- PRO Courses Guides New Tech Help Pro Expert Videos About wikiHow Pro Upgrade Sign In
- EDIT Edit this Article
- EXPLORE Tech Help Pro About Us Random Article Quizzes Request a New Article Community Dashboard This Or That Game Happiness Hub Popular Categories Arts and Entertainment Artwork Books Movies Computers and Electronics Computers Phone Skills Technology Hacks Health Men's Health Mental Health Women's Health Relationships Dating Love Relationship Issues Hobbies and Crafts Crafts Drawing Games Education & Communication Communication Skills Personal Development Studying Personal Care and Style Fashion Hair Care Personal Hygiene Youth Personal Care School Stuff Dating All Categories Arts and Entertainment Finance and Business Home and Garden Relationship Quizzes Cars & Other Vehicles Food and Entertaining Personal Care and Style Sports and Fitness Computers and Electronics Health Pets and Animals Travel Education & Communication Hobbies and Crafts Philosophy and Religion Work World Family Life Holidays and Traditions Relationships Youth
- Browse Articles
- Learn Something New
- Quizzes Hot
- Happiness Hub
- This Or That Game
- Train Your Brain
- Explore More
- Support wikiHow
- About wikiHow
- Log in / Sign up
- Home and Garden
- Heating and Cooling
- Fire Making

How to Make Colored Fire
Last Updated: April 19, 2024 Fact Checked
This article was co-authored by Anthony "TC" Williams . Anthony "TC" Williams is a Professional Landscaper in Idaho. He is the President and Founder of Aqua Conservation Landscape & Irrigation, an Idaho Registered Landscape Business Entity. With over 21 years of landscaping experience, TC has worked on projects such as the Idaho Botanical Garden in Boise, Idaho. He is a Idaho Registered Contractor and a previously Licensed Irrigator in the State of Texas. There are 7 references cited in this article, which can be found at the bottom of the page. This article has been fact-checked, ensuring the accuracy of any cited facts and confirming the authority of its sources. This article has been viewed 798,908 times.
Most fireplace fires or campfires produce yellow and orange flames because firewood contains salts. By adding other chemicals, you can change the color of the flames to suit a special occasion or just to be entertained by the changing color patterns. You can create a colored fire by sprinkling chemicals in the flames, making wax cakes containing chemicals, or by soaking wood in a water and chemical solution. While making colored flames can be a lot of fun, always exercise caution when working with fire and chemical substances.
Choosing the Chemicals

- To create blue flames, use copper chloride or calcium chloride.
- To create turquoise flames, use copper sulfate.
- To create red flames, use strontium chloride
- To create pink flames, use lithium chloride.
- To create light green flames, use borax.
- To create green flames, use alum.
- To create orange flames, use sodium chloride.
- To create purple flames, use potassium chloride.
- To create yellow flames, use sodium carbonate.
- To create white flames, use magnesium sulfate.

- Copper sulfate is used as a tree root killer for plumbers, so you can find it at most hardware or home improvement stores.
- Sodium chloride is table salt, so you can purchase it at any grocery store.
- Potassium chloride is used as a water softener salt, so you can purchase it at many hardware stores.
- Borax is often used to wash clothing, so you can find it in the laundry section of most grocery stores.
- Magnesium sulfate is found in epsom salts, so you can purchase it at most drugstores and pharmacies.
- Copper chloride, calcium chloride, strontium chloride, lithium chloride, sodium carbonate, and alum must be purchased from chemical supply stores, fireplace shops, fireworks suppliers, or online retailers.
Making Wax Cakes

- You can use blocks of canning wax from the grocery store or stubs from old candles for the wax.
- Don't melt the wax over an open flame or near any open flames or you may start a fire.
- Do not heat the container with the paraffin directly with a heat source, this can cause the paraffin to spontaneously ignite or explode! Using a double boiler keeps the temperature at a safe level. [5] X Research source

- If you don't want to mix the chemicals directly into the wax, you can instead fold them into a used dryer sheet, and place the resulting bundle in the bottom of the container you plan to pour the wax into.

- You can also use small paper cups or cardboard egg cartons to form the wax cakes.

- You can add more than one wax cake with different chemicals to the fire at a time, but toss them on different parts of the fire.
- The wax cakes work well in a campfire or a fireplace.
Sprinkling Chemicals on a Fire

- For the best results, the flames should be approximately 1 foot (30 cm) high.

- Sprinkle the chemical on the edge of the fire rather than just tossing it into the middle. This will decrease the chance of a large and dangerous flare-up.
- Wear safety glasses and fire-resistant gloves when you add the chemicals to the fire.
- The smoke produced by many of these chemicals can be a serious irritant, especially for people with breathing problems. Wear a protective breathing mask while adding chemicals to the fire, and be mindful of which way the smoke is going.

Soaking Wood in Chemicals

- You can use a glass container, but avoid metal containers, which may react with the chemicals. Take care not to drop or break any glass containers in your campsite or near the firepit or fireplace.
- Be sure to wear safety glasses, rubber gloves, and a protective mask or respirator when mixing the chemical solution.
- It's best to mix the chemical solution outdoors because some of the chemicals may stain your work area or create hazardous fumes.

- Be sure to wear protective gloves when lifting the wood out of the chemical solution.
- If you don't allow the wood pieces to dry out, you'll have a hard time getting them to burn in your fire.

- If you are burning the wood in an indoor fireplace or campfire tent, make sure the chimney, flue, and dampers are in good working condition, so that you can get good ventilation.
Expert Q&A

- Some woods will produce colored flames without chemical enhancement. Driftwood from oceans makes purple and blue fire. If aged at least 4 years, applewood produces multi-colored flames. Thanks Helpful 3 Not Helpful 0
- Make sure you wear safety gear, such as safety glasses and gloves, when you color the flames. Thanks Helpful 3 Not Helpful 2

- Fire is not a toy and should never be treated as such. It goes without saying that fire is dangerous and can get out of hand quickly. Always have a fire extinguisher or an ample supply of water nearby. Thanks Helpful 2 Not Helpful 0
- If adding chemicals to a fireplace, make sure it is well-ventilated first so your house doesn't fill with chemical-laden smoke. Thanks Helpful 2 Not Helpful 0
- Keep hazardous chemicals stored in airtight containers made of plastic or glass. Don't allow children and pets near these chemicals. Thanks Helpful 2 Not Helpful 0
- Handle all chemicals carefully according to the package directions. Even seemingly harmless chemicals such as sodium chloride can cause skin irritation or burns in large amounts. Thanks Helpful 1 Not Helpful 0
Things You'll Need
- Wax or paraffin
- Cooking pan
- Paper wrappers for muffins or cupcakes
- Wood, pine cones, newspapers
- Glass or plastic container
- Safety glasses
- Rubber gloves
- Face mask or respirator
- Apron or smock to protect you while working with chemicals and wax
- Brick or stone
You Might Also Like

