An official website of the United States government
Official websites use .gov A .gov website belongs to an official government organization in the United States.
Secure .gov websites use HTTPS A lock ( Lock Locked padlock icon ) or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.
- Publications
- Account settings
- Advanced Search
- Journal List


Prevention, diagnosis and treatment of cervical cancer: A systematic review of the impact of COVID-19 on patient care
Pietro ferrara, giulia dallagiacoma, federica alberti, leandro gentile, paola bertuccio.
- Author information
- Article notes
- Copyright and License information
Corresponding author at: Department of Public Health, Experimental and Forensic Medicine, University of Pavia, Via Forlanini, 2, 27100 Pavia, Italy.
Received 2022 Mar 21; Revised 2022 Aug 7; Accepted 2022 Sep 17; Issue date 2022 Nov.
Since January 2020 Elsevier has created a COVID-19 resource centre with free information in English and Mandarin on the novel coronavirus COVID-19. The COVID-19 resource centre is hosted on Elsevier Connect, the company's public news and information website. Elsevier hereby grants permission to make all its COVID-19-related research that is available on the COVID-19 resource centre - including this research content - immediately available in PubMed Central and other publicly funded repositories, such as the WHO COVID database with rights for unrestricted research re-use and analyses in any form or by any means with acknowledgement of the original source. These permissions are granted for free by Elsevier for as long as the COVID-19 resource centre remains active.
Worldwide, the COVID-19 pandemic disrupted healthcare services, including cervical cancer management, and an increased burden for this condition is expected. This systematic review synthetizes the available evidence on the impact of the pandemic on prevention, diagnosis and treatment of cervical cancer. Searches were performed on PubMed, Embase, and Scopus for relevant studies on these topics with the purpose of comparing service access and care delivery before and during COVID-19 pandemic. Due to the methodological heterogeneity among the studies, findings were narratively discussed. Of the 715 screened titles and abstracts, 33 articles were included, corresponding to 42 reports that covered the outcomes of interest: vaccination against human papillomavirus (HPV) (6 reports), cancer screening (19), diagnosis (8), and treatment (8). Seven studies observed reductions in HPV vaccination uptake and coverage during COVID-19. Reports on cervical screening and cancer diagnosis activities showed a substantial impact of the pandemic on access to screening services and diagnostic procedures. All but one study that investigated cervical cancer treatment reported changes in the number of women with cervical lesions who received treatments, as well as treatment delay and interruption. With a major impact during the first wave in 2020, COVID-19 and restriction measures resulted in a substantial disruption in cervical cancer prevention and management, with declines in screening and delays in treatment. Taken together, findings from this systematic review calls for urgent policy interventions for recovering cervical cancer prevention and care.
Keywords: Cervical cancer, COVID-19, Human papillomavirus, Cancer screening, Cancer treatment
1. Introduction
Cervical cancer is one of the most frequently diagnosed cancers, and a leading cause of cancer-related death in women ( Zhao et al., 2021 ). The last iteration of the Global Cancer Statistics 2020–GLOBOCAN censused approximately 600,000 global cases and 340,000 deaths in 2020, and both statistics are expected to increase without broad interventions ( Sung et al., 2021 ).
To contrast cervical cancer as a public health problem, the World Health Organization (WHO) steered a global health strategy, being the first time ever that the world has committed to eliminate a cancer ( World Health Organization, 2020 ). Indeed, cervical cancer is both preventable and treatable, and the reduction of its burden includes tertiary interventions ranging from primary prevention strategies to screening campaigns, to effective treatment options ( Peirson et al., 2013 ; Ferrara et al., 2020a ).
Infection with high-risk types of human papillomavirus (HPV) is a necessary cause of cervical cancer, with 12 oncogenic HPV types classified as group 1 carcinogens by the International Agency for Research on Cancer ( Sung et al., 2021 ; Ferrara et al., 2020a ). Vaccination against HPV has proven to offer protective benefits in the reduction of neoplastic lesions' incidence ( Signorelli et al., 2017 ). Again, robust evidence supports the importance of cervical screening for the early detection of cancerous lesions, which positively impacts on invasive cervical cancers' occurrence and mortality ( Peirson et al., 2013 ; Lozano et al., 2020 ; Ferrara et al., 2020b ). HPV vaccination and cervical screening are therefore essential part components of women's health ( Acuti Martellucci et al., 2022 ), while timely and effective treatments for cervical precancerous lesions and cancers have been shortlisted among the most relevant indicators for an effective universal health coverage for women aged 20 years or older ( Lozano et al., 2020 ).
The rapid spread of the coronavirus disease 2019 (COVID-19) caught unprepared healthcare systems ( Ferrara and Albano, 2020 ), and hospitals and other healthcare facilities responded to the increased demand by internal reorganizations, which resulted in diversion of healthcare delivery for nonurgent conditions especially in the first epidemic months of 2020 ( Voza et al., 2021 ; Matenge et al., 2021 ; Odone et al., 2020 ). This disruption of health services predominantly affected primary care services, leading to limitation in activities, reallocation of healthcare workers (HCW), and reduction of patients' access to facilities, as a consequence of the containment measures and fear of contagion ( Ferrara and Albano, 2020 ; Matenge et al., 2021 ).
Along with broad analyses conducted to quantify the direct and indirect effects of the pandemic, some evidence has highlighted a worrisome impact on care of several cancers, including cervical cancer, but the majority of the studies focused on specific parts of prevention or clinical management ( Acuti Martellucci et al., 2021 ; Saxena et al., 2021 ; Medenwald et al., 2022 ; Bonadio et al., 2021 ). Thus, it is crucial to extensively describe the extent of the COVID-19 impact on cervical cancer patients' and care, providing evidence-based support for the planning of flexible and integrated models of care for women.
In the light of the above and considering the importance of ensuring appropriate care for cervical cancer, we conducted the present systematic review with the aim of summarizing epidemiological research on the impact of COVID-19 pandemic on the prevention, diagnosis and treatment of this condition.
This literature revision was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 2020 ( Page et al., 2021 ). Methods were published in advance in the Prospective Register of Systematic Reviews (PROSPERO) with number CRD42022311206.
