Clearly in the case of the high field taurine triplet region around 3.28 ppm the major contributions to the bucket integral background are TMAO and one of the histidine beta peaks ( Fig. 3 ). However, the presence of a significant integral background in the low field taurine triplet bucket integral around 3.45 ppm (column #4 in Table 2 ) suggests that, in crowded regions of the 1D proton spectra of biofluids, there is an aggregate contribution to bucket integral background coming from many minor constituents. These minor constituents would presumably be present at low concentrations, and would thus be undetected and unrecognized as individual biofluid constituents. It is also important to note that the 1D TOCSY measurements of the two taurine read peaks show a very high degree of statistical correlation over the sample population, giving a Pearson Product Moment Correlation Coefficient of 0.997 (see Table 3 ). In contrast, the bucket integrals of the 1D proton spectra in the corresponding regions give a Pearson product moment correlation coefficient of −0.408, indicating that these two regions are dominated by contributions from species other than taurine.
Measurement of endogenous taurine in a population of six human urine samples
| Column no. | 1D TOCSY read peak | 1D proton spectrum peaks | ||
|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |
| Urine sample | 3.28 ppm | 3.45 ppm | 3.28 ppm | 3.45 ppm |
| a | 0.786 | 0.831 | 3.784 | 1.670 |
| b | 0.466 | 0.455 | 2.733 | 1.443 |
| c | 0.915 | 0.918 | 4.186 | 1.859 |
| d | 0.436 | 0.387 | 2.767 | 1.019 |
| e | 0.157 | 0.130 | 4.584 | 1.353 |
| f | 0.058 | 0.028 | 6.572 | 0.773 |
Peak integration comparison for 1D TOCSY and 1D proton spectra (mM Taurine)
Pearson correlation product moments for integral columns
| Columns | Type | Pearson correlation product moment |
|---|---|---|
| 1 × 2 | TOCSY × TOCSY | 0.997 |
| 1 × 3 | TOCSY × 1D | − 0.494 |
| 1 × 4 | TOCSY × 1D | 0.848 |
| 2 × 3 | TOCSY × 1D | 0.067 |
| 2 × 4 | TOCSY × 1D | 0.777 |
| 3 × 4 | 1D × 1D | − 0.408 |
Measurement of the endogenous taurine concentrations in the set of six urine samples was also made using the Chenomx Profiler spectral library software, Chenomx Inc., Edmonton Canada. In these Chenomx measurements the low field taurine triplet at 3.45 ppm was used as the indicator of taurine concentration, and the 100 µM TSP peak at 0.000 ppm was used as the concentration reference. The resulting taurine concentrations, measured by applying the Chenomx approach to the 1D 1 H NMR spectra, were in general intermediate between the values measured using 1D TOCSY and those determined by integration of the 1D proton spectra. Also in two cases, samples e and f, the endogenous taurine concentrations were too low relative to the obscuring species to allow for identification and measurement using the Chenomx software. A newer version of the software (Chenomx 7.0) was also used to verify the concentration determination, and resulted in essentially the same value.
In order to examine the effect of including the more accurate 1D TOCSY measurements as PCA data inputs, a population of samples was generated by spiking taurine into each of the six samples of human urine discussed above. This resulted in a population of twelve samples with two subpopulations; a “low taurine” subpopulation with taurine concentrations between 0 and 1 mM, and a “high taurine” subpopulation with taurine concentrations between 4 and 5 mM. Interestingly, the PC1 versus PC2 scores plot from PCA calculations performed using bucket integrated 1D proton spectra fail to resolve the “high taurine” and “low taurine” subpopulations to any degree ( Fig. 5a ). However when 1D TOCSY read peak integrals were substituted into the bucket integral matrix in the place of the corresponding taurine 1D proton bucket integrals, PCA calculations produced complete cluster resolution of the two populations along PC2 ( Fig. 5b ). The ANOVA p -value for the PC2 scores drops from 0.87, for the 1D proton spectrum based calculation, to 6.5 × 10 −6 , for the calculation performed with a data matrix including the taurine 1D TOCSY read peak integrals. Similarly, PCA calculations performed using simulated bucket integral data indicate that inclusion of “integral background” in bucket integral based PCA calculations will generally have deleterious effects on scores plot subpopulation cluster resolution (data not shown).

PC1 versus PC2 PCA scores plots calculated from the 1D proton spectra of a set of human urine samples with two subpopulations. Yellow circles “low taurine” subpopulation (0–1 mM taurine). Blue diamonds “high taurine” subpopulation (4–5 mM taurine). Spectral features from urea (6.5–5 ppm) have been excluded. a Calculation based on data matrix of bucket integrated 1D proton spectra. b Repeat of calculation from A, but with taurine 1D TOCSY read peak integrals substituted for taurine 1D proton bucket integrals
It should be noted that in the analysis of the original 1D proton spectra, the sum of bucket integrals for each spectrum were scaled to a numerical value of 1.0. This is a standard procedure in NMR-based metabolomics, especially for urine spectra, and is performed to correct for the variation in total metabolite concentration over the sample population due to dilution effects ( Zhang et al. 2009 ). In order to make certain that the substitution of 1D TOCSY read peak integral data into the bucket integral matrix would not grossly distort this normalization, the highest 1D TOCSY read peak integral (that from the high field triplet of sample “c” spiked with taurine), was set numerically equal to the value found in the corresponding bucket integral for the 3.28 ppm peak in the original 1D proton spectra bucket integral matrix, and the remaining 1D TOCSY read peak integrals were scaled accordingly. Lastly, before the PCA calculations were performed, the data matrix, now incorporating the scaled 1D TOCSY read peak integral data, was renormalized such that the integrals for each spectrum summed to 1.
Examination of the loadings plots from the 1D proton bucket integral based PCA calculation (scores plot shown in Fig. 5a ), indicated that the calculation failed to resolve “low taurine” and “high taurine” subpopulations because the variance in the sample population was dominated by variations in 3 endogenous urine metabolites: ethanol, hippurate, and creatinine. When these metabolites were excluded during the generation of the bucket integral matrix using AMIX software, the scores plot of the 1D proton spectrum PCA calculation did now resolve the “low taurine” and “high taurine” subpopulations along PC1 ( Fig. 6a ). However, when the taurine TOCSY read peak integrals were substituted into the same data set (i.e., excluding the ethanol, hippurate and creatinine signals) there nevertheless was a significant improvement in the cluster resolution, and the p -value for the PC1 scores decreased from 2.9 × 10 −5 to 3.9 × 10 −9 ( Fig. 6 ).

PC1 versus PC2 PCA scores plots calculated from the 1D proton spectra of a population of human urine samples with two subpopulations. Repeat of calculations presented in Fig. 5 , but with spectral features from urea, ethanol, hippurate, and creatinine excluded. Yellow circles “low taurine” subpopulation (0–1 mM taurine). Blue diamonds “high taurine” subpopulation (4–5 mM taurine). a Calculation based on data matrix of bucket integrated 1D proton spectra. b Repeat of calculation from A, but with taurine 1D TOCSY read peak integrals substituted for taurine 1D proton bucket integrals
The selective 1D TOCSY experiment can be relied upon to produce quantifiable read peaks for target metabolites present at concentrations of ~50 µM or better within a 1 min acquisition time using a room temperature 5 mm inverse probe at 500 MHz. If a 5 mm inverse cryoprobe on an 800 MHz instrument were employed the threshold sensitivity of the method could be reduced to 10 µM. Furthermore, quantifiable 1D TOCSY read peaks may be obtained on constituent species even when the peaks of these species are completely obscured in the 1D proton spectrum by the presence of other biofluid constituents.
Because the integral intensities of the 1D TOCSY read peaks are proportional to the concentration of the target metabolite present ( Fig. 4 ; Bauer et al. 1984 ) they can be used as data inputs, in the place of 1D proton spectra bucket integrals, for chemometric calculations such as ANOVA, PCA or supervised multivariate methods. Furthermore, because 1D TOCSY read peaks make measurements free of the integral background inherent in bucket integration of crowded regions of 1D proton NMR biofluid spectra, using 1D TOCSY read peak integrals as input data for PCA calculations should routinely produce better scores plot subpopulation cluster resolution than that obtained using 1D proton spectra bucket integrals.
The use of discrete component concentrations in the place of 1D proton NMR bucket integrals as data input matrices for PCA calculations on biofluid sample populations has previously been shown to produce better scores plot subpopulation cluster resolution ( Sandusky and Raftery 2005a , b ; Weljie et al. 2006 ). The PCA calculation results presented in this paper indicate that two separate factors contribute to this improvement. First, constructing a PCA data matrix from discrete component concentrations allows the analyst to limit the data inputs to metabolite species that are statistically significant in resolving the subpopulations of interest, in effect excluding metabolite species whose presence in the data matrix may be deleterious to subpopulation cluster resolution. This effect is observed in comparing Fig. 6a and b . Second, the use of accurately measured discrete component concentrations, specifically concentration values that are not distorted by integral background, such as those obtained by 1D TOCSY, sharpens the statistical “signal” of the significant metabolite species as they vary over the sample population. This effect is observed in comparing Fig. 5a and b . Certainly other factors, such as the reduction or elimination of noise contributions found in 1D NMR proton bucket integral matrices ( Halouska and Powers 2006 ), also contribute as well, though probably to a lesser extent.
The occurrence of significant integral background in the 1D proton NMR spectrum bucket integral of the low field taurine peak at 3.45 ppm suggests, not surprisingly, that an aggregate of minor constituents will contribute to the integral background in crowded regions of the spectrum, even when these minor constituents are not present at concentrations high enough for them to be detected as individual constituents. 1D proton NMR spectra of urine samples routinely contain detectable peaks from only 30–40 detectable constituent species ( Foxall et al. 1993 ), whereas HSQC spectra of urine samples prepared using 15 N ethanolamine demonstrates that there are nearly 200 carboxyl-containing constituents alone present at NMR detectable concentrations ( Ye et al. 2009 ). Mass spectrometry indicates that perhaps as many as 1,400 constituent species are detectable in typical biofluid samples ( Fischer 2010 ).
Chenomx, Inc. (Edmonton, Canada) has developed a method for data extraction based on an NMR spectral library and software that allows the determination of concentration measurements for constituent chemical species from 1D proton NMR spectra of biofluids. The use of this spectral library software corrects for the problems of peak overlap, and gives more accurate measurements of biofluid constituents than bucket integrals. It has been demonstrated that using these spectral library extracted concentrations in PCA calculations can increase the scores plot subpopulation cluster resolution over that which can be obtained using bucket integrated 1D proton NMR spectra as data inputs ( Weljie et al. 2006 ). The Chenomx spectral library software is a very valuable tool for the analysis of biofluid populations. However, when significant spectral overlap or broad baseline components are present, this approach is more challenging. In such cases, the 1D TOCSY approach may find application. As demonstrated above, quantifiable 1D TOCSY read peaks may be obtained on metabolite species even when the peaks of these species are completely obscured in the 1D proton spectrum by the presence of other biofluid constituents. It seems unlikely that the use of spectral library based software can adequately address the problem presented by the contributions of an aggregate of minor constituents to the integral background as discussed above.
The authors believe that the use of discrete concentration measurements made using 1D TOCSY provides an improved technique in the forensic and diagnostic applications of metabolomics. In passing, we note that other types of TOCSY NMR experiments discussed in the literature may or may not be useful in similar contexts. Statistical TOCSY, or STOCSY, developed by the Nicholson and coworkers, uses statistical covariation over a population of biofluid samples to resolve the proton NMR spectra for certain individual metabolites ( Cloarec et al. 2005 ). The covariation TOCSY approach developed by Bruschweiler and Zhang uses the covariation inherent in an individual molecule’s chemical shift frequencies and J couplings, as they evolve over a sampled time domain, to separate the spectra of the individual chemical species in a mixture using a reduced number of increments in the 2D spectra ( Zhang and Bruschweiler 2004 , 2007 ). Recent work by the Emsley group has shown the utility of intraspectral correlation to better define peak integral limits for improved data analysis ( Holmes et al. 2007 ). Each of these approaches has its own strengths, and is being applied in a variety of metabolomics research studies ( Cloarec et al. 2005 ; Blaise et al. 2009 ; Maher et al. 2007 ; Holmes et al. 2006 ). However, in the present case, the high degree of overlapping peaks and aggregate integral background found in many biofluid samples challenges the statistical methodologies. We note in this regard the Pearson Product Moment analysis of taurine in a population of human urine samples described in Tables 2 and and3. 3 . Reduction of this background signal prior to sophisticated statistical analysis appears to be highly useful.
Conclusions
In this paper we have shown that 1D selective TOCSY measurements remove the integral background that is intrinsically present in the bucket integrals of 1D proton NMR spectra due to the overlap of unresolved peaks from the great many chemical constituents that occur in biofluids. Thus, 1D TOCSY read peak integrals are more accurate measures of the true variances of statistically significant metabolite concentrations in a biofluid sample population than the bucket integrals of 1D proton spectra. While the use of modeling using standard compound spectra can improve quantitation, the presence of such background signals still complicates the analysis and causes errors. We have also shown that using 1D TOCSY data as inputs to PCA calculations performed on a population of human urine samples demonstrates that substituting 1D TOCSY read peak integrals for the corresponding 1D proton bucket integrals produces better subpopulation cluster resolution, even when this substitution is limited to only one statistically significant metabolite.
Acknowledgments
This work was supported by the NIH (NIGMS R01GM085291-02 and 3R01GM085392-02S1). DR is a member of the Purdue University Center for Cancer Research and the Oncological Sciences Center in Discover Park at Purdue University.
Contributor Information
Peter Sandusky, Department of Chemistry, Eckerd College, St. Petersburg, FL 33711, USA.
Emmanuel Appiah-Amponsah, Department of Chemistry, Purdue University, West Lafayette, IN 47907, USA.
Daniel Raftery, Department of Chemistry, Purdue University, West Lafayette, IN 47907, USA.