- ↑ http://sciencing.com/change-color-campfire-flame-2186923.html
- ↑ https://chem.libretexts.org/Bookshelves/Inorganic_Chemistry/Supplemental_Modules_and_Websites_(Inorganic_Chemistry)/Descriptive_Chemistry/Elements_Organized_by_Block/1_s-Block_Elements/Group__1%3A_The_Alkali_Metals/2Reactions_of_the_Group_1_Elements/Flame_Tests
- ↑ http://www.campfiredude.com/campfire-magic.shtml
- ↑ https://www.youtube.com/watch?v=PSg7dwmATPc
- ↑ https://www.youtube.com/watch?v=xzYcB9D8Nt4
- ↑ http://www.popsci.com/diy/article/2007-11/how-make-colored-fire
- ↑ https://www.sciencecompany.com/Creating-Flame-Colors.aspx
About This Article

If you want to make a colored fire, wait until your campfire has burnt down until there's a layer of embers under it, then sprinkle your chosen chemical into the fire to change the flames' color. Choose copper chloride if you want to create blue flames or lithium chloride for pink flames. Alternatively, melt paraffin wax before adding 2 tablespoons of your chosen chemical to it. Next, pour the mixture into paper cupcake wrappers. Once the cakes have set, remove one from the paper and throw it into a burning fire. For tips on how to soak wood in chemicals, keep reading! Did this summary help you? Yes No
- Send fan mail to authors
Reader Success Stories
Olivia Jensen
Mar 6, 2019
Did this article help you?

Oct 21, 2016
Ali Johnson
Sep 9, 2017
Willy Lauder
Nov 12, 2018
Winrock D. Gaces
Sep 8, 2017

Featured Articles

Trending Articles

Watch Articles

- Terms of Use
- Privacy Policy
- Do Not Sell or Share My Info
- Not Selling Info
Get all the best how-tos!
Sign up for wikiHow's weekly email newsletter
Your browser is not supported
Sorry but it looks as if your browser is out of date. To get the best experience using our site we recommend that you upgrade or switch browsers.
Find a solution
- Skip to main content
- Skip to navigation

- Back to parent navigation item
- Primary teacher
- Secondary/FE teacher
- Early career or student teacher
- Higher education
- Curriculum support
- Literacy in science teaching
- Periodic table
- Interactive periodic table
- Climate change and sustainability
- Resources shop
- Collections
- Remote teaching support
- Starters for ten
- Screen experiments
- Assessment for learning
- Microscale chemistry
- Faces of chemistry
- Classic chemistry experiments
- Nuffield practical collection
- Anecdotes for chemistry teachers
- On this day in chemistry
- Global experiments
- PhET interactive simulations
- Chemistry vignettes
- Context and problem based learning
- Journal of the month
- Chemistry and art
- Art analysis
- Pigments and colours
- Ancient art: today's technology
- Psychology and art theory
- Art and archaeology
- Artists as chemists
- The physics of restoration and conservation
- Ancient Egyptian art
- Ancient Greek art
- Ancient Roman art
- Classic chemistry demonstrations
- In search of solutions
- In search of more solutions
- Creative problem-solving in chemistry
- Solar spark
- Chemistry for non-specialists
- Health and safety in higher education
- Analytical chemistry introductions
- Exhibition chemistry
- Introductory maths for higher education
- Commercial skills for chemists
- Kitchen chemistry
- Journals how to guides
- Chemistry in health
- Chemistry in sport
- Chemistry in your cupboard
- Chocolate chemistry
- Adnoddau addysgu cemeg Cymraeg
- The chemistry of fireworks
- Festive chemistry
- Education in Chemistry
- Teach Chemistry
- On-demand online
- Live online
- Selected PD articles
- PD for primary teachers
- PD for secondary teachers
- What we offer
- Chartered Science Teacher (CSciTeach)
- Teacher mentoring
- UK Chemistry Olympiad
- Who can enter?
- How does it work?
- Resources and past papers
- Top of the Bench
- Schools' Analyst
- Regional support
- Education coordinators
- RSC Yusuf Hamied Inspirational Science Programme
- RSC Education News
- Supporting teacher training
- Interest groups