2.1. Search strategy
Studies were identified surfing the electronic databases PubMed/MEDLINE, Scopus and Embase. We combined a search strategy of free-text terms and MeSH headings for the topics of HPV prevention, and cervical cancer diagnosis and treatment, as well as of the COVID-19 pandemic. The complete search strategy is presented in the Supplementary material (Appendix 1). The strategy was first developed for the PubMed database and subsequently adapted for the others. Efforts to include further possible relevant articles included cross-referencing of the citation lists of the retrieved articles. Searches were performed up to February 8, 2022, exploring evidence published from 2020 onwards. No restriction on publication status was applied.
2.2. Study selection and inclusion criteria
Selection criteria for screening titles and abstracts were as follows: (1) primary reports available in full-text (trial or observational studies such as case-control, cohort or cross-sectional studies); (2) reporting primary data on HPV vaccination coverage, as well as screening programs, diagnostic procedures and treatment of cervical cancer; (3) including data comparison before and after COVID-19 pandemic; (4) studies published in English or Italian. Records that met the following criteria were excluded: (1) studies without measures of the outcomes of interest; (2) not considering the impact of COVID-19; (3) published as narrative review, editorial, or letter to editor.
2.3. Data extraction, data synthesis and quality assessment
Two authors (GD and FA) independently evaluated the retrieved titles, abstracts, and full-texts for inclusion. Possible disagreements were solved through discussion and consultation of a senior author (PF). Data extraction was performed using a pre-piloted spreadsheet elaborated in Microsoft Excel® for Windows (Microsoft Corporation, Redmond, WA, USA). The following baseline characteristics were extracted for each article: first author's last name, year and country of publication, study design, population size and characteristics, source of information, type of outcome of interest (HPV vaccination, and/or cervical cancer screening, diagnosis and treatment) and measures, and main findings.
Due to the significant heterogeneity in methods and outcomes across the retrieved studies, results were not pooled in a meta-analysis but discussed according to the aim to analyze the impact of COVID-19 on cervical cancer care.
The two reviewers (GD and FA) also assessed the methodological quality of the body of found evidence through an adapted version of the Newcastle-Ottawa Scale (aNOS) available in literature (Supplementary Material, Appendix 2), in which reports achieving an aNOS score of 5 or greater were considered high-quality studies ( Wells et al., 2014 ).
3.1. Search results and articles overview
The flow chart of included studies and selection process is presented in Fig. 1 . Overall, the search strategy retrieved a total of 715 articles. After titles' and abstracts' screening, 33 articles met the inclusion criteria, corresponding to 41 reports that covered the different outcomes of interest ( Fig. 1 ): HPV vaccination (6), cervical cancer screening (19) and diagnosis (8), treatment (8).
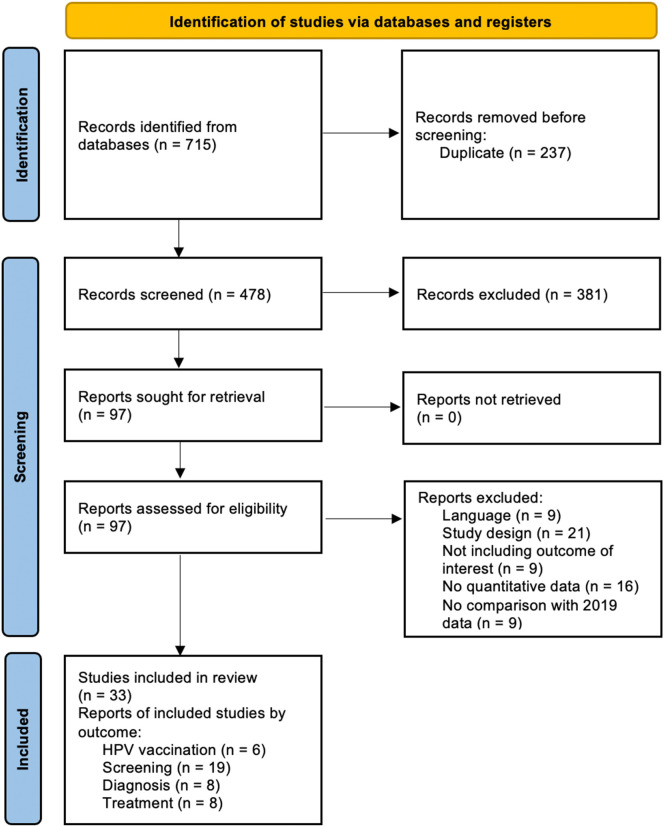
PRISMA flow chart of included studies and selection process.
The characteristics of included studies are presented in Table 1 , Table 2 , Table 3 . All had an observational design and were published after July 2020. Of the total, 16 studies were carried out in America, 13 in Europe, three in Asia, and two in Africa.
Main characteristics of studies including data on HPV vaccination.
Abbreviations: HPV, human papillomavirus; aNOS, adapted version of the Newcastle-Ottawa Scale checklist for assessing the quality of non-randomized studies; NR, not reported; NA, not applicable; 95% CI, 95% confidence interval.
Main characteristics of studies including data on cervical cancer screening.
Abbreviations: aNOS, adapted version of the Newcastle-Ottawa Scale checklist for assessing the quality of non-randomized studies; NR, not reported; NA, not applicable; EMR, electronic medical record; 95% CI 95% confidence interval; CIN, cervical intraepithelial neoplasia; VIA, visual inspection of cervix with acetic acid.
Main characteristics of studies including data on cervical cancer diagnosis.
Abbreviations: aNOS, adapted version of the Newcastle-Ottawa Scale checklist for assessing the quality of non-randomized studies; NR, not reported; NA, not applicable; EMR, electronic medical record; 95% CI 95% confidence interval; CIN, cervical intraepithelial neoplasia.
Methodological quality varied across the 34 studies, of which 32 scored five or more stars on the aNOS quality assessment, while two were classified as low quality given the high risk of bias. Major reasons for bias across studies included lack of representativeness of the sampled participants, as well as substandard assessment of the outcomes as they were mostly self-reported through questionnaires. A more detailed description of the aNOS assessment can be found in Table S1 (Supplementary material).