- Bauer C, Freeman R, Frenkiel T, Keeler J, Shaka AJ. Gaussian pulses. J Magn Res. 1984; 58 :442–457. [ Google Scholar ]
- Bax A, Davis DG. MLEV-17-based two-dimensional homonuclear magnetization transfer spectroscopy. J Magn Res. 1985; 65 :355–360. [ Google Scholar ]
- Belton PS, Colquhoun IJ, Kemsley EK, Delgadillo I, Roma P, Dennis MJ, Sharman M, Holmes E, Nicholson JK, Spraul M. Application of chemometrics to the 1 H NMR spectra of apple juices: discrimination between apple varieties. Food Chem. 1998; 61 :207–213. [ Google Scholar ]
- Blaise BJ, Shintu L, Elena B, Emsley L, Dumas ME, Toulhoat P. Statistical recoupling prior to significance testing in nuclear magnetic resonance based metabonomics. Anal Chem. 2009; 81 :6242–6251. [ PubMed ] [ Google Scholar ]
- Cloarec O, Dumas ME, Craig A, Barton RH, Trygg J, Hudson J, Blancher C, Gauguier D, Lindon JC, Holmes E, Nicholson JK. Statistical total correlation spectroscopy: an exploratory approach for latent biomarker identification from metabolic 1 H NMR data sets. Anal Chem. 2005; 77 :1282–1289. [ PubMed ] [ Google Scholar ]
- Facke T, Berger S. Application of pulsed field gradients in an improved selective TOCSY experiment. J Magn Res Ser A. 1995; 113 :257–259. [ Google Scholar ]
- Fernie AR, Trethewey RN, Krotzky AJ, Willmitzer L. Metabolite profiling: from diagnostics to systems biology. Nature Rev Mol Cell Biol. 2004; 5 :763–769. [ PubMed ] [ Google Scholar ]
- Fiehn O, Kopka J, Dormann P, Altmann T, Trethewey RN, Willmitzer L. Metabolite profiling for plant functional genomics. Nat Biotechnol. 2000; 18 :1157–1161. [ PubMed ] [ Google Scholar ]
- Fischer S. Agilent Technologies. Personal communication. 2010 [ Google Scholar ]
- Foxall PJD, Parkinson JA, Sadler IH, Lindon JC, Nicholson JK. Analysis of biological fluids using 600 MHz proton NMR spectroscopy: application of homonuclear two-dimensional J-resolved spectroscopy to urine and blood plasma for spectral simplification and assignment. J Pharm Biomed Anal. 1993; 11 :21–31. [ PubMed ] [ Google Scholar ]
- Geen H, Freeman R. Band-selective radiofrequency pulses. J Magn Res. 1991; 93 :93–141. [ Google Scholar ]
- Geen H, Wimperis S, Freeman R. Band-selective pulses without phase distortion. A simulated annealing approach. J Magn Res. 1989; 85 :620–627. [ Google Scholar ]
- Gowda GAN, Zhang SC, Gu HW, Asiago V, Shanaiah N, Raftery D. Metabolomics-based methods for early disease diagnostics: a review. Expert Rev Mol Diagn. 2008; 8 :617–633. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Halouska S, Powers R. Negative impact of noise on the principal component analysis of NMR data. J Magn Res. 2006; 178 :88–95. [ PubMed ] [ Google Scholar ]
- Holmes E, Cloarec O, Nicholson JK. Probing latent biomarker signatures and in vivo pathway activity in experimental disease states via statistical total correlation spectroscopy (STOCSY) of biofluids: application to HgCl2 toxicity. J Proteome Res. 2006; 5 :1313–1320. [ PubMed ] [ Google Scholar ]
- Holmes E, Loo RL, Cloaree O, Coen M, Tang H, Maibaum E, Bruce S, Bruce S, Chan Q, Elliott P, Stamler J, Wilson ID, Lindon JC, Nicholson JK. Detection of urinary drug metabolite (xenometabolome) signatures in molecular epidemiology studies via statistical total correlation (NMR) spectroscopy. Anal Chem. 2007; 79 :2629–2640. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Kadkhodaie M, Rivas O, Tan M, Mohebbi A, Shaka AJ. Broadband homonuclear cross polarization using flip-flop spectroscopy. J Magn Res. 1991; 91 :437–443. [ Google Scholar ]
- Kell DB. Metabolomics and systems biology: making sense of the soup. Curr Opin Microbiol. 2004; 7 :296–307. [ PubMed ] [ Google Scholar ]
- Kessler H, Oschkinat H, Griesinger C. Transformation of homonuclear two-dimensional NMR techniques into one-dimensional techniques using Gaussian pulses. J Magn Res. 1986; 70 :106–113. [ Google Scholar ]
- Maher AD, Zirah SFM, Holmes E, Nicholson JK. Experimental and analytical variation in human urine in 1H NMR spectroscopy-based metabolic phenotyping studies. Anal Chem. 2007; 79 :5204–5211. [ PubMed ] [ Google Scholar ]
- Nicholson JK, Foxall PJD, Spraul M, Farrant RD, Lindon JC. 750 MHz 1 H and 1 H– 13 C NMR spectroscopy of human blood plasma. Anal Chem. 1995; 67 :793–811. [ PubMed ] [ Google Scholar ]
- Nicholson JK, Lindon JC, Holmes E. “Metabonomics”: understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica. 1999; 29 :1181–1189. [ PubMed ] [ Google Scholar ]
- Pan Z, Raftery D. Comparing and combining NMR spectroscopy and mass spectrometry in metabolomics. Anal Bioanal Chem. 2007; 387 :525–527. [ PubMed ] [ Google Scholar ]
- Sandusky P, Raftery D. Use of selective TOCSY NMR experiments for quantifying minor components in complex mixtures: application to the metabonomics of amino acids in honey. Anal Chem. 2005a; 77 :2455–2463. [ PubMed ] [ Google Scholar ]
- Sandusky P, Raftery D. Use of semiselective TOCSY and the pearson correlation for the metabonomic analysis of biofluid mixtures: application to urine. Anal Chem. 2005b; 77 :7717–7723. [ PubMed ] [ Google Scholar ]
- Serkova NJ, Niemann CU. Pattern recognition and biomarker validation using quantitative 1 H-NMR-based metabolomics. Expert Rev Mol Diagn. 2006; 6 :717–731. [ PubMed ] [ Google Scholar ]
- Shaka AJ, Lee CJ, Pines A. Iterative scheme for bilinear operators; application to spin decoupling. J Magn Res. 1988; 77 :274–293. [ Google Scholar ]
- Sorensen OW, Rance M, Ernst RR. z filter for purging phase—or multiplet-distorted spectra. J Magn Res. 1984; 56 :527–534. [ Google Scholar ]
- Stott K, Stonehouse J, Keeler J, Hwang TL, Shaka AJ. Excitation sculpting in high-resolution nuclear magnetic resonance spectroscopy: application to selective NOE experiments. J Am Chem Soc. 1995; 117 :4199–4200. [ Google Scholar ]
- Van der Greef J, Smilde AK. Symbiosis of chemometrics and metabolomics: past, present and future. J Chemometr. 2005; 19 :376–386. [ Google Scholar ]
- Van Dien S, Schilling CH. Bringing metabolomics data into the forefront of systems biology. Mol Syst Biol. 2006; 2 :1–2. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Weljie AM, Newton J, Mercier P, Carlson E, Slupsky CM. Targeted profiling: quantitative analysis of 1 H NMR metabolomics data. Anal Chem. 2006; 78 :4430–4442. [ PubMed ] [ Google Scholar ]
- Ye T, Mo H, Shanaiah N, Gowda GAN, Zhang S, Raftery D. Chemoselective 15 N tag for sensitive and high resolution nuclear magnetic resonance profiling of carboxyl-containing metabolome. Anal Chem. 2009; 81 :4882–4888. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Zhang F, Bruschweiler R. Indirect covariance NMR spectroscopy. J Am Chem Soc. 2004; 126 :13180–13181. [ PubMed ] [ Google Scholar ]
- Zhang F, Bruschweiler R. Robust deconvolution of complex mixtures by covariance TOCSY spectroscopy. Angew Chem Int Ed. 2007; 46 :2639–2642. [ PubMed ] [ Google Scholar ]
- Zhang S, Zheng C, Lanza IR, Nair KS, Raftery D, Vitek O. Interdependence of signal processing and analysis of urine 1 H NMR spectra for metabolic profiling. Anal Chem. 2009; 81 :6080–6088. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Zhang S, Gowda GAN, Asiago V, Ye T, Raftery D. Advances in NMR-based biofluid analysis and metabolite profiling. Analyst. 2010; 135 :1490–1498. [ PMC free article ] [ PubMed ] [ Google Scholar ]
Use of optimized 1D TOCSY NMR for improved quantitation and metabolomic analysis of biofluids
- Published: 10 March 2011
- Volume 49 , pages 281–290, ( 2011 )
Cite this article

- Peter Sandusky 1 ,
- Emmanuel Appiah-Amponsah 2 &
- Daniel Raftery 2
927 Accesses
30 Citations
Explore all metrics
One dimensional selective TOCSY experiments have been shown to be advantageous in providing improved data inputs for principle component analysis (PCA) (Sandusky and Raftery 2005a , b ). Better subpopulation cluster resolution in the observed scores plots results from the ability to isolate metabolite signals of interest via the TOCSY based filtering approach. This report reexamines the quantitative aspects of this approach, first by optimizing the 1D TOCSY experiment as it relates to the measurement of biofluid constituent concentrations, and second by comparing the integration of 1D TOCSY read peaks to the bucket integration of 1D proton NMR spectra in terms of precision and accuracy. This comparison indicates that, because of the extensive peak overlap that occurs in the 1D proton NMR spectra of biofluid samples, bucket integrals are often far less accurate as measures of individual constituent concentrations than 1D TOCSY read peaks. Even spectral fitting approaches have proven difficult in the analysis of significantly overlapped spectral regions. Measurements of endogenous taurine made over a sample population of human urine demonstrates that, due to background signals from other constituents, bucket integrals of 1D proton spectra routinely overestimate the taurine concentrations and distort its variation over the sample population. As a result, PCA calculations performed using data matrices incorporating 1D TOCSY determined taurine concentrations produce better scores plot subpopulation cluster resolution.
This is a preview of subscription content, log in via an institution to check access.
Access this article
Subscribe and save.
- Get 10 units per month
- Download Article/Chapter or eBook
- 1 Unit = 1 Article or 1 Chapter
- Cancel anytime
Price includes VAT (Russian Federation)
Instant access to the full article PDF.
