- Monash Online

Student Academic Success
- 1:1 Consultation 1:1 Consultation
- Study better Study better
- Build digital capabilities Build digital capabilities
- Understand assessments Understand assessments
- Excel at writing Excel at writing
- Enhance your thinking Enhance your thinking
- Present confidently Present confidently
- Collaborate with others Collaborate with others
- Improve your academic English Improve your academic English
- Maintain academic integrity Maintain academic integrity
- Advance your graduate studies Advance your graduate studies
- O-week workshops O-week workshops
- Let's Talk Assessments Let's Talk Assessments
- Set up for academic success Set up for academic success
- Writing classes Writing classes
- End of semester End of semester
- PhD writing workshops and courses PhD writing workshops and courses
- First Year Undergraduate Success First Year Undergraduate Success
- Graduate coursework success Graduate coursework success
- Academic Language Skills Analysis Academic Language Skills Analysis
- Mathematics skills analysis Mathematics skills analysis
- Orientation Orientation
- SAS cafe SAS cafe
- SAS pop up SAS pop up
- Written Feedback Written Feedback
- About us About us
- Skip to content
- Skip to navigation
Features of a research proposal
The structure of a research proposal includes various elements. The elements outlined below are applicable to proposals for studies situated in the humanities and those situated in STEM fields but there may be different expectations about the ordering or naming of particular sections. It is best to check any specific discipline or unit/ faculty requirements and preferences.
The title of your proposal should clearly summarise what the research is about – it must be descriptive. Use key terms and be direct. Avoid lengthy titles with too much information.
The examples below are from research proposals. They incorporate key terms, and are direct and descriptive.
Melody recognition at varying frequency ranges in cochlear implant users.
Empowering indigenous women in education in rural Australia.
Structural Analysis of Aromatic Polyketide Synthase for the Design of Novel Aromatic Polyketide Natural Products of Pharmaceutical Importance.
Strain Sensitive Array for the Study of Bone Surface Mechanics.
Evaluation of historical media reporting of gender violence.
Developing Linguistics Data Commons in Australia.
Effects of music therapy on infants undergoing medical procedures.
Integrated social sciences curriculum developments in Australian regional Middle Schools.
Collaborative performance making in communities recovering from catastrophic events.
‘New world’ symbolism in John Donne’s poetry.
Introduction
Introduce the topic that is the focus of this proposal. Continue the introduction by providing a summary overview of what the reader will find in the full research proposal. Include a brief reference to the aims of the research, what will frame the research (this could be the literature that you will review and/or the theory that you will use to guide your study and the methodology and/or methods you will use to gather data. While you might write a draft of this section at the outset, it is often written last to capture the essence of the research project.
Read the example from an IT research proposal. Click to find out more about the contents of a research proposal introduction.
Definitions
You’ll need to provide the meaning of key terms that you refer to in your proposal, if this is relevant. Do not assume your readers are familiar with your topic; so write the proposal as if an unfamiliar but knowledgeable person is reading it. These terms may be integrated in your introduction or clarified in the literature review section. Some research proposals include a glossary of key terms and abbreviations in the Appendix section.
Definitions should be clear and concise, and may include subject-specific vocabulary. Definitions may be descriptive and contain a brief illustrative example.
Source: Deidentified student proposal. (n.d.). Contribution to the science behind alternative energy utilization: Developing the fundamental chemistry behind solar energy conversion using Ni, Pd, Pt metal complexes and small molecule activation. https://archive.urop.uci.edu/SURP/sample_proposals/SURP%20Physical%20Sciences%205.pdf
Source: Deidentified student proposal. (n.d.). Melody recognition at varying frequency ranges in cochlear implant users. https://archive.urop.uci.edu/SURP/sample_proposals/SURP%20Medicine%202.pdf Accessed: 19/1/2023.
Literature review
The literature review provides the scholarly context that positions your research. In academic research, it is always important to acknowledge previous work that has been done that is relevant to work that will be undertaken. It shows what has already been done in the field, the gaps in research, and the relationship between previous research and your proposed research. The synthesis of previous research literature informs the aims, questions and/or hypothesis of your research proposal.
Read the example from a medical research proposal. Click to find out more about the contents of a research proposal literature review.
Research questions / hypothesis
Disciplinary traditions will determine whether a research proposal will identify research questions or a hypothesis. Both provide the focus of the intended research project. The research proposal in its entirety is a justification of an effective approach to answer the research questions or to test the hypothesis.
In this section of the proposal, explicitly state the question(s) that the research will address. They need to be in the form of questions and they need to be specific and answerable through the method that you have proposed in your proposal. They should relate directly to the overall topic you have discussed and incorporate any concepts of variables that you have discussed earlier in the proposal.
How have school intervention programs affected the literacy levels of Grade 6 students in Victorian public schools?
In what ways have parental education levels affected the literacy levels of Grade 6 students in Victorian public schools?
The hypothesis that you wish to test should be a statement beginning with “That …”.
My hypothesis predicts that placenta from Fad104 homozygous mutant embryos will lack, or have reduced, expression of PPAR-γ. To do so, I will test my hypothesis that PPAR-γ expression in vivo is dependent upon Fad104. To do so I will perform western analysis and immunohistochemistry on placentae from wild type and Fad104 homozygous mutant embryos.
Source: Deidentified student proposal. (n.d.) Analysis of the function of FNDC3B during mammalian development. https://archive.urop.uci.edu/SURPsample Accessed: 19/1/2023
Research approach
In this section, you provide an overview of how you will gather the evidence on which the research you propose will be based. This section may be called the research methodology and it would include the research ‘theory’ you will use to guide the data collection (for example, “I am using grounded theory to frame the primary data gathering in this research”) and then the specific methods you will use to gather the data (for example, “I am using interviews”). The methods should include how you will collect data (and secure it) and how you will interpret and analyse it.