3.2. HPV vaccination
Six large-scale studies investigated HPV vaccination uptake and coverage during the COVID-19 pandemic from healthcare administrative databases ( Table 1 ) ( Saxena et al., 2021 ; Casey et al., 2022 ; Daniels et al., 2021 ; Gabutti et al., 2021 ; Ramírez et al., 2022 ; Sabbatucci et al., 2022 ). Findings from five reports, which included adolescents and young girls aged 9–26 years, observed a decrease in vaccination coverage starting from March 2020. Of those, two measured the decrease in the number of vaccine doses administered monthly that dropped by the 96% in March–May 2020 in the research by Casey et al. (2022) Three evaluated the population-level vaccination coverage during the whole year, with slight decreases in 13- ( Sabbatucci et al., 2022 ) and 15-year-old adolescents ( Gabutti et al., 2021 ) (respectively −2.2 and −6.6% compared to 2019), as well as a −77% in girls aged 9–26 years during the stay-at-home period ( Daniels et al., 2021 ) ( Table 1 ). Conversely, Ramírez et al. did not find significant variations in vaccination coverage during the pandemic, describing a 0.4% absolute increase in the temporal trend of vaccination coverage in women aged 15–55 years not included in national vaccination programs ( Ramírez et al., 2022 ).
3.3. Screening
The impact of COVID-19 and restrictive measures on cervical screening was identified in 20 reports, which varied considerably according to study periods and outcomes ( Table 2 ), which sourced data from healthcare administrative databases, hospital medical records, cancer registries, pathology records, or web surveys ( Acuti Martellucci et al., 2021 ; Bonadio et al., 2021 ; Davies et al., 2022 ; de Pelsemaeker et al., 2021 ; DeGroff et al., 2021 ; Dema et al., 2022 ; Dennis et al., 2021 ; Desta et al., 2021 ; Doubova et al., 2021 ; Gorin et al., 2021 ; Istrate-Ofițeru et al., 2021 ; Ivanuš et al., 2021 ; Kim et al., 2022 ; Koczkodaj et al., 2021 ; Laing and Johnston, 2021 ; Li et al., 2021 ; Mantellini et al., 2020 ; Meggetto et al., 2021 ; Miller et al., 2021 ; Morais et al., 2021 ; Nogami et al., 2022 ; Ortiz et al., 2021 ; Van Wyk et al., 2021 ; Walker et al., 2021 ). Seven studies evaluated the impact of COVID-19 on the number of Pap smears performed during the first half of 2020: of those, four were conducted in Europe and reported a reduction ranging from 43.3% and 91.5% ( Acuti Martellucci et al., 2021 ; de Pelsemaeker et al., 2021 ; Ivanuš et al., 2021 ; Mantellini et al., 2020 ), while the other conducted in America observed a reduction between 84% and 91.5%, or of more than two-third during the pandemic, compared to the reference periods ( DeGroff et al., 2021 ; Ortiz et al., 2021 ).
Meggetto et al. examined the average monthly screening tests in Canada, finding a decrease of 63.3% between March–August 2020 ( Meggetto et al., 2021 ). Desta et al. highlighted a 54.8% reduction among 30–49-year-old women screened for cervical cancer using Visual Inspection with Acetic Acid during the second quarter of 2020 in Ethiopia ( Desta et al., 2021 ). Four studies carried out their analysis over the whole 2020 year, in which the rate of Pap smears fell down by −7.5 to −68% ( Doubova et al., 2021 ; Ivanuš et al., 2021 ; Laing and Johnston, 2021 ; Walker et al., 2021 ).
Access to screening services was evaluated in four studies. In the United Kingdom, the number of women using screening services dropped from 6% in 2019 to 2.5% in 2020 during the first four months of the year ( Dema et al., 2022 ). In Poland, cytology coverage decreased by two points comparing data of 2019 and 2020 (from 16.34% to 14.35%, between January–September) ( Koczkodaj et al., 2021 ). In a study conducted in USA, compared with the pre-pandemic period, the odds for screening completion among women decreased of 17% and 31% respectively during the stay-at-home and re-opening phases ( Kim et al., 2022 ). A substantial decrease occurred in screening utilization from January 2016 (2.81 per 100 person-months) to July 2020 (0.72 per 100 person-months) in Puerto Rico ( Ortiz et al., 2021 ).
Acuti Martellucci et al. also evaluated COVID-19-induced variations in screening ambulatory services in a province of central Italy, finding 70.3 and 93.1% increases respectively in the numbers of obstetricians' work hours and Pap smears in the second semester of 2020, compared with the same period of 2019 ( Acuti Martellucci et al., 2021 ). Nogami et al. reported that full screening capacity took six months to recover up to pre-pandemic levels, after having reached values as low as 10% in May 2020 ( Nogami et al., 2022 ).
Li et al. surveyed registered physicians who practiced obstetrics and gynecology in Chinese public hospitals, the 60% of which reported a reduction in cervical screening activities from 25% to 100% 2020 (January–August) due to COVID-19, with the most significant reductions observed in cities with more hospital beds and high-level hospitals ( Li et al., 2021 ).
3.4. Diagnosis
Eight studies specifically analyzed the impact of COVID-19 on cervical cancer diagnosis and diagnostic procedures, comparing 2020 with the pre-pandemic period ( Table 3 ). Among these, Davies et al. censused 25.7% less cancer cases between May and October 2020 in United Kingdom ( Davies et al., 2022 ), and van Wyk et al. -7% in April–June 2020 in South Africa ( Van Wyk et al., 2021 ), and Morais et al. -73.4% during the whole 2020 year in Portugal ( Morais et al., 2021 ). Ivanuš highlighted a decrease of 13% in diagnostic invasive procedures in Slovenia in 2020 ( Ivanuš et al., 2021 ). Istrate-Ofițeru et al. found that the number of biopsies and excisional procedures has been decreasing by more than a factor of three in Romania during the pandemic period (March 2020–March 2021) compared to the year before ( Istrate-Ofițeru et al., 2021 ). A decrease in the number of follow-up colposcopy tests was also seen in the pandemic period in two Canadian studies ( Meggetto et al., 2021 ; Walker et al., 2021 ), while in Brazil Bonadio et al. showed that patients had a more advanced-stage at diagnosis during the pandemic, with the proportion of stages III-IVA increased by 13.5% ( Bonadio et al., 2021 ).