Rent this article via DeepDyve
Institutional subscriptions
Similar content being viewed by others

NMR-Based Metabolomics of Oral Biofluids

Liquid Chromatography-Mass Spectrometry of Biofluids and Extracts

Exploratory GC/MS-Based Metabolomics of Body Fluids
Bauer C, Freeman R, Frenkiel T, Keeler J, Shaka AJ (1984) Gaussian pulses. J Magn Res 58:442–457
Google Scholar
Bax A, Davis DG (1985) MLEV-17-based two-dimensional homonuclear magnetization transfer spectroscopy. J Magn Res 65:355–360
Belton PS, Colquhoun IJ, Kemsley EK, Delgadillo I, Roma P, Dennis MJ, Sharman M, Holmes E, Nicholson JK, Spraul M (1998) Application of chemometrics to the 1 H NMR spectra of apple juices: discrimination between apple varieties. Food Chem 61:207–213
Article Google Scholar
Blaise BJ, Shintu L, Elena B, Emsley L, Dumas ME, Toulhoat P (2009) Statistical recoupling prior to significance testing in nuclear magnetic resonance based metabonomics. Anal Chem 81:6242–6251
Cloarec O, Dumas ME, Craig A, Barton RH, Trygg J, Hudson J, Blancher C, Gauguier D, Lindon JC, Holmes E, Nicholson JK (2005) Statistical total correlation spectroscopy: an exploratory approach for latent biomarker identification from metabolic 1 H NMR data sets. Anal Chem 77:1282–1289
Facke T, Berger S (1995) Application of pulsed field gradients in an improved selective TOCSY experiment. J Magn Res Ser A 113:257–259
Fernie AR, Trethewey RN, Krotzky AJ, Willmitzer L (2004) Metabolite profiling: from diagnostics to systems biology. Nature Rev Mol Cell Biol 5:763–769
Fiehn O, Kopka J, Dormann P, Altmann T, Trethewey RN, Willmitzer L (2000) Metabolite profiling for plant functional genomics. Nat Biotechnol 18:1157–1161
Fischer S (2010) Agilent Technologies. Personal communication
Foxall PJD, Parkinson JA, Sadler IH, Lindon JC, Nicholson JK (1993) Analysis of biological fluids using 600 MHz proton NMR spectroscopy: application of homonuclear two-dimensional J-resolved spectroscopy to urine and blood plasma for spectral simplification and assignment. J Pharm Biomed Anal 11:21–31
Geen H, Freeman R (1991) Band-selective radiofrequency pulses. J Magn Res 93:93–141
Geen H, Wimperis S, Freeman R (1989) Band-selective pulses without phase distortion. A simulated annealing approach. J Magn Res 85:620–627
Gowda GAN, Zhang SC, Gu HW, Asiago V, Shanaiah N, Raftery D (2008) Metabolomics-based methods for early disease diagnostics: a review. Expert Rev Mol Diagn 8:617–633
Halouska S, Powers R (2006) Negative impact of noise on the principal component analysis of NMR data. J Magn Res 178:88–95
Article ADS Google Scholar
Holmes E, Cloarec O, Nicholson JK (2006) Probing latent biomarker signatures and in vivo pathway activity in experimental disease states via statistical total correlation spectroscopy (STOCSY) of biofluids: application to HgCl2 toxicity. J Proteome Res 5:1313–1320
Holmes E, Loo RL, Cloaree O, Coen M, Tang H, Maibaum E, Bruce S, Bruce S, Chan Q, Elliott P, Stamler J, Wilson ID, Lindon JC, Nicholson JK (2007) Detection of urinary drug metabolite (xenometabolome) signatures in molecular epidemiology studies via statistical total correlation (NMR) spectroscopy. Anal Chem 79:2629–2640
Kadkhodaie M, Rivas O, Tan M, Mohebbi A, Shaka AJ (1991) Broadband homonuclear cross polarization using flip-flop spectroscopy. J Magn Res 91:437–443
Kell DB (2004) Metabolomics and systems biology: making sense of the soup. Curr Opin Microbiol 7:296–307
Kessler H, Oschkinat H, Griesinger C (1986) Transformation of homonuclear two-dimensional NMR techniques into one-dimensional techniques using Gaussian pulses. J Magn Res 70:106–113
Maher AD, Zirah SFM, Holmes E, Nicholson JK (2007) Experimental and analytical variation in human urine in 1H NMR spectroscopy-based metabolic phenotyping studies. Anal Chem 79:5204–5211
Nicholson JK, Foxall PJD, Spraul M, Farrant RD, Lindon JC (1995) 750 MHz 1 H and 1 H– 13 C NMR spectroscopy of human blood plasma. Anal Chem 67:793–811
Nicholson JK, Lindon JC, Holmes E (1999) “Metabonomics”: understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica 29:1181–1189
Pan Z, Raftery D (2007) Comparing and combining NMR spectroscopy and mass spectrometry in metabolomics. Anal Bioanal Chem 387:525–527
Sandusky P, Raftery D (2005a) Use of selective TOCSY NMR experiments for quantifying minor components in complex mixtures: application to the metabonomics of amino acids in honey. Anal Chem 77:2455–2463
Sandusky P, Raftery D (2005b) Use of semiselective TOCSY and the pearson correlation for the metabonomic analysis of biofluid mixtures: application to urine. Anal Chem 77:7717–7723
Serkova NJ, Niemann CU (2006) Pattern recognition and biomarker validation using quantitative 1 H-NMR-based metabolomics. Expert Rev Mol Diagn 6:717–731
Shaka AJ, Lee CJ, Pines A (1988) Iterative scheme for bilinear operators; application to spin decoupling. J Magn Res 77:274–293
Sorensen OW, Rance M, Ernst RR (1984) z filter for purging phase—or multiplet-distorted spectra. J Magn Res 56:527–534
Stott K, Stonehouse J, Keeler J, Hwang TL, Shaka AJ (1995) Excitation sculpting in high-resolution nuclear magnetic resonance spectroscopy: application to selective NOE experiments. J Am Chem Soc 117:4199–4200
Van der Greef J, Smilde AK (2005) Symbiosis of chemometrics and metabolomics: past, present and future. J Chemometr 19:376–386
Van Dien S, Schilling CH (2006) Bringing metabolomics data into the forefront of systems biology. Mol Syst Biol 2:1–2
Weljie AM, Newton J, Mercier P, Carlson E, Slupsky CM (2006) Targeted profiling: quantitative analysis of 1 H NMR metabolomics data. Anal Chem 78:4430–4442
Ye T, Mo H, Shanaiah N, Gowda GAN, Zhang S, Raftery D (2009) Chemoselective 15 N tag for sensitive and high resolution nuclear magnetic resonance profiling of carboxyl-containing metabolome. Anal Chem 81:4882–4888
Zhang F, Bruschweiler R (2004) Indirect covariance NMR spectroscopy. J Am Chem Soc 126:13180–13181
Zhang F, Bruschweiler R (2007) Robust deconvolution of complex mixtures by covariance TOCSY spectroscopy. Angew Chem Int Ed 46:2639–2642
Zhang S, Zheng C, Lanza IR, Nair KS, Raftery D, Vitek O (2009) Interdependence of signal processing and analysis of urine 1 H NMR spectra for metabolic profiling. Anal Chem 81:6080–6088
Zhang S, Gowda GAN, Asiago V, Ye T, Raftery D (2010) Advances in NMR-based biofluid analysis and metabolite profiling. Analyst 135:1490–1498
Download references
Acknowledgments
This work was supported by the NIH (NIGMS R01GM085291-02 and 3R01GM085392-02S1). DR is a member of the Purdue University Center for Cancer Research and the Oncological Sciences Center in Discover Park at Purdue University.
Author information
Authors and affiliations.
Department of Chemistry, Eckerd College, St. Petersburg, FL, 33711, USA
Peter Sandusky
Department of Chemistry, Purdue University, West Lafayette, IN, 47907, USA
Emmanuel Appiah-Amponsah & Daniel Raftery
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Daniel Raftery .
Rights and permissions
Reprints and permissions
About this article
Sandusky, P., Appiah-Amponsah, E. & Raftery, D. Use of optimized 1D TOCSY NMR for improved quantitation and metabolomic analysis of biofluids. J Biomol NMR 49 , 281–290 (2011). https://doi.org/10.1007/s10858-011-9483-7
Download citation
Received : 11 October 2010
Accepted : 10 December 2010
Published : 10 March 2011
Issue Date : April 2011
DOI : https://doi.org/10.1007/s10858-011-9483-7
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Metabolomics
- Metabolite profiling
- Quantitative analysis
- Find a journal
- Publish with us
- Track your research
Chemical Communications
Single-scan ultra-selective 1d total correlation spectroscopy †.

* Corresponding authors
a Department of Chemistry, University of Manchester, Oxford Road, Manchester, UK E-mail: [email protected]
Selective 1D TOCSY is a powerful tool in the assignment of NMR spectra of organic molecules. Here an order of magnitude improvement in selectivity, allowing overlapping multiplets to be excited separately, is achieved in a single scan using the very recent GEMSTONE method. The new experiment is illustrated using an antibiotic and a mixture of diastereomers.
Supplementary files
- Supplementary information PDF (1530K)

Article information
Download citation, permissions.
Single-scan ultra-selective 1D total correlation spectroscopy
P. Kiraly, M. Nilsson, G. A. Morris and R. W. Adams, Chem. Commun. , 2021, 57 , 2368 DOI: 10.1039/D0CC08033K
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page .
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page .
Read more about how to correctly acknowledge RSC content .
Social activity
Search articles by author, advertisements.
Duke NMR HELP CENTER
Varian – 1D TOCSY
1h – in a tocsy spectrum, magnetization is allowed to pass from one proton to another that is 3 bonds or less away, and to take such steps repeatedly. thus, magnetization moves from any one proton to all others throughout the spin system of which it is a part. the one caveat of this, is that the proton must be attached to a carbon. furthermore, if the magnetization comes upon a non-carbon or a quaternary carbon the tocsy phenomena stops. this is a great tool for learning which protons are connected through bonds (within a spin system). the selectivity of the tocsy1d sequence is based on a pair of gradient echoes employing selective inversion pulses that will invert the resonance of interest, so that it is rephased by the second of each pair of gradients., commands to be typed in blue ; commands to click on in copper ..
| is powered by WordPress. Read the Sites@Duke Express and , or . |
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Open access
- Published: 21 October 2020
Sensitivity enhancement of homonuclear multidimensional NMR correlations for labile sites in proteins, polysaccharides, and nucleic acids
- Mihajlo Novakovic 1 ,
- Ēriks Kupče 2 ,
- Andreas Oxenfarth 3 ,
- Marcos D. Battistel 4 ,
- Darón I. Freedberg ORCID: orcid.org/0000-0003-1991-5174 4 ,
- Harald Schwalbe 3 &
- Lucio Frydman ORCID: orcid.org/0000-0001-8208-3521 1
Nature Communications volume 11 , Article number: 5317 ( 2020 ) Cite this article
4805 Accesses
20 Citations
4 Altmetric
Metrics details
- Biophysical chemistry
- Solution-state NMR
- Structural biology
Multidimensional TOCSY and NOESY are central experiments in chemical and biophysical NMR. Limited efficiencies are an intrinsic downside of these methods, particularly when targeting labile sites. This study demonstrates that the decoherence imparted on these protons through solvent exchanges can, when suitably manipulated, lead to dramatic sensitivity gains per unit time in the acquisition of these experiments. To achieve this, a priori selected frequencies are encoded according to Hadamard recipes, while concurrently subject to looped selective inversion or selective saturation procedures. Suitable processing then leads to protein, oligosaccharide and nucleic acid cross-peak enhancements of ≈200–1000% per scan, in measurements that are ≈10-fold faster than conventional counterparts. The extent of these gains will depend on the solvent exchange and relaxation rates of the targeted sites; these gains also benefit considerably from the spectral resolution provided by ultrahigh fields, as corroborated by NMR experiments at 600 MHz and 1 GHz. The mechanisms underlying these experiments’ enhanced efficiencies are analyzed on the basis of three-way polarization transfer interplays between the water, labile and non-labile protons, and the experimental results are rationalized using both analytical and numerical derivations. Limitations as well as further extensions of the proposed methods, are also discussed.
Similar content being viewed by others
Relative configuration of micrograms of natural compounds using proton residual chemical shift anisotropy
Room-temperature dynamic nuclear polarization enhanced NMR spectroscopy of small biological molecules in water
SHARPER-DOSY: Sensitivity enhanced diffusion-ordered NMR spectroscopy
Introduction.
Two-dimensional (2D) homonuclear NMR correlations 1 , 2 are an integral part of the tools used to elucidate the structure and dynamics of organic, and biological molecules 3 , 4 . These correlations can be mediated by chemical exchange or nuclear overhauser effects (NOEs) 5 , 6 , and are probed by monitoring how polarization from one spin reservoir travels to another via dipolar interactions or chemical kinetics 7 , 8 , 9 , 10 , 11 , 12 . Magnetization transfer (MT) within a J -coupled spin network as achieved by TOtal Corelation SpectroscopY (TOCSY) 13 , 14 , leads to complementary information based on bond connectivities. Despite being routinely performed these 2D NMR experiments, and particularly NOESY, suffer from relatively low efficiencies, leading to cross-peaks with low signal-to-noise ratio (SNR), and to a need for extensive signal averaging times to improve this SNR. Detection of NOESY and TOCSY cross-peaks becomes even more difficult when involving labile protons ( 1 Hs), as information is then “lost” through chemical exchange with the solvent. Hydroxyl 1 Hs in saccharides, amino 1 Hs in proteins and nucleic acids, amide 1 Hs in disordered proteins, and imino 1 Hs in RNA/DNA, are prototypical examples of such challenging systems: when placed in their natural aqueous environment all of these will undergo a rapid exchange with the solvent, which dramatically reduces the efficiency of their intramolecular polarization transfers.
We have recently introduced Looped PROjective SpectroscopY (L-PROSY) 15 , an approach that alleviates these problems by regarding these exchange processes as “resets” within the framework of Anti-Zeno Effects 16 , 17 , 18 . Instead of applying a single mixing period for homonuclear transfers that will then reach kinetically-compromised amplitudes, L-PROSY “freezes” these transfers after they begin to act with their (fastest) initial rate, resets the labile 1 H states to their initial conditions by exploiting their exchange with an unperturbed solvent polarization reservoir, and repeats this process multiple times 19 , 20 . The ensuing “L-PROSY encoding” acts then as a sort of conveyor belt, causing the NOE/TOCSY cross-peaks to grow with the much more favorable rates characterizing their initial buildups, and lasting for as long as either thermodynamic considerations or the recipient’s T 1 will accommodate them, before performing the latter’s signal detection. By selectively addressing only the targeted 1 Hs and avoiding water perturbation L-PROSY exploits some elements of SOFAST NMR 21 , 22 ; at the same time, by its repeated action, it is also reminiscent of CEST-based polarization transfer 23 , 24 , 25 , 26 . Despite their sensitivity gains, L-PROSY experiments are still long, requiring traditional t 1 evolution periods to build-up multidimensional information. L-PROSY acquisitions can also lead to artifacts arising from an incomplete replenishment of the targeted sites’ polarization by the solvent, appearing as harmonics of genuine evolution frequencies and/or as anti-diagonal peaks. The present study demonstrates a new approach capable of alleviating both drawbacks while achieving even more complete MTs, which relies on Hadamard-encoded 27 , 28 , 29 selective polarization transfers from the targeted labile 1 Hs. It is shown that, whether involving multiple selective inversions or a continuous saturation procedure, this provides the highest per-scan enhancements we have seen on either conventionally- or L-PROSY-encoded NOESY and TOCSY experiments involving labile or fast-relaxing 1 Hs. When combined with the compressed-sensing advantages and multiplexing provided by Hadamard encoding, gains of ca . two orders of magnitude in SNR/unit_time were observed over conventional counterparts. These gains were noted for a variety of biomolecular systems including sugars, nucleic acids, and proteins, illustrating their generality. The physical principles underlying these gains are described on the basis of a simple multi-site exchange model, leading to analytical descriptions that are generalized by numerical calculations, and which reproduce well the experimental observations.
Principles of Hadamard-encoded 2D homonuclear correlations on labile sites
Frequency-domain Hadamard spectroscopy has been proposed as a way to replace the conventional t 1 evolution increment of 2D time-domain NMR, by employing a “comb” of frequency-selective (polychromatic) radiofrequency (RF) pulses, which directly address peaks in the F 1 frequency domain 27 , 28 , 29 . If the frequencies of these peaks are known and well separated, 180˚ phase shift manipulations based on Hadamard encoding principles, followed by suitable addition/subtraction manipulations (the Hadamard transform), have been demonstrated as means for speeding up the acquisition of 2D NMR data. The present study brings this compressed-sensing scheme to bear within the framework of MT experiments targeting labile 1 Hs in biomolecules; thanks to continuous repolarizations with a replenishing solvent pool, this is shown to substantially improve the SNR in basic NOESY and TOCSY experiments involving such sites. To implement the resulting Hadamard-encoded MT (HMT) schemes, the original NOESY/TOCSY indirect-domain and mixing-time manipulations, were here replaced by either selective polychromatic saturations or looped polychromatic inversion pulses, addressing solely the fast-exchanging, labile 1 Hs. This leads to the experimental scheme depicted in Fig. 1a , where what we denote as the “HMT block” encodes via an “on” or “off” irradiation, the labile 1 Hs according to a Hadamard scheme. This is done while neither the solvent nor the peaks that will eventually receive polarization from the labile sites, are perturbed. The fact that the large water spin reservoir is not perturbed provides a constant repolarization of the labile protons during the encoding process 30 , 31 , effectively shortening these spins’ T 1 s. Concurrently, the fact that the recipients’ spins are untouched, prolongs the efficiency of an MT operating through cross-relaxation, J -coupling 32 or chemical exchange, up to times lasting on the order of the latter spins’ T 1 . The resulting asymmetry leads to potentially significant SNR/unit_time gains, as illustrated in Fig. 1b with overlaid conventional and HMT-encoded NOESY and TOCSY spectra addressing the hydroxyl sites of myo-inositol, a prototypical saccharide. It follows that whereas in conventional experiments the exchange that labile 1 Hs undergo with the solvent prevent efficient intramolecular spin-coupling/relaxation transfers, in MT the exchange does the opposite—enhancing correlations and magnifying the labile sites’ cross-peaks.