In a research proposal, this section is written in the future tense as you’re describing what you plan to do. Modal verbs like will , may and could are useful to writing up this section.
The paragraph below is from an IT research project about artificial intelligence. The researcher describes their methods in four numbered and subtitled paragraph sections. This is the first paragraph of the sequence.
- The use of future tense throughout the paragraph – “will” is italicised .
- The researcher describes how they will undertake this part of the research and the theoretical frameworks they’ll draw on. There is appropriate and accurate application of subject-specific vocabulary evident – this is bolded .
The project will be divided into the following four main components:
1) Generalization of relationships in a multi-agent system:
The focus of this part of the research is to develop algorithms for the autonomous agents to form social relationships with each other in a multi-agent community . It will explore how people generalize from previous relationships and other information when originating a new relationship. This project will involve the implementation of a novel construct that we call a Generalizing Emotional Memory (GEM) . It is derived from the Context-Specific Emotional Memory (CSEM) [15]. Specific attention will be paid to the phenomenon of negative stereotyping , a pathological phenomenon that appears to be caused by humans’ broader abilities to generalize and to learn from the experiences of others. This project will consult previous research in multi-agent systems [2, 10, 15], artificial intelligence [16], psychology [4], and animal behaviour [8], among others.
Source: Deidentified student proposal. (n.d.) Protohuman Project . https://archive.urop.uci.edu/SURP/sample_proposals/SURP%20ICS%201.pdf Accessed: 19/1/2023.
The paragraph below is from a research proposal about policing social welfare initiatives. Note the naming of the methodological framework (ethnography), identification of participants and the step-by-step outline of what will be done. The researcher has included in-text citations to justify their research and align it with previous research. This is written in future tense.
- Key terms are bolded and future tense is italicised .
This project will advance an ethnographic methodology , combining a number of in-depth qualitative approaches (Hammersley, 1995; Hammersley and Atkinson, 1995). In the first phase: (1) policy documents, (2) relevant legislation and (3) the political impetus behind the emergence of new forms of policing will be analysed. Next, a number of focus groups in Milton Keynes, and other Thames Valley Police areas will be held in order to ascertain the level of cultural acceptance and beliefs about the service-led, consumerist approach (Westmarland, 2001). An extensive series of ‘open’ interviews will then proceed with senior police officers and other key players , such as members of the Local Police Authority (Sapsford, 1996). The final phase of the research will consist of substantial periods of observational research of police work , through investigation of Thames Valley Police Restorative Justice programme.
Source: Deidentified student proposal. (n.d.) Force to Service? Consumerist Identities in Contemporary Police Governance . https://fass.open.ac.uk/sites/fass.open.ac.uk/files/files/research/sample-research-proposal.pdf Accessed: 19/1/2023.
Research ethics
In this section, you give an overview of any ethical issues inherent in the research project. Outline how informed consent will be obtained from participants, any animal participant ethics, and how you’ll mitigate any potential ethical issues. Importantly, how you’ll describe how you’ll ensure privacy and secure the data. Additionally, you might state the benefits to the participants.
In many cases, the university will have ethics compliance forms for you to complete, including explanation and consent forms for participants and, in this section, you will acknowledge your completion of these.
Anticipated problems/limitations
In this section, outline any foreseeable problems or limitations, and how you might address these or how they might impact on the research.
Significance of study
Why is your study worth doing? In this section, justify your research. You can justify the study on the basis of knowledge gaps, doing the research in a different context than those explored previously, the methodological approach, or the theoretical contributions. This is your chance to pitch your research idea and state its potential contribution.
Resources required
In this section, you list what resources you require to do the project. Do you need lab access? Do you require access to archives? Do you need to establish a budget? How will you procure the required resources?
Proposed timeline
Provide a working plan for the stages of carrying out and completing your research project. This may be a general overview or it may be date-specific. There are many different electronic work-flow programs to help you plan a timetable. The best planning trick is to start at your end date and work backwards by listing what you have to do.
You can present your proposed timeline in visual format.
Additionals
You could include references . Most research proposals and grant or scholarship applications require these. Depending on the purpose of your research proposal, you may also need to identify referees who can vouch for you and your skills to undertake the research.
Appendices can be useful if they provide support material to endorse your proposal. These can be terminologies, sample research instruments, etc.
There are three broad sections in a research proposal: introduction, literature review and methodology.
- The introduction is written in the present tense.
- The literature review is written in the past tense.
- The methodology is written in the future tense.
Cohesive ties
Cohesive ties are linguistic devices that link words, phrases and clauses into fluent sentences and paragraphs. These are important in a research proposal as they help you signpost and order your material. They are especially useful in the methodology section to ensure that your process is clearly and logically set out.
Transitional words
| Purpose | Examples | Purpose | Examples |
|---|---|---|---|
| Indicate time | Indicate result | ||
| Indicate place | Indicate purpose | ||
| Indicate frequency | Indicate coincidence | ||
| Indicate subtraction | Indicate sequence | ||
| Provide example | Indicate comparison | ||
| Indicate contrast | Indicate additional idea | ||
| Indicate opposite idea | Indicate emphasis | ||
| Indicate conclusion or summary |
Prepositional phrases
| Examples | ||
|---|---|---|
The cohesive ties are bolded in this introductory paragraph.
Taking it further
Literature reviews provide an overview of trends and gaps in research on a topic. They analyse, evaluate and synthesise the relevant literature within a particular field.
Developing research questions
Visit this Library resource for more detail on how to structure and refine your literature research questions.
Write clearly
Writing clearly is essential in presenting a convincing and successful research proposal. If you’d like to brush up on skills for writing clearly, click on this resource.
Your feedback matters
We want to hear from you! Let us know what you found most useful or share your suggestions for improving this resource.