3.5. Treatment
Eight of the included reports considered the differences in practice between pre- and pandemic period ( Table 4 ). Data included in the analyses were sourced from population-level healthcare administrative data ( Desta et al., 2021 ; Koczkodaj et al., 2021 ; Meggetto et al., 2021 ), hospital medical records ( Medenwald et al., 2022 ; Istrate-Ofițeru et al., 2021 ; Hathout et al., 2021 ), or cancer screening registry ( Ivanuš et al., 2021 ) in six studies. Of those, all but one reported changes in the delivery of cervical cancer treatment, mostly in terms of decreased number of women with cervical lesion who received treatments, or treatment delays and interruption ( Table 3 ): Altin er al. interviewed 70 gynecologic oncologists: 97.1% reported changes in changes of gynecological cancers due to pandemic situation, in terms of delayed surgery (33.3%) and shift to hypo-fractionated radiotherapy was preferred to standard dose (57.1 vs. 27.1%, respectively), in order to reduce the number of hospital visits ( Altın et al., 2020 ).
Main characteristics of studies including data on cervical cancer treatment.
Abbreviations: aNOS, adapted version of the Newcastle-Ottawa Scale checklist for assessing the quality of non-randomized studies; NR, not reported; NA, not applicable; 95% CI 95% confidence interval; CT, chemotherapy; RT, radiotherapy; LEETZ, Large Loop Excision of the Transformation Zone.

4. Discussion
This is, to the best of our knowledge, the first systematic review that summarizes the current body of evidence about the COVID-19 impact on the care of cervical cancer, with the main goal of comparing service access and care delivery before and during the pandemic.
Although differences in design and setting across the retrieved studies did not allow to provide synthesis measures of this impact, results revealed that COVID-19-related health service reductions have significantly inhibited cervical cancer prevention, diagnosis and treatment, particularly in the very first pandemic months of 2020, which coincided with an important interruption of non-urgent health services ( Walker et al., 2021 ; Conti et al., 2020a ; Balasco et al., 2021 ). More in general, the wide impact on healthcare systems and organizations due to the spread of SARS-CoV-2 have been well-described elsewhere ( Ferrara and Albano, 2020 ; Voza et al., 2021 ; Sabbatucci et al., 2022 ; Conti et al., 2020a ). Major reasons are excessive hospital overload and high shortage of healthcare resources, workload of professionals and their task-shifting to ensure the care of COVID-19 and acute life-threatening conditions, as well as internal rearrangements of their routine activities and closure of certain services, which resulted in a critical amount of patients referred during the pandemic ( Ferrara and Albano, 2020 ; Voza et al., 2021 ; Acuti Martellucci et al., 2021 ; Viganò et al., 2020 ; Conti et al., 2020b ; Della Valle et al., 2021 ), Yet, healthcare users' and patients' fear of contagion and long quarantine due to COVID-19 might have contributed to less usage of preventive health services and facilities ( Wilson et al., 2021 ; Mantica et al., 2020 ; Antonazzo et al., 2022 ; Bittleston et al., 2022 ).
With regards to HPV vaccination, the routine immunization services had significant disruptions amid the COVID-19 pandemic and social distancing measures, with drops in immunization coverage that depended on vaccines, contexts and populations studied ( Ramírez et al., 2022 ; Sabbatucci et al., 2022 ). This review found that the pandemic led to declines in the administration of HPV vaccines, although data vary greatly from context to context. Beyond the crude prevalence of vaccination uptake and coverage during COVID-19, it should be remarked that vaccination of adolescent girls (and other at-risk individuals) is the most effective long-term intervention for reducing the risk of cervical cancer ( World Health Organization, 2020 ), and it is well-recognized that even small decrease in vaccination rates could have significant long-term public health and economic consequences attributable to burden of preventable diseases ( Lo and Hotez, 2017 ). Continued organizational efforts are therefore required to reach at-risk population (particularly young people) to protect from HPV infection ( World Health Organization, 2020 ; Gabutti et al., 2021 ; Sabbatucci et al., 2022 ). It is worth also noting that infection with HPV is a necessary – although not sufficient – cause of human cancer other than to cervix, including carcinoma of the larynx, oropharynx and oral cavity, as well as vulvar, vaginal, anal, and penile tumors ( Ferrara et al., 2020a ; Ferrara et al., 2020b ).
Of particular concern is the lower percentage of screening and diagnosis preneoplastic/neoplastic lesions of the cervix during the COVID-19 pandemic highlighted in this literature review, along with worrisome decreases of screening test performed compared to non-pandemic periods. Results showed huge variations in the extent of the reported reductions, which were likely patterned by screening policies and intensity and types of COVID-19 responses across the different contexts, as well as by study's type, setting and population. In some cases, screening participation was found to remain low despite the efforts made to address the backlog attributable to lockdown measures in the late phase of the pandemic ( Acuti Martellucci et al., 2021 ; Kim et al., 2022 ; Miller et al., 2021 ). This may be due either to a decrease in cervical screening addressability or to a tendency to access the medical system, explained by the fear of not being infected with SARS-CoV-2 virus ( Istrate-Ofițeru et al., 2021 ; Wilson et al., 2021 ).