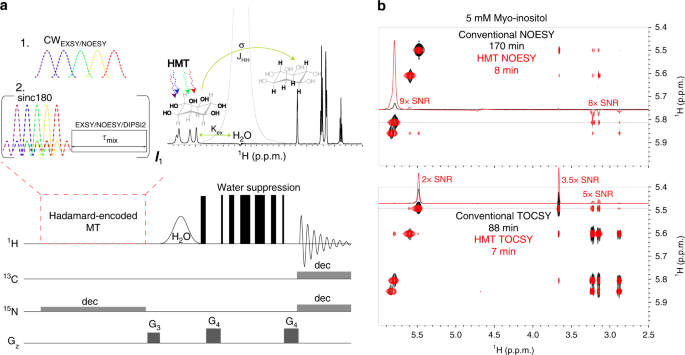
a HMT pulse sequence illustrating the two types of perturbation procedures utilized here for improving the efficiency of homonuclear 2D correlations involving labile sites. These were Hadamard-encoded selective polychromatic saturations, or looped Hadamard-encoded polychromatic inversions (assumed here imparted by sinc pulses) followed by a delay for NOESY/EXSY transfer or by DIPSI2 13 to achieve TOCSY’s isotropic mixing (selective saturations can be used only for the NOESY/EXSY transfers; repeated inversions work for all). During these long MT processes, illustrated here for myo-inositol, a three-way polarization transfer is effectively established, where water constantly repolarizes the targeted labile 1 Hs, enabling a prolonged magnetization transfer to the non-labile counterparts. “dec” refers to GARP4 68 or adiabatic 69 decoupling used during the encoding and the acquisition for labeled samples; water suppression was achieved using excitation sculpting 70 or WATERGATE 3919 71 , 62 , 63 schemes. b Comparisons between conventional NOESY (80 ms mixing) and TOCSY (48 ms DIPSI2 mixing) spectra acquired on myo-inositol, and Hadamard MT counterparts (NOESY obtained using 800 ms CW mixing; TOCSY with 12 loops of 24 ms DIPSI2 mixing). Note the different acquisition times required (≈10–20× faster in the HMT scheme), and the substantial (2–9×) enhancements of the cross- and diagonal peaks. Spectra were acquired at 600 MHz on a Bruker Avance III spectrometer equipped with a Prodigy probe.
The mechanism underlying the resulting enhancements is reminiscent to that in in vivo CEST NMR 24 , 33 , 34 , 35 , and HMT enhancement factors ε vs NOESY/TOCSY can be computed using a similar theoretical framework. For concreteness, we consider here a NOESY spin dynamics as depicted by the Bloch-McConnell-Solomon equations, incorporating a continuous saturating field targeting a labile 1 H; these are then used to evaluate the magnetization build-up arising from labile→non-labile 1 H cross-relaxation-driven transfers in MT and conventional NOESY scenarios 24 , 33 , 36 , 37 . These calculations are detailed in the Supplementary Information ; as shown in Supplementary Equations ( 1 – 5) , the steady-state solutions describing the MT from the labile 1 H’s equilibrium magnetization \(M_{z_l}^0\) into the non-labile 1 H state \(m_{z_{nl}}^{MT_{ss}}\) can, for a cross-relaxation rate σ and saturating field ν 1 , be approximated as
where l and nl subscripts depict parameters for the labile and non-labile 1 Hs, \(p = R_{2_l} + k_{ex}^l - \frac{{k_{ex}^l \ast k_{ex}^w}}{{R_{2_w} + k_{ex}^w}}\) and \(q = \Big( {R_{1_l} + k_{ex}^l + \sigma - \frac{{k_{ex}^l \ast k_{ex}^w}}{{R_{1_w} + k_{ex}^w}}} \Big)(R_{1_{nl}} + \sigma ) - \sigma ^2\) are quantities depending on the exchange rates \(k_{ex}^l\) and \(k_{ex}^w\) between the labile site and the water reservoir (scaled by their population ratio), as well as on the longitudinal and transverse relaxation rates \(R_{1(2)} = 1{\mathrm{/}}T_{1(2)}\) of the labile, non-labile and water 1 Hs. Similar differential Bloch-McConnell-Solomon equations have been solved analytically for the conventional NOESY experiment 8 ; these solutions lead to symmetric \(M_{z_l} \to M_{z_{nl}}\) , \(M_{z_{nl}} \to M_{z_l}\) cross-peaks, reaching maximal values given by:
where \( \lambda _ \pm = \frac{1}{2}\left\{ {(R_{1_l} + R_{1_{nl}} + 2\sigma + k_{ex}^l) \pm \sqrt {(R_{1_l} + k_{ex}^l - R_{1_{nl}})^2 + 4\sigma ^2} } \right\}\) , and other symbols are as introduced above (see Supplementary Equations ( 6 – 10) , for a derivation of these maxima conditions). For the labile 1 H scenario of interest here we can assume that \(k_{ex}^l \gg R_{1l},R_{1nl},\sigma\) ; this greatly simplifies Eqs. ( 1 ) and ( 2 ), leading to MT vs conventional NOESY enhancement factors
Notice that according to this model, the enhancement of the labile 1 H’s signature will, for an efficient \(\nu _1 \,> \,\,k_{ex}^l\) saturation scenario, be given by \(k_{ex}^lT_{1_{nl}}\) . This represents the number of exchanges that labile 1 Hs can imprint on their cross-relaxing non-labile neighbors given the latter’s “memory time”, and is reminiscent of similar behaviors derived for CEST 24 , 35 . More complete analyses of the predictions deriving from this Bloch-McConnell-Solomon model are presented in the Supplementary Information , including numerical solutions of the labile→non-labile transfer upon selectively perturbing the labile spins’ for a variety of solvent exchange rates, couplings and cross-relaxation rates. Supplementary Figures 1 – 4 confirm that substantial sensitivity improvements may arise from this approach, and examine their dependence on irradiation strength, solvent exchange rate and molecular correlation time. Additional insight is provided by Supplementary Fig. 5 , which compares the conceptually similar but practically very different L-PROSY experiment to the current HMT proposal, highlighting the advantages of the latter experiment.
HMT and the effects of increased magnetic field strengths
Like CEST 34 , 38 , 39 , HMT-based methods that target labile 1 Hs could benefit significantly from operating at the highest possible magnetic fields. Under these conditions (i) more rapid exchange rates \(k_{ex}^l\) can be accommodated, leading to higher enhancements ε without resolution penalties (in ppm); (ii) resolution between sites improves, leading to more facile conditions for implementing the 1D Hadamard encoding while relying on more intense ν 1 saturating fields and/or shorter inversion pulses; (iii) \(T_{1_{nl}}\) relaxation times tend to get longer, facilitating the extent of the intramolecular transfers 40 , 41 , 42 ; and (iv) it becomes generally easier to study the exchanging 1 Hs closer to physiologically-relevant temperatures. These advantages reinforce one another when targeting rapidly exchanging groups like the OHs of saccharides, something demonstrated in Fig. 2 for the α2,8-linked sialic acid tetramer (SiA) 4 . Higher fields improve the 1D spectrum for these sites leading, at 5 °C and 1 GHz, to a nearly full resolution of all hydroxyl 1 Hs in the spectrum (Fig. 2b ). Figure 2c compares conventional and HMT NOESY spectra acquired on (SiA) 4 under such conditions; Supplementary Fig. 6a provides similar data but compared at a lower (600 MHz) field. Correlations involving the hydroxyl protons are hard to discern in the conventional experiments: even at 1 GHz, the fast chemical exchange reduces NOESY correlations between hydroxyl and aliphatic 1 Hs to neighbors that are ≤2 Å away, like IVOH8-IV8 (at 1.7 Å) and IVOH8-IV6 (at 1.9 Å). By contrast, HMT NOESY at 1 GHz (and despite the challenges of selective saturation at lower fields, even at 600 MHz) provides a wealth of cross-peaks, in a fraction of the time needed by its conventional counterpart. The fact that in the conventional TOCSY and NOESY spectra many of the cross-peaks are invisible even after protracted acquisitions complicates the quantification of the experimental enhancements; still, Table 1 provides lower bounds for these values. The newly observed correlations involve hydroxyls with a wide rate of solvent exchange rates –ranging from ~10 s −1 for IVOH8, to ~40 s −1 for IIOH7 and even to ~100 s −1 for IIOH9 43 . The reason for this efficiency is as explained earlier: faster exchanges hurt cross-relaxation, but at the same time, supply fresh polarization for the repeated transfer of magnetization (see for instance Supplementary Figs. 1 – 3 ). As for the lineshapes displayed by the 2D HMT plots: these are somewhat artificial, as in the Hadamard processing cross-peak traces were simply placed at the frequency positions used for implementing the selective RF irradiation/encoding. In principle, however, the information in the 2D HMT correlations has a well-defined point-spread function, given by 1D absorptive lineshapes along the direct domain, and by T 1 -, T 2 -, and ν 1 -dependent combinations along the indirect-domain axis. Once again, an analogy to what happens in CEST allows one to compute how these parameters influence the effective F1 spectral peak width 24 , 35 : this will be given by \(\nu _{1/2} \approx \sqrt {\nu _1^2\frac{p}{q} + p^2}\) , where the p and q symbols are as introduced in relation to Eq. ( 1 ). Notice that although line-broadening effects are introduced by the saturating RF into these spectral lineshapes, these are less critical than what they may appear at first sight—primarily owing to the labile nature of the 1 Hs whose 2D correlations these new experiments target. This lability leads to exchange-broadened 1D linewidths, whose spectral resolution is not compromised much further by the RF used in their HMT encoding. In fact, one of the features of HMT is its ability to tailor the saturating field or the inversion bandwidth used in its implementation to each spectral peak individually, enabling an optimal compromise between resolution and the extraction of the information being sought.
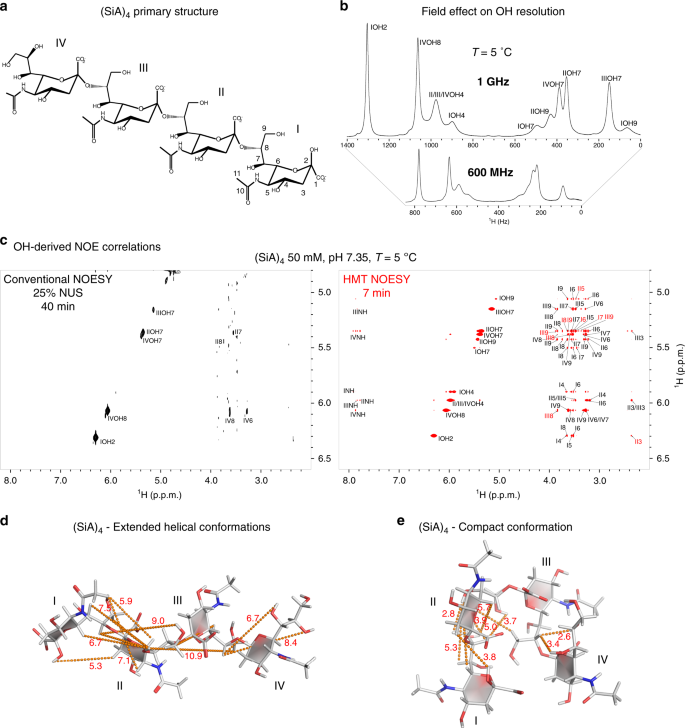
a Primary structure of (SiA) 4 , depicting the rings’ numbering and the multiple hydroxyl and amide labile 1 Hs. b Magnetic field effect on the appearance of (SiA) 4 ’s hydroxyl 1 H spectrum. Note how the chemical exchange broadens certain hydroxyl resonances at 600 MHz, whereas at 1 GHz most of the peaks are well-resolved, thanks to field-induced separation of the exchanging sites. c Comparison between a conventional 2D NOESY acquired using 25% non-uniform sampling for a minimal acquisition time with faithful spectral reconstruction, and Hadamard MT NOESY collected with a 600 ms saturation period. Although conventional NOESY only reveals some of the closest neighbors, richer information (coupled to much shorter acquisitions) is provided by the HMT. These 1 GHz spectra were acquired using a Bruker Avance Neo console equipped with a TCI cryoprobe; Table 1 summarizes some of the enhancements observed for this compound. d Conflicts arising between the cross-relaxation peaks observed in the HMT NOESY spectrum (highlighted in c with red fonts and indicated here by dashed lines) and the extended structure proposed for this homopolymer at −10 °C based on non-labile 1 H NOEs 47 . Notice the long distances (in Å) indicated by the dashed lines for some of the new NOESY-derived experimental connectivities. e Revised, compact structure, compatible with the labile 1 H’s NOEs, showing substantial distance shortenings.