- Customer Reviews
- Extended Essays
- IB Internal Assessment
- Theory of Knowledge
- Literature Review
- Dissertations
- Essay Writing
- Research Writing
- Assignment Help
- Capstone Projects
- College Application
- Online Class
What Are The Elements Of A Good Research Proposal?
by Antony W
March 10, 2023

The key to writing a great research proposal for your upcoming research project is to make sure the document has the right structure.
Your paper must include all the components that your professor expects to see. So in this guide, we’ll outline all the elements of a good research proposal and explain why they’re important.
The elements of a good research proposal are the title, the introduction, literature review, aims and objectives, methodology, scope of the research, outline and timetable, and bibliography.
It’s important to include these elements in your research proposal exactly in the order in which they appear in the list above.
Why The Key Elements Of A Research Proposal Matter
The basic elements of a research proposal are important because they communicate your thought process, present the originality of your ideas, and demonstrate that you’re passionate about the subject in question.
If you structure and write your research proposal well, your paper can convince your professor that your project is feasible and you have what it takes to take your research project to the next level.
Have no time to read this guide and would rather get quick writing help? Let us write your research proposal for you!
7 Key Elements of a Research Proposal
While developing a detailed and comprehensive research proposal requires a lot of planning, attention to details, and academic writing skills , understanding the core elements of the paper is the first step to getting your proposal accepted.
So here are the elements that you should include in your research proposal.
It sounds somewhat obvious when we say that your research proposal with a title. To say the least, you already know you should.
But perhaps the most common mistake that many students make is to write general titles that lack focus.
Instead of writing a long title that’s hard to read or a short title that fails to highlight the theme of your research, write a clear and concise headline that tells your reader what your research proposal is about at a first glance.
2. Introduction
The starting paragraph to a research project is one of the elements of a good research proposal because it introduces the subject you wish to address or a research problem you wish to analyze.
Because the introduction of a research proposal is what sets the tone for the rest of the paper, it’s important to start with a hook and then organize your thoughts in a logical and organized manner.
The introduction to your research proposal should give background information and explain why you believe a research question is worth exploring. While not mandatory, you can briefly describe your methodologies in the introduction and then expand them later on.
Your introduction should be clear and concise. Make sure you include only the most relevant information in this section so you don’t make it unnecessarily too long.
3. Literature Review
Although a research proposal doesn’t include a full literature review , it’s important to include an overview of the most significant studies in your field.
The section should feature evidence and statistical data to demonstrate the significance of your research.
Through the literature review, you can easily draw your reader’s attention to existing research, identify gaps in existing studies, and make your reader understand how your proposal will contribute to the already existing research.
4. Aims and Objectives
Aims and objectives are what you wish your research proposal to accomplish. Your aims will be your overall outcome or what you want the research to achieve.
Objectives tend to be narrower and more focused. More often than not, you need to provide an explanation for each of your objectives to show how they will help to meet the aims of your study.
Unless required, you don’t really have to include a hypothesis that your research proposal looks forward to test.
5. Research Methodology
Methodologies are simply the research methods you will use to conduct your study and they must appear in your research proposal whether or not you’re conducting an experimental research.
The methodologies include analysis and sampling techniques equipment, research approaches, and ethical concerns.
Make sure your explanation for each methodology is clear and precise. It helps to justify why you’ve chosen to use a certain methodology over an alternative. This will go a long way to show that you took your time to think about your methodologies before picking them.
It’s important to explain how you will collect data, the sample size you plan to consider for your research investigation, and the techniques you consider the most appropriate to analyze the data.
6. Scope of the Research
Because you’ll be working with limited time and resource, it’s reasonable to include a section on the scope of the research in your proposal. In other words, you have to show your reader that you can start and complete your research within the constraints of these two resources.
Remember, your research will more than likely have limits, and addressing them in this section not only shows that you have given them a thought but also makes your research proposal strong and authentic.
Don’t just focus on the challenges that you’re likely to come across during your studies. You should also propose alternative solutions that you can use and why they might help.
7. Outline and Timetable
Your professor expects to see an outline and a timetable in your research proposal so it’s important that you include them in your research proposal.
The purpose of the outline is to show how you plan to structure your dissertation . Briefly note what each section will cover and explain how it all fits into the argument of your research project.
The purpose of the timetable is to show how much time you’ll need to complete your research. In particular, you need to make sure you mention exactly how long you expect each stage of your study to take.
Don’t just mention how long the research process will take. Make sure you also indicate how long you’ll take to compile your research.
Get Help with Research Proposal Writing
Knowing the elements of a good research proposal is one thing. Writing the proposal is where there’s a lot of work. If you don’t have the time to complete the work yourself, feel free to take advantage of our research proposal writing and get the paper done on time.
About the author
Antony W is a professional writer and coach at Help for Assessment. He spends countless hours every day researching and writing great content filled with expert advice on how to write engaging essays, research papers, and assignments.