This systematic review also reveals an evident pandemic deficit in the number of diagnoses and diagnostic procedures, compared with pre-COVID-19 period, as well as a decreased number of patients admitted with cervical intraepithelial neoplasia ( Istrate-Ofițeru et al., 2021 ; Ivanuš et al., 2021 ). Again, a higher number of advanced stages at the new diagnosis or particularly long intervals from tumor biopsy to the first cancer center were also seen in the post pandemic period ( Bonadio et al., 2021 ). Of note, the fundamental aims of cervical screening and early diagnosis is to reduce the burden and subsequent mortality from invasive cervical cancer ( Peirson et al., 2013 ). These came in addition to reduced adherence to surgical treatments and chemo- and radio-therapies for patients diagnosed with high-grade dysplasia ( Davies et al., 2022 ; Ivanuš et al., 2021 ), even in those contexts without suspension of cancer screening during the corona lockdown ( Medenwald et al., 2022 ). Indeed, the COVID-19 emergency and related responses have negatively impacted on several components of cervical cancer prevention and care, a type of tumor in which no delays of diagnosis and treatment can be accepted due to the rapidity of its proliferation ( Medenwald et al., 2022 ).
In brief, our systematic review provides context to strengthen the health services response to meet cervical cancer patients' needs, as well as to promote health education initiatives tending to address women's awareness and attitudes towards HPV vaccination and cervical screening. In this frame, more research is needed to understand the exact extent of COVID-19 impact on cervical cancer diagnosis and management, including the potentially damaging effects of the screening program pause and delays in diagnosis on patients' survival ( Sud et al., 2020 ; Smith et al., 2021 ; Burger et al., 2021 ). For instance, recent analyses fitted provisional models to predict the excess of cases caused by pauses in cervical cancer care over the next years, advocating the build of healthcare extra capacity to ensure patients' access to screening programs and cancer therapies before their disease progresses to advanced disease stages ( Davies et al., 2022 ; Castanon et al., 2021 ). Furthermore, other modeling studies have suggested that 6- to 12-month disruptions to screening may result in only nominal changes in cervical cancer burden ( Smith et al., 2021 ; Burger et al., 2021 ). It is also worth mentioning that reduction of new diagnoses, although temporary, results in shift towards higher stage at diagnosis and thus and an increased healthcare and social cancer burden in the next years ( Maringe et al., 2020 ; Cantini et al., 2022 ).
The mentioned WHO strategy for the elimination cervical cancer as a public health problem fixed a 90–70–90 target, which specifically refers to 90% of girls fully vaccinated with HPV vaccine by age 15 years; 70% of women screened with a high-performance test by 35 years of age and again by 45 years of age; 90% of women identified with cervical disease receive treatment (90% of women with precancer treated, and 90% of women with invasive cancer managed) ( World Health Organization, 2020 ). According to the studies here summarized, COVID-19 strongly inhibits the attainment of these goals – which were assumed to be achieved by 2030 –, and the mentioned global public health efforts and urgent policy interventions are needed to create an innovative pipeline for recovering cervical cancer care from prevention to treatment, particularly through additional HPV vaccination and cervical screening campaigns. In doing so, health services research should further analyze local experiences which have successfully addressed lockdown backlog by virtue of prompt adaptation of services and reorganization of obstetrician activities to minimize COVID-19 impact. These experiences may serve as reference model for the implementation of sustainable and effective changes in other on sexual and reproductive healthcare contexts ( Acuti Martellucci et al., 2022 ; Acuti Martellucci et al., 2021 ; Campbell et al., 2021 ).
Some limitations must be considered in this systematic review. First, despite being systematic in nature, the search strategy was limited to literature databases and did not include surveillance reports. However, the assessment of the evidence was in line with the minimum requirements (at least two databases) set by the PRISMA guidelines ( Page et al., 2021 ), and collected the most updated available studies daily on COVID-19. Moreover, cross-referencing of the citation list was also consulted in order to collect and analyze all the available evidence. Nevertheless, at the time of study, evidence about this topic is still relatively sparse and the literature so far available does not allow us to consider the exact extent of the pandemic-related service reduction and its consequences. For these reasons and due to limited quality of data and reporting, these findings should be interpreted with caution, and require further exploration in studies specifically designed to examine the long-term effects of COVID-19 on cervical cancer prevention, diagnosis and treatment. Lastly, disparity across the included reports regarding data types and sources (e.g., administrative data, survey-based self-reported data, individual-level medical data, etc.) makes difficult drawing definite conclusions of this qualitative analysis. Despite the listed limitations, the present study is a comprehensive synthesis of these topics, providing important insights for public health and policymakers.
5. Conclusion
In conclusion, this systematic review provides context to highlight how the prevention, diagnosis and treatment of cervical cancer suffered from the consequences of COVID-19 pandemic. Our results offer actionable metrics of this impact, which could be used to develop health services response, and call for more resilient and sustainable targeted interventions aimed at guaranteeing the access to quality healthcare and prevention to women, as well as at meeting the WHO targets for the elimination of cervical cancer among the major public health problems.
This research received no external funding.
CRediT authorship contribution statement
Pietro Ferrara: Conceptualization, Methodology, Data curation, Project administration, Validation, Writing – original draft, Writing – review & editing. Giulia Dallagiacoma: Conceptualization, Data curation, Formal analysis, Investigation, Writing – review & editing. Federica Alberti: Data curation, Formal analysis, Investigation, Writing – review & editing. Leandro Gentile: Investigation, Visualization, Writing – review & editing. Paola Bertuccio: Investigation, Validation, Visualization, Writing – review & editing. Anna Odone: Conceptualization, Methodology, Project administration, Supervision, Validation, Writing – review & editing.
Declaration of Competing Interest
The authors have no conflicts of interest to disclose.
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ypmed.2022.107264 .
Appendix A. Supplementary data
Supplementary material.
Data availability
Data will be made available on request.