- More navigation items
Flame colours: a demonstration
In association with Nuffield Foundation
- Five out of five
- No comments
Show the flame colours of alkali metals, alkaline earth metals, and other metal salts by safely spraying sodium chloride, potassium chloride, lithium chloride, copper sulfate and ethanol through a bunsen flame.
Demonstration
This demonstration experiment can be used to show the flame colours given by alkali metal, alkaline earth metal, and other metal, salts. This is a spectacular version of the ‘flame tests’ experiment that can be used with chemists and non-chemists alike.
It can be extended as an introduction to atomic spectra for post-16 students.
This experiment must be done as a demonstration. It takes about ten minutes if all is prepared in advance.
Preparation includes making up the spray bottles and conducting a risk assessment. Your employer’s risk assessment must be customised by determining where to spray the flame to guarantee the audience’s safety.
- Eye protection
- Trigger pump operated spray bottles
- Bunsen burner
- Heat resistant mat(s)
- Hand-held spectroscopes or diffraction gratings (optional)
- Samples of the following metal salts (no more than 1 g of each) (see note 2):
- Sodium chloride (Low hazard)
- Potassium chloride (Low hazard) (see note 3)
- Lithium chloride (Harmful) (see note 3)
- Copper sulfate (Harmful, Danger to the environment)
- Ethanol (Highly flammable), approx 10 cm 3 for each metal salt, or IDA (industrial denatured alcohol) (Highly flammable, Harmful)
Health, safety and technical notes
- Read our standard health & safety guidance
- Carry out the whole experiment in a well-ventilated area you have previously shown to be safe. Wear eye protection. Ensure that the spray can be safely directed away from yourself and the audience.
- Sodium chloride is Low hazard. Refer to CLEAPSS Hazcard HC047b .
- Potassium chloride is Low hazard. Refer to CLEAPSS Hazcard HC47b.
- Lithium chloride is Harmful. Refer to CLEAPSS Hazcard HC047b.
- Copper sulfate is Harmful, Danger to the environment. Refer to CLEAPSS Hazcard HC026 .
- Ethanol is Highly flammable. IDA (industrial denatured alcohol) is Highly flammable, Harmful. Refer to CLEAPSS Hazcard HC040a . Do NOT be tempted to use the more toxic and volatile methanol in place of ethanol.
- Spray bottles of the type used for products such as window cleaner should be used.
- These piston-operated spray bottles should be emptied, cleaned thoroughly and finally rinsed with distilled water. Ideally, one bottle is needed for each metal salt. Never use spray bottles with a rubber bulb - the flame may flash back into the container.
- The chlorides of metals are the best but other salts also work. Make a saturated solution of each salt in about 10 cm 3 ethanol. To do this, add the salt to the ethanol in small quantities, with stirring, until no more will dissolve – often only a few mg of salt will be needed. Place each solution in a spray bottle and label the bottle. The solutions can be retained for future use. They can be stored in the plastic bottles for several weeks at least without apparent deterioration of the bottles.
- Potassium iodide and lithium iodide can be used instead. As a general rule, chlorides are usually suggested as they tend to be more volatile and more readily available. These two are in fact a little more volatile than the chloride, and potassium iodide is certainly likely to be available. Other metal salts (e.g. those of calcium and barium) can also be used provided an appropriate risk assessment is carried out. Barium chloride is toxic but gives a different colour (refer to CLEAPSS Hazcard HC010b ), while calcium chloride (Irritant) and strontium chloride (Irritant) are different again (refer to CLEAPSS Hazcard HC047b).
- Darken the room if possible.
- Light the Bunsen and adjust it to give a non-luminous, roaring flame (air hole open).
- Conduct a preliminary spray in a safe direction away from the Bunsen flame.
- Adjust the nozzles of the spray bottles to give a fine mist.
- Choose one spray bottle. Spray the solution into the flame in the direction you have rehearsed. Repeat with the other bottles.
- A spectacular coloured flame or jet should be seen in each case. The colour of the flame depends on the metal in the salt used.
- As an extension, students can view the flames through hand-held spectroscopes or diffraction gratings in order to see the line spectrum of the element. (Diffraction gratings work better. A better way to produce a steady source of light is to use discharge tubes from the Physics Department – with a suitable risk assessment.)
Teaching notes
The colours that should be seen are:
- sodium – yellow-orange (typical ‘street lamp’ yellow)
- potassium – purple-pink, traditionally referred to as ‘lilac’ (often contaminated with small amounts of sodium)
- lithium – crimson red
- copper – green/blue
- calcium – orange-red (probably the least spectacular)
- barium – apple green
- strontium – crimson
The electrons in the metal ions are excited to higher energy levels by the heat. When the electrons fall back to lower energy levels, they emit light of various specific wavelengths (the atomic emission spectrum). Certain bright lines in these spectra cause the characteristic flame colour.
The colour can be used to identify the metal or its compounds (eg sodium vapour in a street lamp). The colours of fireworks are, of course, due to the presence of particular metal salts.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry.
Practical Chemistry activities accompany Practical Physics and Practical Biology .
- 11-14 years
- 14-16 years
- 16-18 years
- Demonstrations
- Elements and the periodic table
- Properties of matter
Specification
- Flame tests can be used to identify some metal ions (cations). Lithium, sodium, potassium, calcium and copper compounds produce distinctive colours in flame tests:
- Lithium compounds result in a crimson flame.
- Sodium compounds result in a yellow flame.
- Potassium compounds result in a lilac flame.
- Calcium compounds result in an orange-red flame.
- Copper compounds result in a green flame.
- If a sample containing a mixture of ions is used some flame colours can be masked.
- 9.2Ca Describe flame tests to identify the following ions in solids: lithium ion, Li⁺ (red)
- 9.2Cb sodium ion, Na⁺ (yellow)
- 9.2Cc potassium ion, K⁺ (lilac)
- 9.2Cd calcium ion, Ca²⁺ (orange-red)
- 9.2Ce copper ion, Cu²⁺ (blue-green)
- 9.3Ca Describe tests to identify the following ions in solids or solutions as appropriate: Aluminium ion, Al³⁺
- 9.3Cb calcium ion, Ca²⁺
- 9.3Cc copper ion, Cu²⁺
- 9.3Cd iron(II) ion, Fe²⁺
- 9.3Ce iron(III) ion, Fe³⁺
- C5.2.2 interpret flame tests to identify metal ions, including the ions of lithium, sodium, potassium, calcium and copper
- C5.2.3 describe the technique of using flame tests to identify metal ions
- C4.2e interpret flame tests to identify metal ions
- Flame tests can identify metals present in a sample.
- When energy is transferred to atoms, electrons within the atoms may be promoted to higher energy levels.
- (t) the identification of Li⁺, Na⁺, K⁺, Ca²⁺ and Ba²⁺ ions by flame tests and Cl⁻, Br⁻ and I⁻ ions by their reactions with silver nitrate solution (including ionic equations)
- (s) the identification of Li⁺, Na⁺, K⁺, Ca²⁺ and Ba²⁺ ions by flame tests and Cl⁻, Br⁻ and I⁻ ions by their reactions with silver nitrate solution (including ionic equations)
- 1.9.13 demonstrate knowledge of the flame colours of different metal ions: lithium (crimson); sodium (yellow/orange); potassium (lilac); calcium (brick red); and copper(II) (blue–green/green–blue);
- 1.9.11 demonstrate knowledge of the flame colours of different metal ions: lithium (crimson); sodium (yellow/orange); potassium (lilac); calcium (brick red); and copper(II) (blue–green/green–blue);
- 1.10.3a use cation tests, including: flame tests to identify the metal ions Li⁺, Na⁺, K⁺, Ca²⁺, Ba²⁺ and Cu²⁺; and
- Mandatory experiment 1.1 - Flame tests (Li, Na, K, Ba, Sr and Cu only)
- Viewing of emission spectra of emission spectra of elements using a spectrascope or a spectrometer.
- Atomic absorption spectrometry (AAS).
- Sodium street lights, fireworks.
Related articles

5 ways to teach reactivity of metals at 14–16
2024-09-04T05:12:00Z By Kristy Turner
Use the importance of metal extraction to help contextualise this topic

Teaching structure and bonding in metals at 14–16
2024-08-22T05:05:00Z By Catherine Smith
Help your learners to master the fundamentals of metallic bonding

Representing elements and compounds | Review my learning worksheets | 14–16 years
By Lyn Nicholls
Identify learning gaps and misconceptions with this set of worksheets offering three levels of support
No comments yet
Only registered users can comment on this article., more resources.