Want to create or adapt books like this? Learn more about how Pressbooks supports open publishing practices.
Chapter 14: The Research Proposal
14.3 Components of a Research Proposal
Krathwohl (2005) suggests and describes a variety of components to include in a research proposal. The following sections – Introductions, Background and significance, Literature Review; Research design and methods, Preliminary suppositions and implications; and Conclusion present these components in a suggested template for you to follow in the preparation of your research proposal.
Introduction
The introduction sets the tone for what follows in your research proposal – treat it as the initial pitch of your idea. After reading the introduction your reader should:
- understand what it is you want to do;
- have a sense of your passion for the topic; and
- be excited about the study’s possible outcomes.
As you begin writing your research proposal, it is helpful to think of the introduction as a narrative of what it is you want to do, written in one to three paragraphs. Within those one to three paragraphs, it is important to briefly answer the following questions:
- What is the central research problem?
- How is the topic of your research proposal related to the problem?
- What methods will you utilize to analyze the research problem?
- Why is it important to undertake this research? What is the significance of your proposed research? Why are the outcomes of your proposed research important? Whom are they important?
Note : You may be asked by your instructor to include an abstract with your research proposal. In such cases, an abstract should provide an overview of what it is you plan to study, your main research question, a brief explanation of your methods to answer the research question, and your expected findings. All of this information must be carefully crafted in 150 to 250 words. A word of advice is to save the writing of your abstract until the very end of your research proposal preparation. If you are asked to provide an abstract, you should include 5 to 7 key words that are of most relevance to your study. List these in order of relevance.
Background and Significance
The purpose of this section is to explain the context of your proposal and to describe, in detail, why it is important to undertake this research. Assume that the person or people who will read your research proposal know nothing or very little about the research problem. While you do not need to include all knowledge you have learned about your topic in this section, it is important to ensure that you include the most relevant material that will help to explain the goals of your research.
While there are no hard and fast rules, you should attempt to address some or all of the following key points:
- State the research problem and provide a more thorough explanation about the purpose of the study than what you stated in the introduction.
- Present the rationale for the proposed research study. Clearly indicate why this research is worth doing. Answer the “so what?” question.
- Describe the major issues or problems to be addressed by your research. Do not forget to explain how and in what ways your proposed research builds upon previous related research.
- Explain how you plan to go about conducting your research.
- Clearly identify the key or most relevant sources of research you intend to use and explain how they will contribute to your analysis of the topic.
- Set the boundaries of your proposed research, in order to provide a clear focus. Where appropriate, state not only what you will study, but what will be excluded from your study.
- Provide clear definitions of key concepts and terms. Since key concepts and terms often have numerous definitions, make sure you state which definition you will be utilizing in your research.
Literature Review
This key component of the research proposal is the most time-consuming aspect in the preparation of your research proposal. As described in Chapter 5 , the literature review provides the background to your study and demonstrates the significance of the proposed research. Specifically, it is a review and synthesis of prior research that is related to the problem you are setting forth to investigate. Essentially, your goal in the literature review is to place your research study within the larger whole of what has been studied in the past, while demonstrating to your reader that your work is original, innovative, and adds to the larger whole.
As the literature review is information dense, it is essential that this section be intelligently structured to enable your reader to grasp the key arguments underpinning your study. However, this can be easier to state and harder to do, simply due to the fact there is usually a plethora of related research to sift through. Consequently, a good strategy for writing the literature review is to break the literature into conceptual categories or themes, rather than attempting to describe various groups of literature you reviewed. Chapter 5 describes a variety of methods to help you organize the themes.
Here are some suggestions on how to approach the writing of your literature review:
- Think about what questions other researchers have asked, what methods they used, what they found, and what they recommended based upon their findings.
- Do not be afraid to challenge previous related research findings and/or conclusions.
- Assess what you believe to be missing from previous research and explain how your research fills in this gap and/or extends previous research.
It is important to note that a significant challenge related to undertaking a literature review is knowing when to stop. As such, it is important to know when you have uncovered the key conceptual categories underlying your research topic. Generally, when you start to see repetition in the conclusions or recommendations, you can have confidence that you have covered all of the significant conceptual categories in your literature review. However, it is also important to acknowledge that researchers often find themselves returning to the literature as they collect and analyze their data. For example, an unexpected finding may develop as you collect and/or analyze the data; in this case, it is important to take the time to step back and review the literature again, to ensure that no other researchers have found a similar finding. This may include looking to research outside your field.