- Acuti Martellucci C., Morettini M., Flacco M.E., et al. Delivering cervical cancer screening during the COVID-19 emergency. BMJ Sex Reprod. Health. 2021;47(4):296–299. doi: 10.1136/bmjsrh-2021-201099. [ DOI ] [ PubMed ] [ Google Scholar ]
- Acuti Martellucci C., Morettini M., Brotherton J.M., et al. Impact of a human papillomavirus vaccination programme within organized cervical cancer screening: cohort study. Cancer Epidemiol. Biomarkers Prev. 2022 doi: 10.1158/1055-9965.EPI-21-0895. Published online January 13. cebp.0895.2021. [ DOI ] [ PubMed ] [ Google Scholar ]
- Altın D., Yalçın İ., Khatib G., et al. Management of gynecological cancers in the COVID-19 era: a survey from Turkey. J. Turk. Ger. Gynecol. Assoc. 2020;21(4):265–271. doi: 10.4274/jtgga.galenos.2020.2020.0071. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Antonazzo I.C., Fornari C., Maumus-Robert S., et al. Antidepressants drug use during COVID-19 waves in the Tuscan general population: an interrupted time-series analysis. J. Pers. Med. 2022;12(2):178. doi: 10.3390/jpm12020178. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Balasco N., d’Alessandro V., Ferrara P., Smaldone G., Vitagliano L. Analysis of the time evolution of COVID-19 lethality during the first epidemic wave in Italy. Acta Biomed. Atenei Parm. 2021;92(2) doi: 10.23750/abm.v92i2.11149. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Bittleston H., Goller J.L., Temple-Smith M., Hocking J.S., Coombe J. “I didn’t want to visit a doctor unless it was extremely necessary”: perspectives on delaying access to sexual and reproductive health care during the COVID-19 pandemic in Australia from an online survey. Aust. J. Prim. Health. 2022 doi: 10.1071/PY21239. Published online February 3. [ DOI ] [ PubMed ] [ Google Scholar ]
- Bonadio R.C., Messias A.P., Moreira O.A., et al. Impact of the COVID-19 pandemic on breast and cervical cancer stage at diagnosis in Brazil. ecancermedicalscience. 2021;15 doi: 10.3332/ecancer.2021.1299. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Burger E.A., Jansen E.E., Killen J., et al. Impact of COVID-19-related care disruptions on cervical cancer screening in the United States. J. Med. Screen. 2021;28(2):213–216. doi: 10.1177/09691413211001097. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Campbell K.J., Barlow-Evans R., Jewell S., Woodhead N., Singh R., Jaffer K. ‘Our COVID-19 cloud silver lining’: the initiation and progress of postnatal contraception services during the COVID-19 pandemic in a UK maternity hospital. BMJ Sex Reprod. Health. 2021;47(3):224–227. doi: 10.1136/bmjsrh-2020-200764. [ DOI ] [ PubMed ] [ Google Scholar ]
- Cantini L., Mentrasti G., Russo G.L., et al. Evaluation of COVID-19 impact on DELAYing diagnostic-therapeutic pathways of lung cancer patients in Italy (COVID-DELAY study): fewer cases and higher stages from a real-world scenario. ESMO Open. 2022;7(2) doi: 10.1016/j.esmoop.2022.100406. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Casey S.M., Jansen E., Drainoni M.L., Schuch T.J., Leschly K.S., Perkins R.B. Long-term multilevel intervention impact on human papillomavirus vaccination rates spanning the COVID-19 pandemic. J. Low Genit. Tract Dis. 2022;26(1):13–19. doi: 10.1097/LGT.0000000000000648. [ DOI ] [ PubMed ] [ Google Scholar ]
- Castanon A., Rebolj M., Pesola F., Sasieni P. Recovery strategies following COVID-19 disruption to cervical cancer screening and their impact on excess diagnoses. Br. J. Cancer. 2021;124(8):1361–1365. doi: 10.1038/s41416-021-01275-3. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Conti S., Ferrara P., Mazzaglia G., et al. Magnitude and time-course of excess mortality during COVID-19 outbreak: population-based empirical evidence from highly impacted provinces in northern Italy. ERJ Open Res. 2020;6(3):00458–02020. doi: 10.1183/23120541.00458-2020. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Conti S., Ferrara P., Fornari C., et al. Estimates of the initial impact of the COVID-19 epidemic on overall mortality: evidence from Italy. ERJ Open Res. 2020;6(2):00179–02020. doi: 10.1183/23120541.00179-2020. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Daniels V., Saxena K., Roberts C., et al. Impact of reduced human papillomavirus vaccination coverage rates due to COVID-19 in the United States: a model based analysis. Vaccine. 2021;39(20):2731–2735. doi: 10.1016/j.vaccine.2021.04.003. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Davies J.M., Spencer A., Macdonald S., et al. Cervical cancer and COVID —an assessment of the initial effect of the pandemic and subsequent projection of impact for women in England: a cohort study. BJOG Int. J. Obstet. Gynaecol. 2022 doi: 10.1111/1471-0528.17098. Published online February 6. 1471-0528.17098. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- de Pelsemaeker M.C., Guiot Y., Vanderveken J., Galant C., Van Bockstal M.R. The impact of the COVID-19 pandemic and the associated Belgian governmental measures on cancer screening, surgical pathology and cytopathology. Pathobiology. 2021;88(1):46–55. doi: 10.1159/000509546. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- DeGroff A., Miller J., Sharma K., et al. COVID-19 impact on screening test volume through the National Breast and Cervical Cancer early detection program, January–June 2020, in the United States. Prev. Med. 2021;151 doi: 10.1016/j.ypmed.2021.106559. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Della Valle P., Fabbri M., Madotto F., et al. Occupational exposure in the Lombardy Region (Italy) to SARS-CoV-2 infection: results from the MUSTANG–OCCUPATION–COVID-19 study. Int. J. Environ. Res. Public Health. 2021;18(5):2567. doi: 10.3390/ijerph18052567. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Dema E., Gibbs J., Clifton S., et al. Initial impacts of the COVID-19 pandemic on sexual and reproductive health service use and unmet need in Britain: findings from a quasi-representative survey (Natsal-COVID) Lancet Public Health. 2022;7(1):e36–e47. doi: 10.1016/S2468-2667(21)00253-X. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Dennis L.K., Hsu C.H., Arrington A.K. Reduction in standard cancer screening in 2020 throughout the U.S. Cancers. 2021;13(23) doi: 10.3390/cancers13235918. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Desta A.A., Woldearegay T.W., Gebremeskel E., et al. Impacts of COVID-19 on essential health services in Tigray, northern Ethiopia: a pre-post study. Pastakia SD, ed. PLoS ONE. 2021;16(8):e0256330. doi: 10.1371/journal.pone.0256330. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Doubova S.V., Leslie H.H., Kruk M.E., Pérez-Cuevas R., Arsenault C. Disruption in essential health services in Mexico during COVID-19: an interrupted time series analysis of health information system data. BMJ Glob. Health. 2021;6(9) doi: 10.1136/bmjgh-2021-006204. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Ferrara P., Albano L. COVID-19 and healthcare systems: what should we do next? Public Health. 2020;185:1–2. doi: 10.1016/j.puhe.2020.05.014. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Ferrara P., Conti S., Agüero F., et al. Estimates of cancer mortality attributable to carcinogenic infections in Italy. Int. J. Environ. Res. Public Health. 2020;17(23):8723. doi: 10.3390/ijerph17238723. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Ferrara P., Agüero F., Masuet-Aumatell C., Ramon-Torrell J.M. Burden of cancer mortality attributable to carcinogenic infections in Spain. Med. Clín. 2020;154(10):394–397. doi: 10.1016/j.medcli.2019.11.005. [ DOI ] [ PubMed ] [ Google Scholar ]
- Gabutti G., d’Anchera E., De Motoli F., Savio M., Stefanati A. Human Papilloma Virus vaccination: focus on the Italian situation. Vaccines. 2021;9(12):1374. doi: 10.3390/vaccines9121374. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Gorin S.N.S., Jimbo M., Heizelman R., Harmes K.M., Harper D.M. The future of cancer screening after COVID-19 may be at home. Cancer. 2021;127(4):498–503. doi: 10.1002/cncr.33274. [ DOI ] [ PubMed ] [ Google Scholar ]
- Hathout L., Ennis R.D., Mattes M.D., et al. The impact of COVID-19 on brachytherapy during the pandemic: a Rutgers-Robert Wood Johnson Barnabas health multisite experience. Adv. Radiat. Oncol. 2021;6(1) doi: 10.1016/j.adro.2020.10.013. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Istrate-Ofițeru A.M., Berbecaru E.I.A., Ruican D., et al. The influence of SARS-CoV-2 pandemic in the diagnosis and treatment of cervical dysplasia. Med. (Mex). 2021;57(10):1101. doi: 10.3390/medicina57101101. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Ivanuš U., Jerman T., Gašper Oblak U., et al. The impact of the COVID-19 pandemic on organised cervical cancer screening: the first results of the Slovenian cervical screening programme and registry. Lancet Reg. Health - Eur. 2021;5 doi: 10.1016/j.lanepe.2021.100101. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Kim E., Kojima N., Vangala S., et al. Impact of COVID-19 on primary care quality measures in an academic integrated health system. J. Gen. Intern. Med. 2022 doi: 10.1007/s11606-021-07193-7. Published online January 26. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Koczkodaj P., Sulkowsa U., Kaminski M. SARS-CoV-2 as a new possible long-lasting determining factor impacting cancer death numbers. Based on the example of breast, colorectal and cervical cancer in Poland. Nowotw. J Oncol. 2021;71(1):42–46. doi: 10.5603/NJO.2021.0007. [ DOI ] [ Google Scholar ]
- Laing S., Johnston S. Estimated impact of COVID-19 on preventive care service delivery: an observational cohort study. BMC Health Serv. Res. 2021;21(1):1107. doi: 10.1186/s12913-021-07131-7. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Li L., Cao Y., Fan J., et al. Impact of COVID-19 pandemic on the clinical activities in obstetrics and gynecology: a national survey in China. Front. Med. 2021;8 doi: 10.3389/fmed.2021.633477. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Lo N.C., Hotez P.J. Public health and economic consequences of vaccine hesitancy for measles in the United States. JAMA Pediatr. 2017;171(9):887. doi: 10.1001/jamapediatrics.2017.1695. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Lozano R., Fullman N., Mumford J.E., et al. Measuring universal health coverage based on an index of effective coverage of health services in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396(10258):1250–1284. doi: 10.1016/S0140-6736(20)30750-9. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Mantellini P., Battisti F., Armaroli P., et al. Ritardi maturati dai programmi di screening oncologici ai tempi del COVID-19 in Italia, velocita della ripartenza e stima dei possibili ritardi diagnostici. Epidemiol. Prev. 2020;44(5–6 Suppl 2):344–352. doi: 10.19191/EP20.5-6.S2.136. [ DOI ] [ PubMed ] [ Google Scholar ]
- Mantica G., Riccardi N., Terrone C., Gratarola A. Non-COVID-19 visits to emergency departments during the pandemic: the impact of fear. Public Health. 2020;183:40–41. doi: 10.1016/j.puhe.2020.04.046. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Maringe C., Spicer J., Morris M., et al. The impact of the COVID-19 pandemic on cancer deaths due to delays in diagnosis in England, UK: a national, population-based, modelling study. Lancet Oncol. 2020;21(8):1023–1034. doi: 10.1016/S1470-2045(20)30388-0. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Matenge S., Sturgiss E., Desborough J., Hall Dykgraaf S., Dut G., Kidd M. Ensuring the continuation of routine primary care during the COVID-19 pandemic: a review of the international literature. Fam. Pract. 2021 doi: 10.1093/fampra/cmab115. Published online October 6. cmab115. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Medenwald D., Brunner T., Christiansen H., et al. Shift of radiotherapy use during the first wave of the COVID-19 pandemic? An analysis of German inpatient data. Strahlenther Onkol. 2022 doi: 10.1007/s00066-021-01883-1. Published online January 7. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Meggetto O., Jembere N., Gao J., et al. The impact of the COVID-19 pandemic on the Ontario Cervical Screening Program, colposcopy and treatment services in Ontario, Canada: a population-based study. BJOG Int. J. Obstet. Gynaecol. 2021;128(9):1503–1510. doi: 10.1111/1471-0528.16741. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Miller M.J., Xu L., Qin J., et al. Impact of COVID-19 on cervical cancer screening rates among women aged 21–65 years in a large integrated health care system — Southern California, January 1–September 30, 2019, and January 1–September 30, 2020. MMWR Morb. Mortal. Wkly Rep. 2021;70(4):109–113. doi: 10.15585/mmwr.mm7004a1. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Morais S., Antunes L., Rodrigues J., Fontes F., Bento M.J., Lunet N. The impact of the coronavirus disease 2019 pandemic on the diagnosis and treatment of cancer in Northern Portugal. Eur. J. Cancer Prev. 2021 doi: 10.1097/CEJ.0000000000000686. Publish Ahead of Print. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Nogami Y., Makabe T., Komatsu H., et al. Impact of COVID-19 on cervical cancer screening in Japan: a survey of population-based screening in urban Japan by the Japan Society of Gynecologic Oncology. J. Obstet. Gynaecol. Res. 2022;48(3):757–765. doi: 10.1111/jog.15130. [ DOI ] [ PubMed ] [ Google Scholar ]