Fractional distillation and hydrocarbons | Review my learning worksheets | 14–16 years

Chromatography | Review my learning worksheets | 14–16 years
2024-05-10T13:33:00Z By Lyn Nicholls

Solubility | Review my learning worksheets | 14–16 years
- Contributors
- Email alerts
Site powered by Webvision Cloud

Flame Test – Colorful Elements
Burning metals reveal themselves from unexpected, awesome colors in a simple flame test.
Print this Experiment

Blue, orange, and yellow flames are pretty common. What about green or purple flames? When you’re used to the everyday colors of flames, colorful changes like these can be pretty cool. These surprising colors are the result of the presence of specific metals in the burning material. Here are a couple of household materials that contain metals that are easily seen and identifiable in a Flame Test.
Experiment Videos
Here's What You'll Need
2 popsicle sticks, cream of tartar, small glass dish for each powder tested, flame source, small cup of water, container of water to douse the flame, adult supervision, let's try it.

Pour a little powder to test into each small dish. Dip one end of a popsicle stick into the water to moisten it.

Dip the wetted popsicle stick into the boric acid. Coat the tip of the stick completely.

It helps to dim the lights in your lab so the colors are easier to see. Light the flame and place the boric acid-covered popsicle stick into the flame. Move the flame under the stick to find the best color. Look for an unexpected color in portions of the flame. An assistant could take a picture of it. Douse the flame in the large container of water.

Dip the end of another popsicle stick in water. Coat the tip with the cream of tartar.

Follow the procedure in Step 3 for this test. You may see some surprising flashes. This color is harder to see but it’s there.

How Does It Work
This activity is called a flame test and it’s a real procedure used in labs. Its purpose is to identify specific elements in a material. When the boric acid was in the flame, you probably notice a bright green portion of the flame. You may have seen it only briefly but it was there. The green color denotes the presence of the element boron (B) which you’d expect in boric acid. The cream of tartar yielded a purple-colored flame. Purple is associated with the presence of potassium (K). That’s because cream of tartar is a potassium salt.
These element-specific colors are catalogued in an emission spectrum. The emission spectral color of an element occurs when certain electrons in an atom are excited to a higher energy level and then make a transition from that level to their normal energy state. In that downward transition, energy is released as a photon of light at a specific wavelength of color. The hiding element is revealed by color!
Colors of Other Elements
| As | Arsenic | Blue |
| B | Boron | Bright green |
| Ba | Barium | Pale/Yellow-green |
| Ca | Calcium | Orange-red |
| Cu (I) | Copper (I) | Blue |
| Cu (II) | Copper (II) non-halide | Green |
| Cu (II) | Copper (II) halide | Blue-green |
| Fe | Iron | Gold |
| In | Indium | Blue |
| K | Potassium | Light purple to red |
| Li | Lithium | Deep pink to dark red |
| Mg | Magnesium | Bright white |
| Mn (II) | Manganese (II) | Yellow-green |
| Mo | Molybdenum | Yellow-green |
| Na | Sodium | Bright yellow |
| P | Phosphorous | Pale blue-green |
| Pb | Lead | Blue |
| Rb | Rubidium | Red/Purple-red |
| Sb | Antimony | Pale green |
| Se | Selenium | Bright blue |
| Sr | Strontium | Crimson |
| Te | Tellurium | Pale green |
| Tl | Thallium | Bright green |
| Zn | Zinc | Blue-green to pale green |
Related Experiments

Fireproof Balloon
Common sense tells you that it’s impossible to boil water in a paper bag, but this classic parlor trick was a favorite of Victorian magicians. […]

Holiday Fire - Red
Everyone likes to cuddle up next to a nice warm fire, but we couldn’t possibly settle for any normal fire. Why not make a more […]

Burning Money
Do you have money burning a hole in your pocket? It’s probably not a wise idea to soak a $20 bill in a flammable liquid […]

CO2 Fire Extinguisher
Carbon dioxide (CO2) is a gas that humans interact with every day. For instance, you exhale it from your lungs. Drive a vehicle and it’s in the engine […]

Color Changing Liquids with Cabbage Juice and Dry Ice
If a science activity bubbles furiously, belches clouds of water vapor while changing colors, and does it safely all at the same time, then you’ve […]

Flame Light Relight - Science Magic
There’s a lot of chemistry behind the simple lighting or extinguishing of a flame. Would you believe us if we told you that you could […]
Browse more experiments by concept:

- Science Notes Posts
- Contact Science Notes
- Todd Helmenstine Biography
- Anne Helmenstine Biography
- Free Printable Periodic Tables (PDF and PNG)
- Periodic Table Wallpapers
- Interactive Periodic Table
- Periodic Table Posters
- Science Experiments for Kids
- How to Grow Crystals
- Chemistry Projects
- Fire and Flames Projects
- Holiday Science
- Chemistry Problems With Answers
- Physics Problems
- Unit Conversion Example Problems
- Chemistry Worksheets
- Biology Worksheets
- Periodic Table Worksheets
- Physical Science Worksheets
- Science Lab Worksheets
- My Amazon Books
Safe Method of Doing the Colored Flames Chemistry Demonstration 9