This situation occurred with one of this textbook’s authors’ research related to community resilience. During the interviews, the researchers heard many participants discuss individual resilience factors and how they believed these individual factors helped make the community more resilient, overall. Sheppard and Williams (2016) had not discovered these individual factors in their original literature review on community and environmental resilience. However, when they returned to the literature to search for individual resilience factors, they discovered a small body of literature in the child and youth psychology field. Consequently, Sheppard and Williams had to go back and add a new section to their literature review on individual resilience factors. Interestingly, their research appeared to be the first research to link individual resilience factors with community resilience factors.
Research design and methods
The objective of this section of the research proposal is to convince the reader that your overall research design and methods of analysis will enable you to solve the research problem you have identified and also enable you to accurately and effectively interpret the results of your research. Consequently, it is critical that the research design and methods section is well-written, clear, and logically organized. This demonstrates to your reader that you know what you are going to do and how you are going to do it. Overall, you want to leave your reader feeling confident that you have what it takes to get this research study completed in a timely fashion.
Essentially, this section of the research proposal should be clearly tied to the specific objectives of your study; however, it is also important to draw upon and include examples from the literature review that relate to your design and intended methods. In other words, you must clearly demonstrate how your study utilizes and builds upon past studies, as it relates to the research design and intended methods. For example, what methods have been used by other researchers in similar studies?
While it is important to consider the methods that other researchers have employed, it is equally, if not more, important to consider what methods have not been but could be employed. Remember, the methods section is not simply a list of tasks to be undertaken. It is also an argument as to why and how the tasks you have outlined will help you investigate the research problem and answer your research question(s).
Tips for writing the research design and methods section
Specify the methodological approaches you intend to employ to obtain information and the techniques you will use to analyze the data.
Specify the research operations you will undertake and the way you will interpret the results of those operations in relation to the research problem.
Go beyond stating what you hope to achieve through the methods you have chosen. State how you will actually implement the methods (i.e., coding interview text, running regression analysis, etc.).
Anticipate and acknowledge any potential barriers you may encounter when undertaking your research, and describe how you will address these barriers.
Explain where you believe you will find challenges related to data collection, including access to participants and information.
Preliminary Suppositions and Implications
The purpose of this section is to argue how you anticipate that your research will refine, revise, or extend existing knowledge in the area of your study. Depending upon the aims and objectives of your study, you should also discuss how your anticipated findings may impact future research. For example, is it possible that your research may lead to a new policy, theoretical understanding, or method for analyzing data? How might your study influence future studies? What might your study mean for future practitioners working in the field? Who or what might benefit from your study? How might your study contribute to social, economic or environmental issues? While it is important to think about and discuss possibilities such as these, it is equally important to be realistic in stating your anticipated findings. In other words, you do not want to delve into idle speculation. Rather, the purpose here is to reflect upon gaps in the current body of literature and to describe how you anticipate your research will begin to fill in some or all of those gaps.
The conclusion reiterates the importance and significance of your research proposal, and provides a brief summary of the entire proposed study. Essentially, this section should only be one or two paragraphs in length. Here is a potential outline for your conclusion:
Discuss why the study should be done. Specifically discuss how you expect your study will advance existing knowledge and how your study is unique.
Explain the specific purpose of the study and the research questions that the study will answer.
Explain why the research design and methods chosen for this study are appropriate, and why other designs and methods were not chosen.
State the potential implications you expect to emerge from your proposed study,
Provide a sense of how your study fits within the broader scholarship currently in existence, related to the research problem.
Citations and References
As with any scholarly research paper, you must cite the sources you used in composing your research proposal. In a research proposal, this can take two forms: a reference list or a bibliography. A reference list lists the literature you referenced in the body of your research proposal. All references in the reference list must appear in the body of the research proposal. Remember, it is not acceptable to say “as cited in …” As a researcher you must always go to the original source and check it for yourself. Many errors are made in referencing, even by top researchers, and so it is important not to perpetuate an error made by someone else. While this can be time consuming, it is the proper way to undertake a literature review.
In contrast, a bibliography , is a list of everything you used or cited in your research proposal, with additional citations to any key sources relevant to understanding the research problem. In other words, sources cited in your bibliography may not necessarily appear in the body of your research proposal. Make sure you check with your instructor to see which of the two you are expected to produce.
Overall, your list of citations should be a testament to the fact that you have done a sufficient level of preliminary research to ensure that your project will complement, but not duplicate, previous research efforts. For social sciences, the reference list or bibliography should be prepared in American Psychological Association (APA) referencing format. Usually, the reference list (or bibliography) is not included in the word count of the research proposal. Again, make sure you check with your instructor to confirm.
Research Methods for the Social Sciences: An Introduction Copyright © 2020 by Valerie Sheppard is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License , except where otherwise noted.
Share This Book
- Research Process
- Manuscript Preparation
- Manuscript Review
- Publication Process
- Publication Recognition
Language Editing Services
- Translation Services