- Odone A., Delmonte D., Scognamiglio T., Signorelli C. COVID-19 deaths in Lombardy, Italy: data in context. Lancet Public Health. 2020;5(6) doi: 10.1016/S2468-2667(20)30099-2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Ortiz A.P., Gierbolini-Bermúdez A., Ramos-Cartagena J.M., et al. Cervical cancer screening among medicaid patients during natural disasters and the COVID-19 pandemic in Puerto Rico, 2016 to 2020. JAMA Netw. Open. 2021;4(10) doi: 10.1001/jamanetworkopen.2021.28806. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Page M.J., McKenzie J.E., Bossuyt P.M., et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021 doi: 10.1136/bmj.n71. Published online March 29. n71. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Peirson L., Fitzpatrick-Lewis D., Ciliska D., Warren R. Screening for cervical cancer: a systematic review and meta-analysis. Syst. Rev. 2013;2(1):35. doi: 10.1186/2046-4053-2-35. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Ramírez M., Fuente J., Andía D., José Hernández J., Fiol G., Torné A. HPV vaccination coverage in women between 15 and 55 years old in Spain: temporal trend during the period 2007–2020. Int. J. Gynecol. Obstet. 2022 doi: 10.1002/ijgo.14067. Published online January 7. ijgo.14067. [ DOI ] [ PubMed ] [ Google Scholar ]
- Sabbatucci M., Odone A., Signorelli C., et al. Childhood immunisation coverage during the COVID-19 epidemic in Italy. Vaccines. 2022;10(1):120. doi: 10.3390/vaccines10010120. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Saxena K., Marden J.R., Carias C., et al. Impact of the COVID-19 pandemic on adolescent vaccinations: projected time to reverse deficits in routine adolescent vaccination in the United States. Curr. Med. Res. Opin. 2021;37(12):2077–2087. doi: 10.1080/03007995.2021.1981842. [ DOI ] [ PubMed ] [ Google Scholar ]
- Signorelli C., Odone A., Ciorba V., et al. Human papillomavirus 9-valent vaccine for cancer prevention: a systematic review of the available evidence. Epidemiol. Infect. 2017;145(10):1962–1982. doi: 10.1017/S0950268817000747. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Smith M.A., Burger E.A., Castanon A., et al. Impact of disruptions and recovery for established cervical screening programs across a range of high-income country program designs, using COVID-19 as an example: a modelled analysis. Prev. Med. 2021;151 doi: 10.1016/j.ypmed.2021.106623. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Sud A., Torr B., Jones M.E., et al. Effect of delays in the 2-week-wait cancer referral pathway during the COVID-19 pandemic on cancer survival in the UK: a modelling study. Lancet Oncol. 2020;21(8):1035–1044. doi: 10.1016/S1470-2045(20)30392-2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Sung H., Ferlay J., Siegel R.L., et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021;71(3):209–249. doi: 10.3322/caac.21660. [ DOI ] [ PubMed ] [ Google Scholar ]
- Van Wyk A.C., De Jager L.J., Razack R., et al. The initial impact of the COVID-19 pandemic on the diagnosis of new cancers at a large pathology laboratory in the public health sector, Western Cape Province, South Africa. S. Afr. Med. J. Suid-Afr. Tydskr Vir Geneeskd. 2021;111(6):570–574. [ PubMed ] [ Google Scholar ]
- Viganò M., Voza A., Harari S., et al. Letter to the editor: clinical management of nonrespiratory diseases in the COVID-19 pandemic: what have we done and what needs to be done? Telemed. E-Health. 2020;26(10):1206–1208. doi: 10.1089/tmj.2020.0148. [ DOI ] [ PubMed ] [ Google Scholar ]
- Voza A., Desai A., Luzzi S., et al. Clinical outcomes in the second versus first pandemic wave in Italy: impact of hospital changes and reorganization. Appl. Sci. 2021;11(19):9342. doi: 10.3390/app11199342. [ DOI ] [ Google Scholar ]
- Walker M.J., Meggetto O., Gao J., et al. Measuring the impact of the COVID-19 pandemic on organized cancer screening and diagnostic follow-up care in Ontario, Canada: a provincial, population-based study. Prev. Med. 2021;151 doi: 10.1016/j.ypmed.2021.106586. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Wells G.A., Shea B., O’Connell D., Paterson J., Welch V., Losos M. The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Published online. 2014 http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp [ Google Scholar ]
- Wilson R., Quinn-Scoggins H., Moriarty Y., et al. Intentions to participate in cervical and colorectal cancer screening during the COVID-19 pandemic: a mixed-methods study. Prev. Med. 2021;153 doi: 10.1016/j.ypmed.2021.106826. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- World Health Organization . World Health Organization; 2020. Global Strategy to Accelerate the Elimination of Cervical Cancer as a Public Health Problem. https://apps.who.int/iris/handle/10665/336583 Accessed March 9, 2022. [ Google Scholar ]
- Zhao M., Wu Q., Hao Y., et al. Global, regional, and national burden of cervical cancer for 195 countries and territories, 2007–2017: findings from the Global Burden of Disease Study 2017. BMC Womens Health. 2021;21(1):419. doi: 10.1186/s12905-021-01571-3. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data availability statement.
- View on publisher site
- PDF (976.9 KB)
- Collections
Similar articles
Cited by other articles, links to ncbi databases.
- Download .nbib .nbib
- Format: AMA APA MLA NLM
Add to Collections

IMAGES
VIDEO