People have been coloring flames pretty much since the days of Prometheus. Even before chemistry was recognized as a science, colored fire was a useful chemistry demonstration. A person might wonder why a fire made with driftwood would burn in a different color from an ordinary wood fire. Why would adding minerals or salts to a fire change its flames?
Colored fire raises questions and piques scientific inquiry. As a modern chemistry demonstration, colored flames are used to evoke wonder in students and also to illustrate the emission spectra of metal salts ( the flame test ). Yet, the demonstration has come under fire because… well… fire is fire. It can burn you. It can spread. Like any lab hazard, fire needs to be treated with respect and handled using proper safety precautions. Numerous accidents have occurred with the colored fire demonstration. Yet, other instructors have performed the demonstration hundreds or possibly thousands of times without incident. Should the demonstration be banned because it’s potentially dangerous? Instead, why don’t we keep it in the repertoire, but perform it more safely. It’s really pretty simple. Don’t use liquid fuel. Don’t use toxic chemicals.
Here is one way to make colored flames. If you’re not a chemist or you just want to minimize risk while performing the demonstration, give it a try and see if it feels safer to you. In addition to being safer than the tradition demo, you don’t need any exotic chemicals to produce the colors.
Colored Flames Materials
You need a fuel and a collection of salts:
- hand sanitizer (around 65% alcohol)
- red – strontium salt (take from the inside of an emergency flare)
- orange – mix red and tiny amount of yellow chemicals
- yellow – table salt – sodium chloride
- green – boric acid or borax or copper sulfate
- blue – natural color of the flame
- violet – salt substitute – potassium chloride
Set Up the Demonstration
- I went low-tech here, setting a potholder on my kitchen counter, with a cookie sheet for the demonstration. You can use any fire-safe surface. It’s nice to use a container with a lip because the liquid (water) will run and can make a mess.
- Sprinkle a small amount of each chemical in a line running across your dish. You can mix the colors, but be aware some emission spectra are much stronger than others. For example, any contamination of the other colors with the sodium from table salt will turn your flame mostly yellow, regardless of the other salts that are present.
- When you are ready to perform the demonstration, pump a layer of hand sanitizer across the salts.
- Dim the lights.
- Use a long match or long handled lighter to ignite one end of the gel. The flame will spread across, displaying all the colors.
- Now, the nice thing about hand sanitizer is it’s mostly water. The flame goes out as soon as enough alcohol burns off to make the gel watery enough to extinguish the fire. It doesn’t take long.
- If you want to re-start the demonstration, wait until the flame is out. This is the key mistake people make with the traditional colored fire project, too. Don’t add fuel to a burning fire and expect good things to happen. Once the flame is out, you can apply more alcohol gel and light it. You do not need to add more salts.
Do you have any additional tips or advice for people wishing to perform this demonstration? Feel free to comment.
Here’s the video made when the photograph was taken. Easy. Safe.
Related Posts
Leave a reply cancel reply.
This site uses Akismet to reduce spam. Learn how your comment data is processed .
9 thoughts on “ Safe Method of Doing the Colored Flames Chemistry Demonstration ”
Can I use rubbing alcohol or acetone for it.Ca(NO3)2 for orange???
I use rubbing alcohol for it all the time at home, but that increases the risk of a fire, of course. Yes, calcium compounds yield an orange flame. If you have it, use it :)
I have noticed the bigger and more crystalline the salt the better color it gives to flames
Same here. I’ve found crystalline chunks of solid to show up better than a line of crumbly powder. Perhaps because it has more height and is more accessible to the flame?
Sometimes the blue of the ethanol drowns out the color of the salt, but when this happens, I use a little less hand sanitizer. Unfortunately, that means it doesn’t burn as long.
Any tips for trying to make this look as dramatic and clear as the usual flame test (or even getting the intensity of color in the post picture at the top)?
I credit the intense color I got for the photo to the solvent I used. You may wish to experiment with different types of alcohol or with water-alcohol gels. The solubility of the salt comes into play, so what works best for one compound may not be optimal for another.
The metal Sterno cans used for catering work nicely too, they’re just methanol. You’ll notice though that once you add the chemicals, the gel will become a liquid. My favorites are boric acid for green and lithium perchlorate for pink/red.
Thanks for the Info!
Just remember that it is the vapors that burn; so if you are going to add more solvent, make sure that the container has cooled. Be careful with methanol since it has a higher flash point than hand sanitizer.
Excellent advice! One way to help avoid the chance of a flash is to refresh the fuel by first adding hand sanitizer gel (which helps cool the surface) and then adding methanol (if you’re using both).
- svg]:fill-accent-900">
Gray Matter: How to make colored fire
By Theodore Gray
Posted on Nov 14, 2007 12:00 PM EST
Wood fires are yellow, right? Well, typically, but not necessarily. All hot bodies emit “black body” radiation in a color spectrum that depends on temperature: red for fairly hot, orange for hotter, yellow for really quite hot, all the way up through white for, well, white-hot. Most wood fires burn at about yellow-hot.
But there’s also a second reason for the usual yellow color: something called the “spectral emission line.” Wood (along with most natural substances) contains sodium, which produces a yellow emission when ignited, regardless of the temperature.
A word from our attorney: Conduct this experiment with care, and always wear appropriate safety gear. Children should not attempt this experiment without adult supervision. Theodore Gray is a scientist trained in lab safety procedures. For more information on Gray’s scientific pursuits, visit his website .
If you mix substances with different spectral-emissions lines, they’ll burn different colors. Pyrotechnicians have a long list of chemicals that emit particular colors—combine lines with enough gunpowder to make a big bang and… boom ! Fireworks are born.
One particularly fun chemical used in fireworks is copper (II) chloride, CuCl2, which has a strong blue emission line. Most fireworks-coloring chemicals produce tints only under fairly specific conditions, but with copper chloride you can just throw a handful into an ordinary wood fire (Achtung! Do not try this with a gas fire) , and it will instantly turn the flames a fascinating mixture of blue, green and yellow. The color lasts pretty much as long as the logs do.
Is this safe? Well, sort of. Copper chloride is only moderately toxic, which is to say it’s about the same as ordinary wood smoke. (There’s a reason people have chimneys, and whether you’re using extra chemicals or not, wood smoke is not something you should breathe much of.) Be sensible, have an adult present, and you should be fine.
Another fun and reasonably safe substance to throw into fire is lycopodium powder. This ultrafine yellowish powder, available from fireworks- and magic-supply shops, is a classic substance employed by magicians and alchemists for centuries for impressing the public with a nice ball of fire. A pinch tossed or blown into a fire or candle flame will give off a huge, hot and loud fireball that vanishes as quickly as it appeared. I won’t say it’s harmless—this is real fire, and it could set things around it on fire—but on the other hand, it lasts for only a fraction of a second. ( Achtung! Keep clothes and hair away from the fire ).
Remarkably, lycopodium powder is not a chemical but actually the spores of a certain type of club moss. It burns fast simply because its texture is so fine. Almost any organic matter, even flour or corn dust, can burn explosively if it’s granular enough—lycopodium just happens to be a convenient all-natural fireball powder that is readily available.