Based on a previous (SiA) 4 study performed at lower fields under super-cooled conditions 44 , the new data arising from HMT NOESY and TOCSY experiments can be used for both assignments and for structural refinements. The latter data are illustrated in Supplementary Fig. 6b , showing a number of sizable intra-residue J-driven correlations (Table 1 ); in fact, these TOCSY enhancements are often larger than their NOESY counterparts, reflecting perhaps the slightly higher temperatures at which they were measured. The HMT NOESY experiments also reveal correlations that had so far remained unseen, including inter-residue cross-peaks between IOH9-II5/II6, IIOH7-I6/I7/I8/I9, IIOH9-I8, IVOH7-III8, IVOH8-III8. When considering the structure proposed for the glycan under super-cooled conditions 44 a number of conflicts arise, as, according to it, some of the cross-peaks observed by HMT NOESY would then correspond to internuclear distances between 1 Hs positioned ≈7–9 Å apart (yellow dashes in Fig. 2d ). As no strong additional cross-peaks that could justify such long-distance correlations on the basis of a relaying mechanism are visible, this called for a refinement of the glycan’s structure. Further investigations to be discussed in further detail separately evidence that, with temperature, (SiA) 4 may undergo a rearrangement; this rearranges the oligosaccharide into the average structure shown in Fig. 2e , making it compatible with the observed HMT NOESY cross-peaks.
Amide, amino, and imino proton correlations in proteins and nucleic acids
HMT at ultrahigh magnetic field turns out to be especially informative when implemented on the imino protons of nucleic acids. At 1 GHz these imino resonances, which can be broadened by chemical exchange with the solvent at lower fields, are sharper and better resolved. Figure 3 shows the superiority of the HMT experiment over conventional NOESY for detecting cross-peaks involving imino resonances, utilizing a 14mer hairpin RNA as prototypical example. This can be visualized by comparing 1D traces extracted from the conventional and HMT NOESY spectra (Fig. 3 ). Notice that several of the correlations are only detected in the HMT experiment, especially for iminos arising from bases that, like U7 and U8, are positioned in loop regions that undergo facile solvent exchanges. The wealth of peaks in the HMT experiment facilitates the elucidation of peaks in the amine, aromatic and sugar regions: with these, it is possible to obtain many more spectral assignments than what is possible with conventional experiments. Also illustrated in this Figure is HMT’s ability to fine-tune the extent of the saturation to the rate of solvent exchange for various labile 1 Hs—a potentially important feature for heterogeneous structures, as the tailored excitation helps overcome the fast exchange arising in flexible regions without compromising the resolving power needed to address other, more rigid regions in the RNA. Even for more slowly exchanging iminos, like U11H3, enhancements are significant, both for correlations with aromatic protons that like A4H2 can be clearly detected in the conventional experiment, as well as with labile amino protons that like C10/C3H41, barely yield cross-peaks in the conventional NOESY. A summary of the resulting enhancements for representative peaks is presented in Table 1 . Comparisons of conventional and HMT NOESY experiments for this 14mer were also performed at 600 MHz; although slices extracted from these data for various imino sites also lead to substantial enhancements, the relative enhancements provided by HMT over conventional experiments were larger at 1 GHz than at 600 MHz (Table 1 ). This reflects one of the aforementioned field-derived advantages: at 1 GHz, the inversion/saturation pulses addressing the labile 1 Hs can be stronger, leading to a more efficient MT and to ~25–100% larger enhancements when assessed against their conventionally collected counterparts.
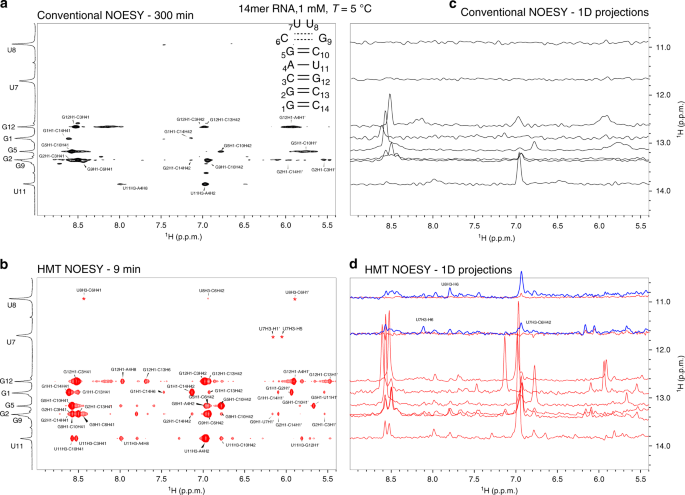
a Conventional (100 ms mixing) vs b HMT NOESY spectra (17 loops with a 20 Hz inversion pulse followed by 40 ms mixing each) recorded on the 14mer on top. Both 2D spectra show the imino resonances placed along the vertical direction; for maximizing its efficiency, however, the conventional spectrum was acquired with the imino resonances along the direct domain, and was flipped in this graph to match the HMT data (otherwise, if encoding the iminos in t 1 , the conventional experiment showed no peaks altogether). As this was an 15 N-labeled sample, looped inversions/mixing periods were preferred over continuous saturations: this avoided a constant 15 N decoupling during the saturation period, which would have led to sample heating. Assignments are as labeled in the spectra; cross peaks labeled with asterisk are not visible at the given contour level. To better appreciate the enhancements, c and d show various 1D slices extracted from a and b , respectively. Shown in blue for the HMT NOESY are traces addressing U7 and U8 with a stronger (40 Hz continuous) saturation HMT scheme, revealing new correlations despite these sites’ >30 s −1 solvent exchange rates. All spectra were acquired at 1 GHz using a Bruker Avance Neo spectrometer equipped with a TCI cryoprobe.
Another advantage of operating at ultrahigh fields is that most amide backbone signals in small and medium-sized proteins become well resolved in 1D experiments, enabling HMT experiments on these N-bound 1 H resonances as well. Figure 4 illustrates the benefits resulting from this with comparisons between conventional and HMT NOESY acquisitions recorded on LA5, a 40-residue protein 45 , and on ubiquitin, a 76-residue protein. As in the case of the RNA, these HMT experiments utilized a polychromatic looped encoding instead of single long saturation pulses, as both of these proteins were 15 N labeled and looping facilitated heteronuclear decoupling during the encoding (sequence in Fig. 1a ). Extracted 1D projections show a 1:1 match between the HMT and conventional spectra, with sensitivity enhancements of ≈2x provided by the faster former scheme. These enhancements are smaller than those observed for saccharides and nucleic acids owing to the slower exchange rates that amide protons exhibit in the structured environments of these proteins; still when combined with fivefold shorter acquisitions, it is clear that HMT also brings substantial gains in SNR/unit_time to proteins (Table 1 ). Even larger enhancements could result in disordered proteins; in such cases, however, limited spectral dispersion would require the incorporation of a third, heteronuclear-encoding dimension in order to resolve the amide peaks. Such experiments will be considered in a separate study.
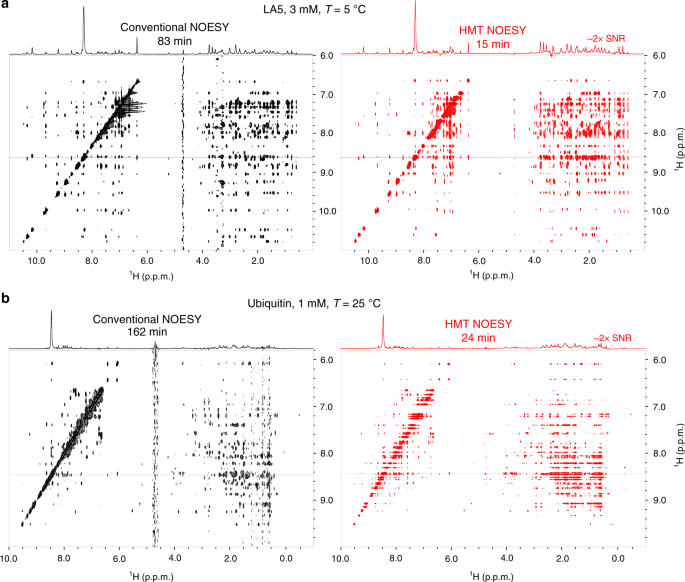
Conventional vs HMT NOESY spectra recorded for LA5 a and ubiquitin b samples. Notice how the 23.5 T field is sufficient to resolve almost entirely the amide/amino resonances in these structured peptides (regions between 6.6–9.5 ppm) and enable fast, highly sensitive NOESY experiments by Hadamard MT. Conventional experiments were acquired with 300 ms and 250 ms mixing, respectively (for maximum NOESY cross-peaks); HMT employed 6 × 150 ms and 6 × 140 ms looped encoding. Spectra were acquired at 1 GHz using a Bruker Avance Neo console equipped with a TCI cryoprobe.
The strong correlations that HMT can deliver also open up the possibility of better exploiting amino groups, whose 1 Hs are sometimes underutilized in NOESY and TOCSY experiments both in proteins and nucleic acids. Figure 5 shows how such 2D correlations can be put to good use, with HMT NOESY examples targeting amino groups in both ubiquitin and in the 14mer RNA sample. Strong correlations are observed in both spectra among the amino protons themselves, arising from combined chemical exchange and Overhauser effects among these moieties. Similar correlations are detected, much more weakly, in the conventional experiments depicted in Supplementary Fig. 7c for ubiquitin and in Supplementary Fig. 8b for the RNA. Moreover, HMT experiments reveal many additional long-range NOE-driven cross-peaks with the aliphatic protons for the protein case, and with the imino protons of nucleotides that are both in the same and in neighboring base pairs throughout the nucleic acid chain. When compared ith the amide protons, it is clear that the faster chemical exchange of these amine sites endows their HMT data with sizable gains when considering SNR/unit_time.
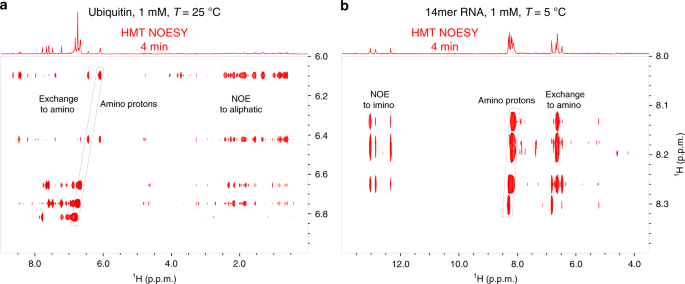
a Ubiquitin sample. b 14mer RNA in Fig. 3 . The spectrum in a was acquired with 12 loops and 80 ms per loop, whereas in b a 600 ms long saturation pulse was used instead. The strongest correlations correspond to exchange and NOE cross-peaks to aliphatic and amino protons respectively; notice in the protein and the RNA, however, interesting amide→imino cross-peaks emerge as well. Spectra were acquired at 1 GHz using a Bruker Avance Neo console equipped with a TCI cryoprobe.
HMT: exploiting hydroxyl correlations in proteins and nucleic acids
In addition to nitrogen-bound labile protons, hydroxyl 1 Hs in sidechains and in sugars are notoriously challenging targets to work with in protein and nucleic acid NMR, respectively 46 , 47 , 48 . In both cases, these -OH peaks are often buried under other, sharper, and more intense amine and amide resonances. On the other hand, hydroxyl 1 Hs usually undergo faster chemical exchange with water than the latter; as illustrated in Figs. 1 and 2 , this qualifies them for potentially large cross-peak enhancements when targeted by the HMT scheme. Figure 6a shows a version of the HMT pulse sequence that could allow such usually “hidden” hydroxyl 1 Hs be encoded, by incorporating additional 15 N/ 13 C-based filters 49 , 50 aimed at suppressing the intense signals from 1 Hs bound to 15 N and 13 C that would otherwise complicate the OH’s observation. To investigate if the OH 1 Hs could be targeted in such experiments, a series of 1D variable-temperature (5–25 °C) 15 N/ 13 C-suppressed spectra were acquired; 1 H resonances that survived the N-H and C-H suppression and became sharper at lower temperatures owing to slower chemical exchange with water, and were confirmed as likely candidates to arise from the labile hydroxyl 1 Hs (Supplementary Fig. 7b ). Figure 6b, c exemplify the cross-correlations arising from this experiment on a doubly 13 C/ 15 N-labeled ubiquitin and on a 14mer RNA sample, respectively. Highlighted in these spectra are the hydroxyl 1 Hs addressed in the HMT NOESY; particularly interesting are OH-OH inter-residue correlations detected in ubiquitin, and the long-range correlations between the OHs of sugar and the base 1 Hs resonating between 7 and 8 ppm in the 14mer RNA that are mostly missing in the conventional spectrum (Supplementary Fig. 8c ). The latter include interesting correlations between the 2′-OH groups and aromatic nucleotide sites in the 5′-direction 46 , 51 . Whereas in the conventional NOESY only a single such interaction is observed (between the 2′-OH group of C13 and the aromatic proton of C14), in the HMT NOESY a total of 12 such correlations were detected. These give hitherto unavailable inter-residue correlations and distance information –including interactions between the 2′-OH of C10 and the H6 of U11, 2′OH of C3 and H8 of A4, and 2′-OH of G5 and H6 of C6. Additional cross-peaks are observed, but their unambiguous assignment is still challenging in this 2D NOESY spectrum due to resonance overlap persisting even at 1 GHz. The incorporation of the HMT NOESY segment into a 3D experiment incorporating further information regarding the ribose 1 Hs, could potentially lift such ambiguities and contribute substantially to 3D structure determinations.