Writing a Scientific Research Project Proposal
- 5 minute read
- 116.6K views
Table of Contents
The importance of a well-written research proposal cannot be underestimated. Your research really is only as good as your proposal. A poorly written, or poorly conceived research proposal will doom even an otherwise worthy project. On the other hand, a well-written, high-quality proposal will increase your chances for success.
In this article, we’ll outline the basics of writing an effective scientific research proposal, including the differences between research proposals, grants and cover letters. We’ll also touch on common mistakes made when submitting research proposals, as well as a simple example or template that you can follow.
What is a scientific research proposal?
The main purpose of a scientific research proposal is to convince your audience that your project is worthwhile, and that you have the expertise and wherewithal to complete it. The elements of an effective research proposal mirror those of the research process itself, which we’ll outline below. Essentially, the research proposal should include enough information for the reader to determine if your proposed study is worth pursuing.
It is not an uncommon misunderstanding to think that a research proposal and a cover letter are the same things. However, they are different. The main difference between a research proposal vs cover letter content is distinct. Whereas the research proposal summarizes the proposal for future research, the cover letter connects you to the research, and how you are the right person to complete the proposed research.
There is also sometimes confusion around a research proposal vs grant application. Whereas a research proposal is a statement of intent, related to answering a research question, a grant application is a specific request for funding to complete the research proposed. Of course, there are elements of overlap between the two documents; it’s the purpose of the document that defines one or the other.
Scientific Research Proposal Format
Although there is no one way to write a scientific research proposal, there are specific guidelines. A lot depends on which journal you’re submitting your research proposal to, so you may need to follow their scientific research proposal template.
In general, however, there are fairly universal sections to every scientific research proposal. These include:
- Title: Make sure the title of your proposal is descriptive and concise. Make it catch and informative at the same time, avoiding dry phrases like, “An investigation…” Your title should pique the interest of the reader.
- Abstract: This is a brief (300-500 words) summary that includes the research question, your rationale for the study, and any applicable hypothesis. You should also include a brief description of your methodology, including procedures, samples, instruments, etc.
- Introduction: The opening paragraph of your research proposal is, perhaps, the most important. Here you want to introduce the research problem in a creative way, and demonstrate your understanding of the need for the research. You want the reader to think that your proposed research is current, important and relevant.
- Background: Include a brief history of the topic and link it to a contemporary context to show its relevance for today. Identify key researchers and institutions also looking at the problem
- Literature Review: This is the section that may take the longest amount of time to assemble. Here you want to synthesize prior research, and place your proposed research into the larger picture of what’s been studied in the past. You want to show your reader that your work is original, and adds to the current knowledge.
- Research Design and Methodology: This section should be very clearly and logically written and organized. You are letting your reader know that you know what you are going to do, and how. The reader should feel confident that you have the skills and knowledge needed to get the project done.
- Preliminary Implications: Here you’ll be outlining how you anticipate your research will extend current knowledge in your field. You might also want to discuss how your findings will impact future research needs.
- Conclusion: This section reinforces the significance and importance of your proposed research, and summarizes the entire proposal.
- References/Citations: Of course, you need to include a full and accurate list of any and all sources you used to write your research proposal.
Common Mistakes in Writing a Scientific Research Project Proposal
Remember, the best research proposal can be rejected if it’s not well written or is ill-conceived. The most common mistakes made include:
- Not providing the proper context for your research question or the problem
- Failing to reference landmark/key studies
- Losing focus of the research question or problem
- Not accurately presenting contributions by other researchers and institutions
- Incompletely developing a persuasive argument for the research that is being proposed
- Misplaced attention on minor points and/or not enough detail on major issues
- Sloppy, low-quality writing without effective logic and flow
- Incorrect or lapses in references and citations, and/or references not in proper format
- The proposal is too long – or too short
Scientific Research Proposal Example
There are countless examples that you can find for successful research proposals. In addition, you can also find examples of unsuccessful research proposals. Search for successful research proposals in your field, and even for your target journal, to get a good idea on what specifically your audience may be looking for.
While there’s no one example that will show you everything you need to know, looking at a few will give you a good idea of what you need to include in your own research proposal. Talk, also, to colleagues in your field, especially if you are a student or a new researcher. We can often learn from the mistakes of others. The more prepared and knowledgeable you are prior to writing your research proposal, the more likely you are to succeed.
One of the top reasons scientific research proposals are rejected is due to poor logic and flow. Check out our Language Editing Services to ensure a great proposal , that’s clear and concise, and properly referenced. Check our video for more information, and get started today.