How to Make Colored Fire With Home Items

On this new, easy science for kids, you’ll learn how to make colored fire with household items from your own home. Its an entertaining experiment which has few simple steps. Step 1: what do you need? You can choose any of these items to use, each one gives you a different hue. Violet-Ketone, Blue-Alkaline alcohol, Green-Alkaline alcohol, or Copper sulfate (or borax).
Next, get a big container to put the mix in. Preferably large enough to hold at least one quart. A plastic food container will work well. This mixture will make a fairly good hot fire, so it may take a little while to get it going.
When the material is ready, you should have two sticks in the same shapes lying about. The color of the stick depends on the compound you are using. For instance, a blue stick would produce a purple fire. The green stick produces a yellow fire.
Now get a bucket to put the mix into. It should be large enough to hold at least a quart. You want it to be big enough to allow for a long, hot flame. If your fire is short, you may want to increase the size to four quarts.
Finally, light the fire. Put a burner on top of the pan. You can choose a simple ceramic burner if this is all you have. If you have more experience in working with fires, consider getting a gas burner. This type produces smoke rather than flames, and you should not have any problem getting it going.
If you want to know how to make colored fire with home items like clay, this may be easier. Clay will heat up nicely, though you do want to be careful. Do not start out with a full pot. Just heat up some water and stir in the clay. You will also want to add a colorant to help the colors stick.
How To Make Colored Fire With Household Items?
So, how to make colored fire with home items like clay? Again, start with plenty of water. Then make a paste out of cornstarch and water by using a blender or your hands. Next, add a colorant to help the colors stick. Once you have the paste ready, just put it in a jar and heat it over an open flame. It will bubble up as it heats up, but you can release it once it turns black.
Finally, how to make colored fire with home items that you made yourself is to paint it. There are different types of paint, so you may need to get special tools or learn a bit about color tricks. Get a gallon of white paint and a brush. Also make sure you have covered everything with the clay. The only other thing you need is an iron that you can paint on. Get the paint, the iron and a container to hold it in and you are done.
If you get items made by other people, you should ask them how to make painted candles. Of course, you will need the raw materials first. However, if you can find some left over items, you might not need to get new ones. Just repaint them and they will glow in your house. They will also be a lot cheaper than purchasing new.
You can also find how to make colored fire with home items online. Usually you will find instructions for items made out of brass, copper, stainless steel and other metal. However, there are some items made from glass that glow and are sold online as well.
You can even make painted glow sticks and glow bracelets. To make the glow sticks, you will need black construction paper, some rope, wire, and a marker. To make the bracelets, you will need some clear plastic or glass beads, some double-sided tape, some rope, and a marker. You can buy these items at dollar stores or arts and crafts stores in your area.
As you can see, learning how to make painted items is easy. You can find everything you need at a good craft store or online. The sky is the limit when it comes to the projects you can do with these items. So start looking at items made from glass today and start creating some unique projects.
- Recent Posts
- Hiring a Driver for Pets: What to Expect - August 26, 2024
- Innovations in Insurance Analytics SolutionS - June 26, 2024
- The Future of AI-Driven Climate Change Solutions: Innovations and Impact Ahead - May 9, 2024
Related Topics:

How to Test a Product on a Customer
How To Tie Yourself Up With Household Items

Reach out to us for Guest Post opportunities
+44 (0) 1630 312154
[email protected]
177 Lower Road, Hookgate TF9 4QJ
© 2024 Science Oxford Live
Privacy Overview
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |

How to Make Purple Fire – Easy & Interesting Methods
- May 11, 2022
- Chemistry , Science Facts
IIf you have been wondering how to make purple fire, then you have come to the right place. There are many ways to make purple fire.
Today, I will be showing you my personal favorite and most effective ways using simple items you can find at home at the end of the article.
Making Purple Fire – Chemistry Project
Ingredients to make purple fire.
1) Salts of Potassium Chloride in powdered form
2) Red Emergency Flare that contains Strontium Nitrate
3) Fuels that produce a blue flame-like Alcohol-based Sanitizer , Ethanol , Lighter Fluid , Rubbing Alcohol , Propane, or Methanol
That’s it! Collect these supplies to make a purple flame.
Directions to Make Purple Fire
Since purple color is not in the spectrum wavelength, it is a little tricky to produce the purple shade.
But as I said, we can still create purple color from the chemicals that produce the emission spectrum.
To get purple fire, we need to produce and combine red and violet flames. So, let us learn the easy and simple directions that produce a purple flame.

Step-1: As a first step, arrange a campfire set up using either coal or firewood in your room or backyard.
Step-2: For the second step, you need to add some amount of fuel to the campfire set up and light it up using matches. Then, let the fire grows up to one foot a distance upwards.
Note: Fuel is the key element while creating purple color fire because it dominates the yellow embers from the sodium salts of wood. Thus, it results in offering us a beautiful purple fire. In addition, fuel helps in burning with the invisible or blue flames and makes purple color visible to the human eye.
Step-3: The third step is to collect the powdery strontium from the flare! The emergency flare comes in a long cardboard tube accompanied by a striker at one end. And to remove the strontium powder, you need to peel off the cardboard material present on the opposite side to the striker part. Then, you can collect the revealed powdery substance, nothing but strontium into a bowl. Strontium powder is useful for making the purple color much more intense and visible clearly.
Step-4: In the fourth step, we are going to use a very little amount of strontium powder and the chemical salts of potassium chloride to create purple color. All you need to do is sprinkle the gathered materials one after the other over the tip of the fire embers slowly.
Note: Do not drop the chemicals in large quantities and collectively in the middle of the flame. Because this may cause unnecessary fire accidents with the sudden rise in the fire flare-ups.
Step-5: The fifth step is to check there are no harmful gases released because some chemicals are dangerous to people dealing with breathing issues. Continue to do the activity only the fumes released after the chemicals react with the fire are safe! If you feel you are suffocating with the fumes, just step away from the campfire and let the others do the activity and you just watch and enjoy!
Step-6: You land in the sixth step only when you feel it is safe to continue the activity! In the sixth step, drop the chemical powder i.e. potassium chloride slowly over the tip of the fire flames. Also, continue to add strontium powder and fuel in between to see the best results.

Results: The first and little addition of chemicals to the fire may not give many changes to the color of the fire. But still, you need to continue adding chemicals until you see the color change in the fire fumes. So, you must keep adding more and more chemical powders to change the color and make it visible.
Safety Tips
As you are dealing with a lot of chemicals and fire, it is mandatory to wear safety glasses and hand gloves that resist fire and chemical contact. Keep a fire suppressant for safety. We strongly advice you to try this at your backyard in safe conditions.
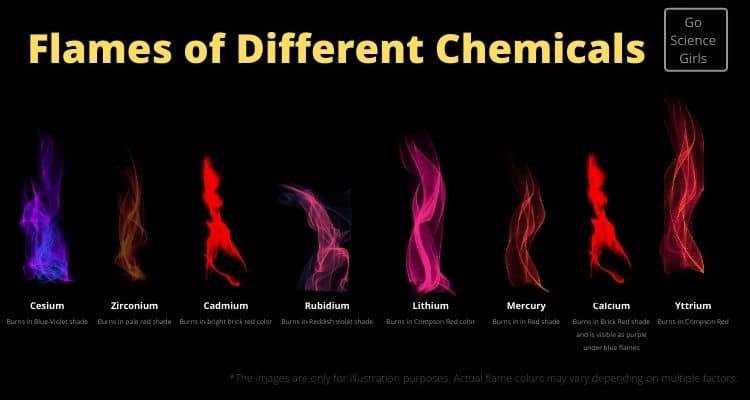
What chemicals burn purple?
Here are the other surprising chemicals that burn purple and its lighter and darker shades:
1) Cesium —Burns in Blue-Violet shade
2) Zirconium —Burns in pale red shade
3) Cadmium —Burns in bright brick red color it is dangerous to use
4) Rubidium —Burns in Reddish violet shade
5) Lithium —Burns in Crimpson Red color
6) Mercury —It is highly toxic and burns in Red shade
7) Calcium —Burns in Brick Red shade and is visible as purple under blue flames
8) Yttrium —Burns in Crimpson Red
How hot is purple fire?
Different color flames possess different degrees of temperature! So, the temperature change in fumes is the main characteristic that represents fuel temperature. As purple fire is the result of a combination of red fire and blue fire, it exhibits lesser temperature than blue fire. So, let us learn the temperatures of a variety of fuels that produce a purple flame.
Ethanol — 1920 °C or 3488 °F
Methanol — 1870 °C or 3398 °F
Propane — 1980 °C or 3596 °F (This temperature recorded in the presence of air)
In the case of black body radiations, the temperature of flames is calculated according to the color of the fire. Have a look at the temperatures of different color flames!
Orange-yellow shaded flames possess 1100 °C or 2012 °F
Deep red color flames exhibit 600-800 °C or 1112-1800 °F
Blue color ones lie in the range between 1400-1650 °C or 2600-3000 °F
White color fumes show 1300-1500 °C or 2400-2700 °F
Violet shade fumes reveal temperatures at 39400 °C or 71000 °F