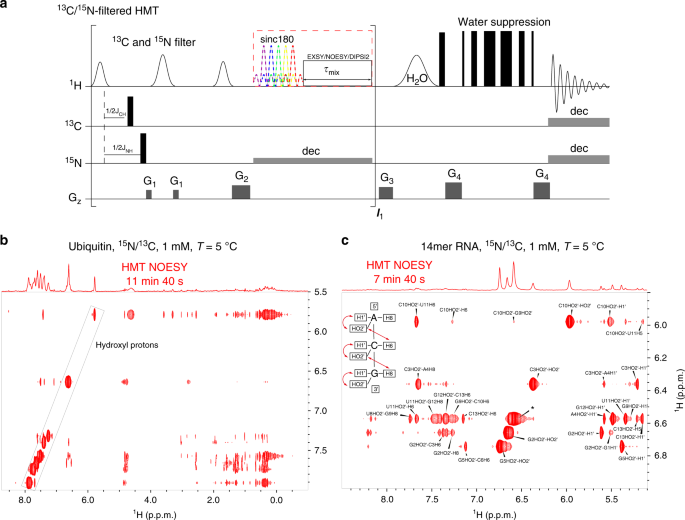
a Pulse sequence incorporating a filter is applied at the beginning of every loop in order to prevent the suppressed 13 C- and/or 15 N-bound 1 Hs to start recovering during mixing periods. Consequently, short mixing times have to be used per loop; this is not a hindrance in this case thanks to the OH’s fast chemical exchange with water. In the illustrated filter, a selective 1 Hs spin-echo is applied only to the HO-bearing regions, so as not to disturb other 1 Hs that maybe receiving the magnetization from these targeted sites. Examples are illustrated with b ubiquitin acquired using 10 loops and 50 ms per loop, and c the 14mer RNA using 15 loops and 40 ms mixing time per loop. The structure in the inset illustrates the latter’s expected NOE correlations involving hydroxyl 1 Hs, and a nearly complete assignment of these resonances revealing the many long-range correlations provided by the HMT spectrum. Resonances labeled with an asterisk does not have unambiguous assignment, as they involve 4 or 5 overlapping hydroxyl 1 Hs. Spectra were acquired at 1 GHz using a Bruker Avance Neo console equipped with a TCI cryoprobe.
Hadamard MT was introduced here as an extension of projective-measurement experiments where, instead of a looped t 1 time-domain encoding 15 , selective irradiations are used to impart significant SNR/unit_time gains in homonuclear correlation experiments involving fast-exchanging protons. To achieve these gains, HMT exploits the flow of water polarization to reset the states of the targeted sites, which are then perturbed away from equilibrium via selective saturation or inversions. Polarization transfer processes spreading throughout the molecules via dipole–dipole relaxation or J -couplings are thus magnified, in a manner that resembles CEST-derived enhancements 52 , 53 . Thus, although chemical exchange with the solvent deteriorates conventional homonuclear transfer experiments, the abundant, slowly relaxing water resonance improves these processes by several-fold when switching to this new encoding scheme. Relaxation properties of the non-labile 1 Hs on the receiving end of these transfers will limit the extent of these gains, as the MT process will only be effective over their “memory times” T 1 . The efficiency of these experiments improves by operating at ultrahigh fields, where the labile 1 Hs are better resolved, and the extent of the saturation-derived transfer can be increased. Possible drawbacks of relying on such high-field MT processes concern potential sample heating effects, but these were not found to be a problem even when operating at 1 GHz. Specificity in these MT experiments could be compromised by spin-diffusion among the non-labile 1 Hs in the system; we investigated a similar possibility for the case of L-PROSY, where it was found that a 1:1 correlation with conventional NOESY cross-peak intensities was preserved even in the presence of a spin-diffusion sink pool 15 . This correspondence will persist over a wide range of solvent exchange rates and correlation times; it remains to be investigated whether the continuous-irradiation or the repeated inversion versions of HMT, might perform differently in terms of potential spin-diffusion effects. It is also interesting to note that while HMT and L-PROSY operate based on related principles, the per-scan enhancements in HMT will generally be equal to or higher than in L-PROSY. This is a consequence of: (i) the additional degree of freedom that tailoring the intensity of the saturating/inverting RF field provides to the MT experiments, and (ii) the higher per-scan efficiency enjoyed by frequency-based Hadamard methods over time-domain encoded counterparts, where signals decay owing to T 2 and exchange-driven losses over the course of the t 1 evolutions. On top of this there is Hadamard’s “compressed sensing” time savings, whereby in the absence of signal averaging needs, the overall duration of the experiment should not require more scans than the actual number of peaks; given sufficient SNR, suitably regularized non-uniform-sampling Fourier reconstruction schemes can also enjoy from such benefits.
Although this study focused on homonuclear transfers originating from labile 1 Hs being replenished by the solvent, the HMT concept could be exploited in additional NMR settings where fast-relaxing sites can be individually addressed; in such instances, cross-peaks to slower-relaxing sites could be enhanced without a need for exchanges with the solvent. These cases may include methyl groups in otherwise deuterated proteins, small molecules interacting dynamically with bigger ones (e.g., drug–protein binding processes) 54 , 55 , 56 , as well as fast-relaxing sites in paramagnetic biomolecules. It is also worth exploring extension of the method to incorporate other mixing schemes, particularly those that could serve to distinguish spin-diffusion and relaxation-driven transfers from chemical exchange effects 57 , 58 , 59 . The high efficiency and short-acquisition of HMT experiments can be further utilized for fast multidimensional reaction monitoring, especially involving small reagents/products in the fast tumbling regime that are traditionally hard to correlate with NOESY. Last but not least, the 2D Hadamard concepts introduced here could also be included as part of correlations with heteronuclei in 3D spectral acquisitions 27 , 60 . Heteronuclear analogs of the homonuclear polarization transfer processes discussed here can also be envisaged. Examples of these developments will be discussed in upcoming studies.
Sample preparation
Myo-inositol was purchased from Sigma Aldrich (Israel) and prepared as 5 mM solution at pH 6.0. 5 mM sucrose was prepared using household sugar at pH 6.5. Natural abundance α2–8 (SiA) 4 was purchased from EY Laboratories Inc (San Mateo, CA); 25 mg of this tetramer were dissolved in 400 μL of 20 mM phosphate buffer at pH 6.5 containing 0.05% NaN 3 , yielding a ~50 mM final solution at pH 7.35. The 13 C/ 15 N-labeled 14mer gCUUGc tetraloop (5′-pppGGCAGCUUGCUGCC-3′) was prepared from a linearized plasmid DNA by a run-off in vitro transcription using the T7 RNA polymerase 61 . In addition, the plasmid DNA contained a self-cleaving HDV ribozyme to ensure 3′ homogeneity. Labeled rNTPs were purchased from Silantes (Munich, Germany). The RNA was folded in NMR buffer (10 mM phosphate buffer + 1 mM EDTA pH: 6.4) in 90% H 2 O and 10% D 2 O by denaturing it for 5 min at 95 °C and subsequently slowly cooling down to room temperature. The final concentration of the RNA was 1 mM. Ubiquitin was purchased from Asla Biotech and was dissolved in PBS (Dulbecco’s Phosphate Buffer Saline at physiological pH, purchased from Biological Industries) at a concentration of 1 mM; LA5, the ligand binding domain 5 of the low-density lipoprotein receptor LDLR, was prepared as described by Szekely et al. 45 at pH 7.4 and concentration 3 mM in 10 mM Tris buffer with 1 mM CaCl 2 . All protein samples were prepared in H 2 O/D 2 O (90%:10%) solutions containing NaN 3 . Although the use of 13 C/ 15 N spin labels in these protein and RNA samples was not needed within the context of this study, they were used as a result of their immediate availability. It is worth noting that, because of this labeling, efficient composite pulse or adiabatic decoupling could be implemented throughout the acquisition of all 2D NOESY/TOCSY data (rather than just during t 2 and at t 1 /2).
NMR experiments
NMR experiments were conducted using either a 14.1 T Bruker magnet equipped with an Avance III console and TCI Prodigy probe; or a 1 GHz, 23.5 T Bruker Avance Neo equipped with a TCI cryoprobe. Hadamard experiments were carried out using 8x8 through 64 × 64 Hadamard encoding matrices, depending on the number of peaks in the spectrum. In total, 10–40 Hz nutation fields were used for saturation, whereas 20–25 Hz bandwidth sinc1 pulses were used in looped inversion method. The number of loops and/or duration of the saturation, were determined according to T 1 values of the receiving protons. Optimal values for NOESY and TOCSY mixing times were used in conventional experiments. Conventional TOCSY were acquired using dipsi2gpph19/dipsi2esgpph Bruker sequences employing DIPSI2 isotropic mixing 13 , whereas for NOESY experiments noesyfpgpph19/noesyesgpph was used employing Watergate 3919 62 with a flip-back pulse 63 , or excitation sculpting for water suppression. For most of the compounds, Watergate 3919 with water flip-back pulses showed excellent performance in keeping the water unperturbed 31 , 64 , 65 , 66 , and was preferred over jump-return techniques owing to the larger bandwidth of their binomial excitation. In all comparisons between HMT and conventional NOESY or TOCSY acquisitions, the experiments differed only in their encoding and mixing principles; water-suppression techniques, recovery delays d 1 and receiver gains were otherwise set identical, in order to ensure faithful comparisons. Mixing times and Watergate delays for the binomial water suppression were optimized according to the magnetic field. All spectra were processed in Bruker ® TopSpin ® 4.0.6 and 4.0.9. All spectra were apodized with QSINE or SINE window functions and while conventional spectra were zero-filled once, all Hadamard spectra were zero-filled to 256 × 1024 points. Spectra were analyzed using NMRFAM-SPARKY 67 .
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The data sets generated and analysed during the current study are available from the corresponding author on reasonable request.
Code availability
HMT sequences were deposited in an RNA-specialized Bruker NMR User Library, and its details/uses further explained in the Bruker-sponsored website https://www.bruker.com/service/information-communication/nmr-pulse-program-lib/bruker-user-library/liquids/avance-neo.html . Parameters there are given for the imino-based experiments; adjustments to target other kinds of 1 Hs may require adjusting offsets and powers.
Jeener, J., Meier, B. H., Bachmann, P. & Ernst, R. R. Investigation of exchange processes by two-dimensional NMR spectroscopy. J. Chem. Phys. 71 , 4546–4553 (1979).
Article ADS CAS Google Scholar
Aue, W. P., Bartholdi, E. & Ernst, R. R. Two-dimensional spectroscopy. Application to nuclear magnetic resonance. J. Chem. Phys. 64 , 2229–2246 (1976).
Macura, S., Huang, Y., Suter, D. & Ernst, R. R. Two-dimensional chemical exchange and cross-relaxation spectroscopy of coupled nuclear spins. J. Magn. Reson. 43 , 259–281 (1981).
ADS CAS Google Scholar
Macura, S. & Ernst, R. R. Elucidation of cross relaxation in liquids by two-dimensional N.M.R. spectroscopy. Mol. Phys. 41 , 95–117 (1980).
Overhauser, A. W. Polarization of nuclei in metals. Phys. Rev. 92 , 411–415 (1953).
Article ADS CAS MATH Google Scholar
Noggle, J. H. & Schirmer, R. E. The Nuclear Overhauser Effect . (Academic Press Inc., 1971).
Neuhaus, D. & Williamson, M. P. The Nuclear Overhauser Effect in Structural and Conformational Analysis. VCH 75 (1989).
Cavanagh, J., Fairbrother, W. J., Palmer, A. G., Rance, M. & J, S. N. Protein NMR Spectroscopy https://doi.org/10.1016/B978-012164491-8/50005-1 (Elsevier, 2007).
Wuthrich, K. NMR of proteins and nucleic acids. (Wiley, 1986).
Kumar, A., Wagner, G., Ernst, R. R. & Wuethrich, K. Buildup rates of the nuclear Overhauser effect measured by two-dimensional proton magnetic resonance spectroscopy: implications for studies of protein conformation. J. Am. Chem. Soc. 103 , 3654–3658 (1981).
Article CAS Google Scholar
Huang, Y., Macura, S. & Ernst, R. R. Carbon-13 exchange maps for the elucidation of chemical exchange networks. J. Am. Chem. Soc. 103 , 5327–5333 (1981).
Macura, S., Westler, W. M. & Markley, J. L. Two-dimensional exchange spectroscopy of proteins. Methods Enzymol. 239 , 106–144 (1994).
Article CAS PubMed Google Scholar
Rucker, S. & Shaka, A. J. Broadband homonuclear cross polarization in 2D N.M.R. using DIPSI-2. Mol. Phys. 68 , 509–517 (1989).
Furrer, J., Kramer, F., Marino, J. P., Glaser, S. J. & Luy, B. Homonuclear Hartmann-Hahn transfer with reduced relaxation losses by use of the MOCCA-XY16 multiple pulse sequence. J. Magn. Reson. 166 , 39–46 (2004).
Article ADS CAS PubMed Google Scholar
Novakovic, M., Cousin, S. F., Jaroszewicz, M. J., Rosenzweig, R. & Frydman, L. Looped-PROjected SpectroscopY (L-PROSY): a simple approach to enhance backbone/sidechain cross-peaks in 1H NMR. J. Magn. Reson. 294 , 169–180 (2018).
Facchi, P., Lidar, D. A. & Pascazio, S. Unification of dynamical decoupling and the quantum Zeno effect. Phys. Rev. A 69 , 032314 (2004).
Facchi, P. & Pascazio, S. Quantum Zeno dynamics: mathematical and physical aspects. J. Phys. A Math. Theor . 41 (2008).
Zheng, W. et al. Experimental demonstration of the quantum Zeno effect in NMR with entanglement-based measurements. Phys. Rev. A —. Mol. Opt. Phys. 87 , 1–6 (2013).
Google Scholar
Álvarez, G. A., Rao, D. D. B., Frydman, L. & Kurizki, G. Zeno and anti-zeno polarization control of spin ensembles by induced dephasing. Phys. Rev. Lett. 105 , 1–4 (2010).
Bretschneider, C. O., Alvarez, G. A., Kurizki, G. & Frydman, L. Controlling spin-spin network dynamics by repeated projective measurements. Phys. Rev. Lett. 108 , 1–5 (2012).
Schanda, P., Kupče, Ē. & Brutscher, B. SOFAST-HMQC experiments for recording two-dimensional deteronuclear correlation spectra of proteins within a few seconds. J. Biomol. NMR 33 , 199–211 (2005).
Schulze-Sünninghausen, D., Becker, J. & Luy, B. Rapid heteronuclear single quantum correlation NMR spectra at natural abundance. J. Am. Chem. Soc. 136 , 1242–1245 (2014).