Research Fraud: Falsification and Fabrication in Research Data

Research Team Structure
You may also like.

Descriptive Research Design and Its Myriad Uses

Five Common Mistakes to Avoid When Writing a Biomedical Research Paper

Making Technical Writing in Environmental Engineering Accessible

To Err is Not Human: The Dangers of AI-assisted Academic Writing

When Data Speak, Listen: Importance of Data Collection and Analysis Methods

Choosing the Right Research Methodology: A Guide for Researchers

Why is data validation important in research?

Writing a good review article
Input your search keywords and press Enter.
- Locations and Hours
- UCLA Library
- Research Guides
- Research Tips and Tools
Advanced Research Methods
- Writing a Research Proposal
- What Is Research?
- Library Research
What Is a Research Proposal?
Reference books.
- Writing the Research Paper
- Presenting the Research Paper
When applying for a research grant or scholarship, or, just before you start a major research project, you may be asked to write a preliminary document that includes basic information about your future research. This is the information that is usually needed in your proposal:
- The topic and goal of the research project.
- The kind of result expected from the research.
- The theory or framework in which the research will be done and presented.
- What kind of methods will be used (statistical, empirical, etc.).
- Short reference on the preliminary scholarship and why your research project is needed; how will it continue/justify/disprove the previous scholarship.
- How much will the research project cost; how will it be budgeted (what for the money will be spent).
- Why is it you who can do this research and not somebody else.
Most agencies that offer scholarships or grants provide information about the required format of the proposal. It may include filling out templates, types of information they need, suggested/maximum length of the proposal, etc.
Research proposal formats vary depending on the size of the planned research, the number of participants, the discipline, the characteristics of the research, etc. The following outline assumes an individual researcher. This is just a SAMPLE; several other ways are equally good and can be successful. If possible, discuss your research proposal with an expert in writing, a professor, your colleague, another student who already wrote successful proposals, etc.
- Author, author's affiliation
- Explain the topic and why you chose it. If possible explain your goal/outcome of the research . How much time you need to complete the research?
- Give a brief summary of previous scholarship and explain why your topic and goals are important.
- Relate your planned research to previous scholarship. What will your research add to our knowledge of the topic.
- Break down the main topic into smaller research questions. List them one by one and explain why these questions need to be investigated. Relate them to previous scholarship.
- Include your hypothesis into the descriptions of the detailed research issues if you have one. Explain why it is important to justify your hypothesis.
- This part depends of the methods conducted in the research process. List the methods; explain how the results will be presented; how they will be assessed.
- Explain what kind of results will justify or disprove your hypothesis.
- Explain how much money you need.
- Explain the details of the budget (how much you want to spend for what).
- Describe why your research is important.
- List the sources you have used for writing the research proposal, including a few main citations of the preliminary scholarship.
- << Previous: Library Research
- Next: Writing the Research Paper >>
- Last Updated: Aug 22, 2024 3:43 PM
- URL: https://guides.library.ucla.edu/research-methods