How to make purple fire at home
There are two methods through which we can make purple fire easily at home. Let us see what they are:
Method-1: Making Wax Cup Cakes
1) Arrange a double boiler set up on the stove and put some old candle wax into it. Let the wax melts on medium flame.
2) Once the wax melts completely, add little amounts of desired chemical powder into it. You need to stir the wax and chemical powder well to avoid lumps formation.
3) Then, switch off the stove and pour the mixture into a mold or paper cup molds.
4) Let the mixture completely cools down. After cooling, the liquid mixture turns hard again in the shape of the mold you used.
5) Now, set up a campfire in your backyard and add these wax cupcakes one or two into the fire. You can see the beautiful purple flames from the fire.

Method-2: Soaking Wood in Chemicals
1) Gather some lightweight fire ingredients like wood, pine cones, scraps of lumber, etc.
2) Take some amount of water in a plastic container and add a tablespoon of your desired chemical compound into it.
3) Then, drop the wood fire materials into the chemical solution prepared. And let them soak for 24 hours.
4) After a day, remove the wood materials and allow them to dry completely. After drying completely, these wood materials are said chemically treated.
5) Now bring these chemically treated wood materials into a campfire.
6) The campfire releases the beautiful purple flames after a minute or a couple of minutes.
Leave a Reply Cancel Reply
Your email address will not be published. Required fields are marked *
Name *
Email *
Add Comment *
Save my name, email, and website in this browser for the next time I comment.
Post Comment
Making Colored Candle Flames
Tips for Making Colored Fire Using a Wick
Philip Evans / Getty Images
- Projects & Experiments
- Chemical Laws
- Periodic Table
- Scientific Method
- Biochemistry
- Physical Chemistry
- Medical Chemistry
- Chemistry In Everyday Life
- Famous Chemists
- Activities for Kids
- Abbreviations & Acronyms
- Weather & Climate
- Ph.D., Biomedical Sciences, University of Tennessee at Knoxville
- B.A., Physics and Mathematics, Hastings College
Have you ever wanted to color the flames of your candles? I've received several questions about how this might be achieved, including the following email:
I just posted this question to the forum but I am also interested in your take on it. I read the article about colored fire and decided to try to make a candle with a color flame!
First I tried dissolving the chems you suggested in the article (such as cupric chloride) into water until it was fully concentrated, and soaking some wicks overnight. After drying the wicks I found that on their own they do burn with a pretty flame (well, some of the chemicals ), but once I tried adding wax to the mixture the natural color of the wax burning completely took away any desired effects.
Next I tried grinding up the chems into a fine powder and mixing as uniformly as possible with the wax. This was also unsuccessful and resulted in sporadic and weak color at best and often wouldn't even stay lit. Even when I could keep the particles from sinking to the bottom of the molten wax, they still [do] not burn correctly. I am convinced that in order to make a functioning candle with a color flame it is necessary to fully dissolve the salts and minerals listed in the article into the wax. Obviously the salts do not naturally dissolve and this got me thinking that maybe an emulsifier is necessary? Does that make sense? Thanks!
If making colored candle flames was easy, these candles would likely be available for sale. They are, but only when the candles burn liquid fuel. I would think you could make an alcohol lamp that burns a colored flame by attaching a wick to an alcohol lamp filled with fuel containing metal salts. The salts could be dissolved in a small amount of water , which would be mixable in alcohol. Some salts dissolve directly in alcohol. It's possible something similar could be achieved using fuel oil. I'm not sure a wax candle would ever work as well. Soaking the wick will produce a colored flame, much as if you burned paper or wood that has been soaked with metal salts, but the wick of a candle burns very slowly. Most of the flame results from the combustion of vaporized wax.
Has anyone tried making candles with colored flames? Do you have any suggestions for the reader who sent this e-mail or any tips about what will/won't work?
I too tried using paraffin wax but to no avail. I searched around and US patent 6921260 is probably the best description on the previous art and its own design, careful reading of the patent reveals that it should be possible to make colored flame candles at home if you know what you’re doing.
Arnold says:
There is an old pdf article dated Dec 26, 1939 entitled Colored Flame Candle. In it William Fredericks used petroleum jelly as a fuel source with the mineral salt suspended in it. Although I haven’t built the whole project, I did suspend copper chloride in petroleum jelly, and it burned very nicely. A nice blue flame. You have to play with the ratios. As I see it, there are two approaches. A. Drill an existing candle from the top, and fill the hole with warmed jelly, or B. Follow the instructions in the article by building a candle around an inner core of jelly. But I was asked a question which I need to answer: Is breathing the smoke of colored flame candles healthy? i.e. copper , strontium , potassium
Perhaps we can put our heads together on this project. I would like to get the colored flame candle project started. I saw that you have tried some things, but found they didn’t work.
I would ask you not post this information yet. I would rather think this through with you and present the final project, rather than to publish the raw thinking of it. On the net I have found very chemically complicated candles (ethanolamine etc.)
I mixed copper I chloride with petroleum jelly, put a wick in it, and it burned very nicely blue. There was some moisture there, so it did stink a bit.
I read in one of the patent papers online that one of the problems is the amount of carbon particles in a candle flame. The suggestion was to use a palladium, vanadium or platinum chloride as a catalyst/accelerant (absorbing a small amount of this material on the wick) to increase the temperature. Not exactly cheap or readily available. But supposedly the orange flame is gone.
The other alternative is to burn smaller chain organic compounds, like citric acid or benzoic acid. I haven’t tried these. Faerie flames advertises their candles are not paraffin, but crystals. Perhaps you have some ideas on other smaller molecules.
I find that alcohol flames color very nicely, but paraffin is just not very hot burning.
Yes, I am knowledgeable in chemistry with a B.Sc. in chemistry.
Chels says:
I am trying to make a color flame candle myself. I think the first step would be producing a candle that burns with a light blue/luminous flame, you need to get rid of the yellow. To do this you need a fuel that has a low carbon content. Things like paraffin and stearin burn yellow due to their high carbon content.
I don’t think it’s possible to make a good color flame candle with paraffin. A lot of patents seem to recommend Trimethyl Citrate. It’s a waxy/crystalline solid that burns a light blue. But I can’t find a place to get it, unless I want to buy it in industrial quantities!
Does anyone know where I can find trimethyl citrate? It’s used as a food additive and cosmetic ingredient so I figure it isn’t toxic.
Amber says:
I see a lot of soy candles on the market. I am wondering if perhaps this may work with soy or beeswax?
Bryan says:
I have had a little success making a bluish candle flame by using copper desoldering braid.
It makes a surprisingly good candle wick. In order to get the color, however, I first heated it up to melt out the impregnated rosin. I then put it in saltwater, put another wire in saltwater (pretty much any metal except aluminum), made sure they didn’t touch, and attached a 9 V battery to the wires—negative to the bare wire, positive to the copper braid. Within seconds, tiny bubbles will come off the – wire and blue-green stuff will form on the + braid. Leave it in for a while. Most of the green stuff will come off the braid into the water. The stuff is most likely copper chloride, formed from the chloride in the salt. After the braid is green (but before it falls apart), pull it out, trying not to knock off too much stuff. Dry it, preferably by hanging. Then try that as a wick.
I’ve only tried limited experiments, so your mileage may vary.
I’m working on Bryan’s idea of using desoldering braid as a wick. I’ve had limited success so far. The theory is good it seems, but the main problem I’ve had is that the "wick" doesn’t seem to be very good at drawing the molten wax up to the flame. The longest I’ve been able to keep one lit is about thirty seconds.
I’m thinking that either I did not allow the wick to remain in the saltwater solution long enough or perhaps I might benefit from a different variety of wax or possibly weaving the braid together with a more traditional wick.
Priyanka says:
take 1.5 cups of water and add 2 tbsp of salt (NaCl). dissolve 4 tbsp of borax. Then dissolve Add 1 tsp. of one of the following chemicals for colored flames: strontium chloride for a brilliant red flame, boric acid for a deep red flame, calcium for a red-orange flame, calcium chloride for a yellow-orange flame, table salt for a bright yellow flame, borax for a yellow-green flame, copper sulfate (blue vitriol/bluestone) for a green flame, calcium chloride for a blue flame, potassium sulfate or potassium nitrate (saltpeter) for a violet flame or Epsom salt for a white flame.
David Tran says:
Wouldn’t the NaCl contaminate the flame with yellow and overpower the other colors?
Tim Billman says:
Check your colors. Boric acid burns green, calcium chloride burns orange/yellow, etc.
I can make solutions of boric acid (which can be bought at Ace Hardware-type stores 99% pure as a cockroach killer) and strontium chloride (an additive from pet stores for saltwater fish tanks) which burn nicely in a mixture of acetone and rubbing alcohol , but those solutions do not mix with melted candle wax (because it is non-polar.) The next thing I was going to try was finding an emulsifying agent that was safe to burn (i.e., probably not soap) to make a semisolid colloid with the compounds dissolved in the wax.
Any ideas on what my emulsifier could be? What can make oil and water mix besides soap?
For colored flames the element burn:
Lithium = Red Potassium = Purple Sulfur = Yellow Copper/copper oxide = Blue/Green
I would just look at the elements and chemicals that they use in fireworks because those burn with different colors.
- Color Change Chemistry Experiments
- Light a Candle With Smoke
- 18 Fun Christmas Chemistry Projects
- Special Effects Science
- How to Get Lithium From a Battery
- Halloween Chemistry Demonstrations
- How To Make Green Flames
- 10 Amazing Chemical Reactions
- How to Do the Flame Test
- Top Halloween Chemistry Projects
- 10 Fun Chemistry Demonstrations and Experiments
- Hydrogen Balloon Explosion Experiment
- How to Make Colored Flowers
- How to Make a Sparkler
- How To Make a Rainbow in a Glass
- How to Make Bath Salts (Scented and Colored)

IMAGES
VIDEO
COMMENTS
One method of getting purple fire is using a chemical that burns violet in the flame test, ideally with a colorless or blue-burning fuel. Alternatively, use a chemical that yields a pink or red flame test and combine this color with a fuel that has a blue flame.
This handy chart shows the chemicals needed to make different flame colors. It’s easy to make colored fire at home in the fireplace or a campfire. All you need to do is sprinkle on a salt to color the flames. Here is a list of colorants, the colors they produce, and a look at where to find them.
You can create a colored fire by sprinkling chemicals in the flames, making wax cakes containing chemicals, or by soaking wood in a water and chemical solution. While making colored flames can be a lot of fun, always exercise caution when working with fire and chemical substances.
Explore how different elements rect when exposed to a flame, and discuss how alkali metals, alkaline earth metals, and metal salts change the colour of fire.
The cream of tartar yielded a purple-colored flame. Purple is associated with the presence of potassium (K). That’s because cream of tartar is a potassium salt.
Why would adding minerals or salts to a fire change its flames? Colored fire raises questions and piques scientific inquiry. As a modern chemistry demonstration, colored flames are used to evoke wonder in students and also to illustrate the emission spectra of metal salts ( the flame test ).
Certain substances make fire burn a specific color. PopSci's columnist, Theodore Gray, explains the science behind these colorful flames.
January 9, 2021. On this new, easy science for kids, you’ll learn how to make colored fire with household items from your own home. Its an entertaining experiment which has few simple steps. Step 1: what do you need? You can choose any of these items to use, each one gives you a different hue.
A quick and simple tutorial for an easy way to make purple fire without using dangerous chemicals. Also we discuss ways to make purple fire at home at the end.
I read the article about colored fire and decided to try to make a candle with a color flame! First I tried dissolving the chems you suggested in the article (such as cupric chloride) into water until it was fully concentrated, and soaking some wicks overnight.