Article PubMed CAS Google Scholar
Guivel-Scharen, V., Sinnwell, T., Wolff, S. D. & Balaban, R. S. Detection of proton chemical exchange between metabolites and water in biological tissues. J. Magn. Reson. 133 , 36–45 (1998).
Zhou, J. & van Zijl, P. C. M. Chemical exchange saturation transfer imaging and spectroscopy. Prog. Nucl. Magn. Reson. Spectrosc. 48 , 109–136 (2006).
Martinho, R. P., Novakovic, M., Olsen, G. L. & Frydman, L. Heteronuclear 1D and 2D NMR resonances detected by chemical exchange saturation transfer to water. Angew. Chem. Int. Ed. 56 , 3521–3525 (2017).
Novakovic, M., Martinho, R. P., Olsen, G. L., Lustig, M. S. & Frydman, L. Sensitivity-enhanced detection of non-labile proton and carbon NMR spectra on water resonances. Phys. Chem. Chem. Phys. 20 , 56–62 (2018).
Kupče, E., Nishida, T. & Freeman, R. Hadamard NMR spectroscopy. Prog. Nucl. Magn. Reson. Spectrosc. 42 , 95–122 (2003).
Kupče, Ē. & Freeman, R. Two-dimensional Hadamard spectroscopy. J. Magn. Reson. 162 , 300–310 (2003).
Article ADS PubMed CAS Google Scholar
Kupče, E. & Freeman, R. Frequency-domain Hadamard spectroscopy. J. Magn. Reson. 162 , 158–165 (2003).
Grzesiek, S. & Bax, A. The importance of not saturating H2O in protein NMR. Application to sensitivity enhancement and NOE measurements. J. Am. Chem. Soc. 115 , 12593–12594 (1993).
Guéron, M., Plateau, P. & Decorps, M. Solvent signal suppression in NMR. Prog. Nucl. Magn. Reson. Spectrosc. 23 , 135–209 (1991).
Article Google Scholar
Yuwen, T. & Skrynnikov, N. R. CP-HISQC: a better version of HSQC experiment for intrinsically disordered proteins under physiological conditions. J. Biomol. NMR 58 , 175–192 (2014).
Zaiss, M. & Bachert, P. Chemical exchange saturation transfer (CEST) and MR Z-spectroscopy in vivo: a review of theoretical approaches and methods. Phys. Med. Biol. 58 , 221–269 (2013).
van Zijl, P. C. M., Lam, W. W., Xu, J., Knutsson, L. & Stanisz, G. J. Magnetization transfer contrast and chemical exchange saturation transfer MRI. Features and analysis of the field-dependent saturation spectrum. Neuroimage 168 , 222–241 (2018).
Article PubMed Google Scholar
Zaiß, M., Schmitt, B. & Bachert, P. Quantitative separation of CEST effect from magnetization transfer and spillover effects by Lorentzian-line-fit analysis of z-spectra. J. Magn. Reson. 211 , 149–155 (2011).
McConnell, H. M. Reaction rates by nuclear magnetic resonance. J. Chem. Phys. 28 , 430–431 (1958).
Helgstrand, M., Hard, T. & Allard, P. Simulations of NMR pulse sequences during equilibrium and non-equilibrium chemical exchange. J. Biomol. NMR 18 , 49–63 (2000).
Roussel, T., Rosenberg, J. T., Grant, S. C. & Frydman, L. Brain investigations of rodent disease models by chemical exchange saturation transfer at 21.1 T. NMR Biomed. 31 , e3995 (2018).
Article PubMed PubMed Central CAS Google Scholar
Chung, J. J., Choi, W., Jin, T., Lee, J. H. & Kim, S.-G. Chemical-exchange-sensitive MRI of amide, amine and NOE at 9.4 T versus 15.2 T. NMR Biomed. 30 , e3740 (2017).
Korb, J. P. & Bryant, R. G. Magnetic field dependence of proton spin-lattice relaxation times. Magn. Reson. Med. 48 , 21–26 (2002).
Levitt, M. Spin Dynamics: Basics of Nuclear Magnetic Resonance https://doi.org/10.1002/cmr.a.20130 (John Wiley & Sons Ltd, 2000).
Kowalewski, J. & Maler, L. Nuclear Spin Relaxation in Liquids. Nuclear Magnetic Resonance https://doi.org/10.1201/9781420012194 (CRC Press, 2006).
Shinar, H. et al. Sialo-CEST: chemical exchange saturation transfer NMR of oligo- and poly-sialic acids and the assignment of their hydroxyl groups using selective- and HSQC-TOCSY. Carbohydr. Res. 389 , 165–173 (2014).
Battistel, M. D., Shangold, M., Trinh, L., Shiloach, J. & Freedberg, I. Evidence for helical structure in a tetramer of α 2–8 sialic acid: unveiling a structural antigen. J. Am. Chem. Soc. 134 , 8–11 (2012).
Szekely, O. et al. Identification and rationalization of kinetic folding intermediates for a low-density lipoprotein receptor ligand-binding module. Biochemistry 57 , 4776–4787 (2018).
Hennig, M., Fohrer, J. & Carlomagno, T. Assignment and NOE analysis of 2′-hydroxyl protons in RNA: implications for stabilization of RNA A-form duplexes. J. Am. Chem. Soc . 127 , 2028–2029 (2005).
Grzesiek, S. & Bax, A. Measurement of amide proton exchange rates and NOEs with water in 13C/15N-enriched calcineurin B. J. Biomol. NMR 3 , 627–638 (1993).
CAS PubMed Google Scholar
Wang, Y. X. et al. Mapping hydration water molecules in the HIV-1 protease/DMP323 complex in solution by NMR spectroscopy. Biochemistry 35 , 12694–12704 (1996).
Ogura, K., Terasawa, H. & Inagaki, F. An improved double-tuned and isotope-filtered pulse scheme based on a pulsed field gradient and a wide-band inversion shaped pulse. J. Biomol. NMR 8 , 492–498 (1996).
Brockerman, J. A., Okon, M. & McIntosh, L. P. Detection and characterization of serine and threonine hydroxyl protons in Bacillus circulans xylanase by NMR spectroscopy. J. Biomol. NMR 58 , 17–25 (2014).
Nozinovic, S., Fürtig, B., Jonker, H. R. A., Richter, C. & Schwalbe, H. High-resolution NMR structure of an RNA model system: the 14-mer cUUCGg tetraloop hairpin RNA. Nucleic Acids Res. 38 , 683–694 (2009).
Ward, K. M., Aletras, A. H. & Balaban, R. S. A new class of contrast agents for MRI based on proton chemical exchange dependent saturation transfer (CEST). J. Magn. Reson. 143 , 79–87 (2000).
Van Zijl, P. C. M. & Yadav, N. N. Chemical exchange saturation transfer (CEST): what is in a name and what isn’t? Magn. Reson. Med. 65 , 927–948 (2011).
Dalvit, C. et al. Identification of compounds with binding affinity to proteins via magnetization transfer from bulk water. J. Biomol. NMR 18 , 65–68 (2000).
Dalvit, C., Fogliatto, G., Stewart, A., Veronesi, M. & Stockman, B. WaterLOGSY as a method for primary NMR screening: practical aspects and range of applicability. J. Biomol. NMR 21 , 349–359 (2001).
Mayer, M. & Meyer, B. Characterization of ligand binding by saturation transfer difference NMR spectroscopy. Angew. Chem. - Int. Ed. 38 , 1784–1788 (1999).
Fejzo, J., Westler, W. M., Macura, S. & Markley, J. L. Elimination of cross-relaxation effects from two-dimensional chemical-exchange spectra of macromolecules. J. Am. Chem. Soc. 112 , 2574–2577 (1990).
Fejzo, J., Westler, W. M., Markley, J. L. & Macura, S. Complete elimination of spin diffusion from selected resonances in two-dimensional cross-relaxation spectra of macromolecules by a novel pulse sequence (SNOESY). J. Am. Chem. Soc. 114 , 1523–1524 (1992).
Hwang, T., Mori, S., Shaka, A. J. & van Zijl, P. C. M. Application of phase-modulated CLEAN chemical EXchange spectroscopy (CLEANEX-PM) to detect water−protein proton exchange and intermolecular NOEs. J. Am. Chem. Soc. 119 , 6203–6204 (1997).
Brutscher, B. Combined frequency- and time-domain NMR spectroscopy. Application to fast protein resonance assignment. J. Biomol. NMR 29 , 57–64 (2004).
Guilleres, J., Lopez, P. J., Proux, F., Launay, H. & Dreyfus, M. A mutation in T7 RNA polymerase that facilitates promoter clearance. Proc. Natl Acad. Sci. USA 102 , 5958–5963 (2005).
Piotto, M., Saudek, V. & Sklenář, V. Gradient-tailored excitation for single-quantum NMR spectroscopy of aqueous solutions. J. Biomol. NMR 2 , 661–665 (1992).
Lippens, G., Dhalluin, C. & Wieruszeski, J. M. Use of a water flip-back pulse in the homonuclear NOESY experiment. J. Biomol. NMR 5 , 327–331 (1995).
Redfield, A. G., Kunz, S. D. & Ralph, E. K. Dynamic range in Fourier transform proton magnetic resonance. J. Magn. Reson. 19 , 114–117 (1975).
Plateau, P. & Guéron, M. Exchangeable proton nmr without base-line distortion, using new strong-pulse sequences. J. Am. Chem. Soc. 104 , 7310–7311 (1982).
Louis-Joseph, A., Abergel, D., Lebars, I. & Lallemand, J. Y. Enhancement of water suppression by radiation damping-based manipulation of residual water in Jump and Return NMR experiments. Chem. Phys. Lett. 337 , 92–96 (2001).
Lee, W., Tonelli, M. & Markley, J. L. NMRFAM-SPARKY: enhanced software for biomolecular NMR spectroscopy. Bioinformatics 31 , 1325–1327 (2015).
Zhou, Z. et al. A new decoupling method for accurate quantification of polyethylene copolymer composition and triad sequence distribution with 13C NMR. J. Magn. Reson. 187 , 225–233 (2007).
Fu, R. & Bodenhausen, G. Evaluation of adiabatic frequency-modulated schemes for broadband decoupling in isotropic liquids. J. Magn. Reson. - Ser. A 119 , 129–133 (1996).
Hwang, T. & Shaka, A. J. Water suppression that works. Excitation sculpting using arbitrary wave-forms and pulsed-field gradients. J. Magn. Reson. - Ser. A 112 , (1995).
Davis, A. L. & Wimperis, S. A solvent suppression technique giving NMR spectra with minimal amplitude and phase distortion. J. Magn. Reson. 84 , 620–626 (1989).
Download references
Acknowledgements
We are grateful to professor D. Fass (Weizmann Institute) for the LA5 sample and to Dr. Tali Scherf (Weizmann Institute) for the assistance in the GHz experiments. This work was supported by the Kimmel Institute for Magnetic Resonance (Weizmann Institute), the EU Horizon 2020 program (FET-OPEN Grant 828946, PATHOS), Israel Science Foundation Grant 965/18, and the Perlman Family Foundation. H.S. was supported by DFG-funded collaborative research center 902. Work at BMRZ is supported by the state of Hesse. Joint support to L.F., H.S. was given by the German-Israel Foundation (grant G-1501-302). We wish to thank Boris Fürtig, Robbin Schnieders, and Christian Richter for stimulating discussions.
Author information
Authors and affiliations.
Department of Chemical and Biological Physics, Weizmann Institute of Science, 7610001, Rehovot, Israel
Mihajlo Novakovic & Lucio Frydman
Bruker UK Ltd., Banner Lane, Coventry, UK
Ēriks Kupče
Institute for Organic Chemistry and Chemical Biology, Center for Biomolecular Magnetic Resonance, Johann Wolfgang Goethe-University, D-60438, Frankfurt/Main, Germany
Andreas Oxenfarth & Harald Schwalbe
Laboratory of Bacterial Polysaccharides, Center for Biologics Evaluation and Research, Food and Drug Administration, 10903 New Hampshire Ave, Silver Spring, MD, 20993, USA
Marcos D. Battistel & Darón I. Freedberg
You can also search for this author in PubMed Google Scholar
Contributions
M.N., E.K., and L.F. conceived the project. M.N., E.K., and L.F. implemented the method. M.N. carried out the NMR measurements and the spin simulations. M.N. and E.K. processed the data. M.N., A.O., H.S., and L.F. conceived the nucleic acids applications and evaluated their NMR data. M.N., M.D.B., D.F., and L.F. conceived the oligosaccharide applications and evaluated their NMR data. M.N. and L.F. wrote the paper. All authors contributed to the discussions leading to the final manuscript.
Corresponding author
Correspondence to Lucio Frydman .
Ethics declarations
Competing interests.
Dr. Eriks Kupce is an employee of the Bruker Corporation. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information, peer review file, rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/ .
Reprints and permissions
About this article
Cite this article.
Novakovic, M., Kupče, Ē., Oxenfarth, A. et al. Sensitivity enhancement of homonuclear multidimensional NMR correlations for labile sites in proteins, polysaccharides, and nucleic acids. Nat Commun 11 , 5317 (2020). https://doi.org/10.1038/s41467-020-19108-x
Download citation
Received : 07 May 2020
Accepted : 17 September 2020
Published : 21 October 2020
DOI : https://doi.org/10.1038/s41467-020-19108-x
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
This article is cited by
- George Peat
- Patrick J. Boaler
- Dušan Uhrín
Nature Communications (2023)
By submitting a comment you agree to abide by our Terms and Community Guidelines . If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.

Shared Instrumentation Facility
Department of Chemistry
- NMR Experiment Guide
- Attached Proton Test
- Refocused Insensitive Nuclei Enhanced by Polarization Transfer
TOCSY – Selective 1D
- NOESY – Selective 1D
- Solvent Suppression – Presaturation
- Solvent Suppression – Excitation Sculpting
- COSY-gpmfphpp
- NOESY-gpphpp
- HMBC – phase sensitive
- T1 Relaxation
- T2 Relaxation
- Pseudo 2D Sequence (zg2d)
- multi_batchdel
- Command Queue / Spooler
- Processing bulk data (multicmd, multi_integ2)
- 1D Experiments
| | SELMLGP | selmlgp | ||||
| One peak is selected and signal is transferred from it to all -coupled protons, up to three bonds away, in a stepwise process. | ||||||
| 4 | 2 | 1 | 64k | 20 | 6.18 | |
| or to read in proper pulse parameters = mixing time (default = 60 ms); depends on how far magnetization is to be transferred. Longer times will allow further magnetization transfer, but will affect intensity of shorter couplings. is the offset frequency of the shaped pulse. ) is calculated by the difference between the valued determined in steps 2 and 1 (ν2 – ν1) . Make note of the sign | ||||||
| Enlarge | ||||||
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- My Bibliography
- Collections
- Citation manager
Save citation to file
Email citation, add to collections.
- Create a new collection
- Add to an existing collection
Add to My Bibliography
Your saved search, create a file for external citation management software, your rss feed.
- Search in PubMed
- Search in NLM Catalog
- Add to Search
Saturation transfer difference 1D-TOCSY experiments to map the topography of oligosaccharides recognized by a monoclonal antibody directed against the cell-wall polysaccharide of group A streptococcus
Affiliation.
- 1 Department of Chemistry, Simon Fraser University, Burnaby, British Columbia, Canada V5A 1S6. [email protected]
- PMID: 12487612
- DOI: 10.1021/ja020983v
A new saturation transfer difference 1D-TOCSY NMR experiment that allows the investigation of complex ligands interacting with proteins and its application in the mapping of which portions of oligosaccharide ligands (epitope) interact with a complementary antibody are described. The interaction between trisaccharide and hexasaccharide ligands, corresponding to fragments of the cell-wall polysaccharide of Streptococcus Group A, and a monoclonal antibody directed against the polysaccharide is investigated at the molecular level. The polysaccharide consists of alternating alpha-(1-->2) and alpha-(1-->3) linked L-rhamnopyranose (Rha) residues with branching N-acetyl-D-glucopyranosylamine (GlcNAc) residues linked beta-(1-->3) to alternate rhamnopyranose rings. The epitope is proven to consist not only of the immunodominant GlcNAc sugar but also of an entire branched trisaccharide repeating unit. The experimental NMR data serve to check and validate the computed models of the oligosaccharide-antibody complexes.
PubMed Disclaimer
Similar articles
- NMR studies of the antibody-bound conformation of a carbohydrate-mimetic peptide. Johnson MA, Rotondo A, Pinto BM. Johnson MA, et al. Biochemistry. 2002 Feb 19;41(7):2149-57. doi: 10.1021/bi011927u. Biochemistry. 2002. PMID: 11841205
- Bivalency and epitope specificity of a high-affinity IgG3 monoclonal antibody to the Streptococcus group A carbohydrate antigen. Molecular modeling of a Fv fragment. Pitner JB, Beyer WF, Venetta TM, Nycz C, Mitchell MJ, Harris SL, Mariño-Albernas JR, Auzanneau FI, Forooghian F, Pinto BM. Pitner JB, et al. Carbohydr Res. 2000 Jan 29;324(1):17-29. doi: 10.1016/s0008-6215(99)00279-7. Carbohydr Res. 2000. PMID: 10723608
- Conformational analysis of oligosaccharides corresponding to the cell-wall polysaccharide of the Streptococcus group A by Metropolis Monte Carlo simulations. Stuike-Prill R, Pinto BM. Stuike-Prill R, et al. Carbohydr Res. 1995 Dec 27;279:59-73. doi: 10.1016/0008-6215(95)00279-0. Carbohydr Res. 1995. PMID: 8593633
- Application of two-dimensional NMR spectroscopy and molecular dynamics simulations to the conformational analysis of oligosaccharides corresponding to the cell-wall polysaccharide of Streptococcus group A. Kreis UC, Varma V, Pinto BM. Kreis UC, et al. Int J Biol Macromol. 1995 Jun;17(3-4):117-30. doi: 10.1016/0141-8130(95)92678-j. Int J Biol Macromol. 1995. PMID: 7577810
- A review of NMR analysis in polysaccharide structure and conformation: Progress, challenge and perspective. Yao HY, Wang JQ, Yin JY, Nie SP, Xie MY. Yao HY, et al. Food Res Int. 2021 May;143:110290. doi: 10.1016/j.foodres.2021.110290. Epub 2021 Mar 9. Food Res Int. 2021. PMID: 33992390 Review.
- Progress towards a glycoconjugate vaccine against Group A Streptococcus. Burns K, Dorfmueller HC, Wren BW, Mawas F, Shaw HA. Burns K, et al. NPJ Vaccines. 2023 Mar 28;8(1):48. doi: 10.1038/s41541-023-00639-5. NPJ Vaccines. 2023. PMID: 36977677 Free PMC article. Review.
- Synthetic Glycans to Improve Current Glycoconjugate Vaccines and Fight Antimicrobial Resistance. Del Bino L, Østerlid KE, Wu DY, Nonne F, Romano MR, Codée J, Adamo R. Del Bino L, et al. Chem Rev. 2022 Oct 26;122(20):15672-15716. doi: 10.1021/acs.chemrev.2c00021. Epub 2022 May 24. Chem Rev. 2022. PMID: 35608633 Free PMC article. Review.
- Recent Advances in the Synthesis of Glycoconjugates for Vaccine Development. Colombo C, Pitirollo O, Lay L. Colombo C, et al. Molecules. 2018 Jul 13;23(7):1712. doi: 10.3390/molecules23071712. Molecules. 2018. PMID: 30011851 Free PMC article. Review.
- Total synthesis of a Streptococcus pneumoniae serotype 12F CPS repeating unit hexasaccharide. Seeberger PH, Pereira CL, Govindan S. Seeberger PH, et al. Beilstein J Org Chem. 2017 Jan 25;13:164-173. doi: 10.3762/bjoc.13.19. eCollection 2017. Beilstein J Org Chem. 2017. PMID: 28228857 Free PMC article.
- Automated glycan assembly of a S. pneumoniae serotype 3 CPS antigen. Weishaupt MW, Matthies S, Hurevich M, Pereira CL, Hahm HS, Seeberger PH. Weishaupt MW, et al. Beilstein J Org Chem. 2016 Jul 12;12:1440-6. doi: 10.3762/bjoc.12.139. eCollection 2016. Beilstein J Org Chem. 2016. PMID: 27559395 Free PMC article.
Publication types
- Search in MeSH
Related information
- PubChem Compound
- PubChem Substance
LinkOut - more resources
Full text sources.
- American Chemical Society
Other Literature Sources
- The Lens - Patent Citations
Molecular Biology Databases
- Immune Epitope Database and Analysis Resource

- Citation Manager
NCBI Literature Resources
MeSH PMC Bookshelf Disclaimer
The PubMed wordmark and PubMed logo are registered trademarks of the U.S. Department of Health and Human Services (HHS). Unauthorized use of these marks is strictly prohibited.

IMAGES
VIDEO
COMMENTS
The basic idea behind this so-called 1D TOCSY experiment is quite straightforward: first, a combination of shaped rf pulses and magnetic field gradients is used to selectively excite one signal in the spectrum; then, a special pulse sequence element called a "spinlock" is used to transfer magnetization from the "excited" spins to the ...
The 1D TOCSY experiment was proposed by Bax and co-workers [4, 5], and by Kessler et al. [6]. The essential features of the pulse sequence involve the use of selective excitation of a desired resonance, followed by a homonu- clear Hartman-Hahn (or isotropic) mixing period [2, 7]. That is, the unit Pnonsel-t l in the 2D TOCSY pulse sequence is ...
1D TOCSY (Total correlation spectroscopy), like 2D TOCSY gives correlations between all protons within a given spin system. In 1D TOCSY, one peak is selected and signal is transferred from it to to all J-coupled protons in a stepwise process. Instead of cross peaks, magnetization transfer is seen as increased multiplet intensity. ...
NMR Methodological Overview. Zoltán Szakács, Zsuzsanna Sánta, in Anthropic Awareness, 2015. 7.3.2 2D TOCSY. The 2D variant of the 1D TOCSY experiment discussed above reveals in one single measurement the coupling network that any proton is involved in. This feature is useful if the 1D proton spectrum happens to be too crowded to start a 1D TOCSY spectrum by the selective excitation of the ...
The selective 1D TOCSY (HOHAHA) experiment has become one of the most important NMR techniques for establishing 1 H-1 H connectivity via scalar coupling in small and medium-size molecules. Magnetization transfer between protons within the same coupling network affords 1D proton subspectra of individual spin systems.
During spin-locking, the TOCSY experiment manipulates the isotropic mixing condition to generate crosspeaks between all hydrogen nuclei forming part of a continuous chain of coupled spins. A chain of four hydrogens—labeled A, B, C, and D—can be considered. ... The structure (top) and 1D 1 H spectrum (bottom) of trans-2-hexenoic acid ...
The selective 1D TOCSY experiment can be relied upon to produce quantifiable read peaks for target metabolites present at concentrations of ~50 µM or better within a 1 min acquisition time using a room temperature 5 mm inverse probe at 500 MHz. If a 5 mm inverse cryoprobe on an 800 MHz instrument were employed the threshold sensitivity of the ...
The selective 1D TOCSY experiment can be relied upon to produce quantifiable read peaks for target metabolites present at concentrations of ~50 μM or better within a 1 min acquisition time using a room temperature 5 mm inverse probe at 500 MHz. If a 5 mm inverse cryoprobe on an 800 MHz instrument were employed the threshold sensitivity of the ...
Selective 1D TOCSY is a powerful tool in the assignment of NMR spectra of organic molecules. Here an order of magnitude improvement in selectivity, allowing overlapping multiplets to be excited separately, is achieved in a single scan using the very recent GEMSTONE method. The new experiment is illustrated u
This chapter discusses the one-dimensional (1-D) TOCSY (TOtal Correlation SpectroscopY) experiment, which, together with 1-D nuclear Overhauser enhancement spectroscopy (NOESY), is probably the most frequently and routinely used selective 1-D experiment for elucidating the spin-spin coupling network, and obtaining homonuclear coupling constants.
In conclusion, a new ultraselective, ultrahigh resolution 1D TOCSY experiment has been introduced that can be used to perform targeted analyses of spin systems where spectral overlap prohibits the use of conventional selective methods. The ultrahigh resolution nature of the experiment enables unambiguous identification of chemical environments ...
1d in experiment A (A is a number) move the parameters to experiment b (B is a number) by typing mp(A, B). If you are currently in A at the time you issue this command, you need only type mp(B). Set up the parameters for a 1D TOCSY In experiment
Steps to run 1D TOCSY on VNMRJ (assumes you have tuned, locked, and shimmed your sample) (1) ... Integrate new pulse into the 1D TOCSY experiment ... go to experiment 2 (type: jexp2), be sure that the experiment uses the identical tof value as the one in which you created the pulse.
The principle is demonstrated for the TOCSY experiment, generating a chemical shift correlation map in which a single peak is seen for each coupled relationship, but the principle is general and readily extensible to other homonuclear correlation experiments. ... Pure Shift NMR: Application of 1D PSYCHE and 1D TOCSY-PSYCHE Techniques for ...
Multidimensional TOCSY and NOESY are central experiments in chemical and biophysical NMR. ... leading to more facile conditions for implementing the 1D Hadamard encoding ... The wealth of peaks in ...
TOCSY - Selective 1D. Parameter Set. SELMLGP. Pulse Sequence. selmlgp. Experiment Description. 1D Total Correlation Spectroscopy. Correlates chemical shifts of spins that are in the same J-coupling network. One peak is selected and signal is transferred from it to all J-coupled protons, up to three bonds away, in a stepwise process.
The mixing time in 1D TOCSY experiment was adjusted (Figure S1). The spectra (Fig. 3 a-d) recorded by 1D CSSF TOCSY with appropriate mixing time at the frequencies of 7.067 ppm, 6.990 ppm, 7.058 ppm, and 7.279 ppm provided the desired results. By comparing the spectra obtained by 1D TOCSY with the standard NMR spectra of EB, OX, MX and PX, we ...
A new saturation transfer difference 1D-TOCSY NMR experiment that allows the investigation of complex ligands interacting with proteins and its application in the mapping of which portions of oligosaccharide ligands (epitope) interact with a complementary antibody are described. The interaction between trisaccharide and hexasaccharide ligands ...
SELCOGP selective cosy 1D set d1 = T1(longest of interest) 1-1.5 adjust d4 depending on the JHH desired this experiment is relatively new to our facility; its most likely utility is for observing small couplings (which cannot be done with the selective tocsy experiment), or perhaps to measure coupling constants;
A new saturation transfer difference 1D-TOCSY NMR experiment that allows the investigation of complex ligands interacting with proteins and its application in the mapping of which portions of oligosaccharide ligands (epitope) interact with a complementary antibody are described. The interaction between trisaccharide and hexasaccharide ligands, corresponding to fragments of the cell-wall ...
The TOCSY experiment shows couplings down to ca. 2 Hz., which can include 5- and occasionally 6-bond couplings. The NOESY experiment gives information about ... 1D spectrum of that axis, taken from a separate 1D experiment (called the "external" projection), or they are the actual skyline projection of the 2D data matrix in that dimension ...
• Highlight your experiment and click the Add buon and select the SELNOGPZS.2 for 1D NOESY (or SDELDIGPZS for 1D TOCSY). Make sure the F2 reference spectrum for your selecve experiment is the 1H experiment you just collected (typically experiment number 10). • Click the = buon to edit parameters for either NOESY or TOCSY:
A 1D version of TOCSY is also available, and by irradiating a single proton the rest of the spin system can be revealed. Recent advances in this technique include the 1D-CSSF (chemical shift selective filter) TOCSY experiment, which produces higher quality spectra and allows coupling constants to be reliably extracted and used to help determine ...