Writing a Research Proposal
Parts of a research proposal, prosana model, introduction, research question, methodology.
- Structure of a Research Proposal
- Common Proposal Writing Mistakes
- Proposal Writing Resources
A research proposal's purpose is to capture the evaluator's attention, demonstrate the study's potential benefits, and prove that it is a logical and consistent approach (Van Ekelenburg, 2010). To ensure that your research proposal contains these elements, there are several aspects to include in your proposal (Al-Riyami, 2008):
- Objective(s)
- Variables (independent and dependent)
- Research Question and/or hypothesis
Details about what to include in each element are included in the boxes below. Depending on the topic of your study, some parts may not apply to your proposal. You can also watch the video below for a brief overview about writing a successful research proposal.
Van Ekelenburg (2010) uses the PROSANA Model to guide researchers in developing rationale and justification for their research projects. It is an acronym that connects the problem, solution, and benefits of a particular research project. It is an easy way to remember the critical parts of a research proposal and how they relate to one another. It includes the following letters (Van Ekelenburg, 2010):
- Problem: Describing the main problem that the researcher is trying to solve.
- Root causes: Describing what is causing the problem. Why is the topic an issue?
- fOcus: Narrowing down one of the underlying causes on which the researcher will focus for their research project.
- Solutions: Listing potential solutions or approaches to fix to the problem. There could be more than one.
- Approach: Selecting the solution that the researcher will want to focus on.
- Novelty: Describing how the solution will address or solve the problem.
- Arguments: Explaining how the proposed solution will benefit the problem.
Research proposal titles should be concise and to the point, but informative. The title of your proposal may be different from the title of your final research project, but that is completely normal! Your findings may help you come up with a title that is more fitting for the final project. Characteristics of good proposal titles are (Al-Riyami, 2008):
- Catchy: It catches the reader's attention by peaking their interest.
- Positive: It spins your project in a positive way towards the reader.
- Transparent: It identifies the independent and dependent variables.
It is also common for proposal titles to be very similar to your research question, hypothesis, or thesis statement (Locke et al., 2007).
An abstract is a brief summary (about 300 words) of the study you are proposing. It includes the following elements (Al-Riyami, 2008):
- Your primary research question(s).
- Hypothesis or main argument.
- Method you will use to complete the study. This may include the design, sample population, or measuring instruments that you plan to use.
Our guide on writing summaries may help you with this step.
The purpose of the introduction is to give readers background information about your topic. it gives the readers a basic understanding of your topic so that they can further understand the significance of your proposal. A good introduction will explain (Al-Riyami, 2008):
- How it relates to other research done on the topic
- Why your research is significant to the field
- The relevance of your study
Your research objectives are the desired outcomes that you will achieve from the research project. Depending on your research design, these may be generic or very specific. You may also have more than one objective (Al-Riyami, 2008).
- General objectives are what the research project will accomplish
- Specific objectives relate to the research questions that the researcher aims to answer through the study.
Be careful not to have too many objectives in your proposal, as having too many can make your project lose focus. Plus, it may not be possible to achieve several objectives in one study.
This section describes the different types of variables that you plan to have in your study and how you will measure them. According to Al-Riyami (2008), there are four types of research variables:
- Independent: The person, object, or idea that is manipulated by the researcher.
- Dependent: The person, object, or idea whose changes are dependent upon the independent variable. Typically, it is the item that the researcher is measuring for the study.
- Confounding/Intervening: Factors that may influence the effect of the independent variable on the dependent variable. These include physical and mental barriers. Not every study will have intervening variables, but they should be studied if applicable.
- Background: Factors that are relevant to the study's data and how it can be generalized. Examples include demographic information such as age, sex, and ethnicity.
Your research proposal should describe each of your variables and how they relate to one another. Depending on your study, you may not have all four types of variables present. However, there will always be an independent and dependent variable.
A research question is the main piece of your research project because it explains what your study will discover to the reader. It is the question that fuels the study, so it is important for it to be precise and unique. You do not want it to be too broad, and it should identify a relationship between two variables (an independent and a dependent) (Al-Riyami, 2008). There are six types of research questions (Academic Writer, n.d.):
- Example: "Do people get nervous before speaking in front of an audience?"
- Example: "What are the study habits of college freshmen at Tiffin University?"
- Example: "What primary traits create a successful romantic relationship?"
- Example: "Is there a relationship between a child's performance in school and their parents' socioeconomic status?"
- Example: "Are high school seniors more motivated than high school freshmen?"
- Example: "Do news media outlets impact a person's political opinions?"
For more information on the different types of research questions, you can view the "Research Questions and Hypotheses" tutorial on Academic Writer, located below. If you are unfamiliar with Academic Writer, we also have a tutorial on using the database located below.
Compose papers in pre-formatted APA templates. Manage references in forms that help craft APA citations. Learn the rules of APA style through tutorials and practice quizzes.
Academic Writer will continue to use the 6th edition guidelines until August 2020. A preview of the 7th edition is available in the footer of the resource's site. Previously known as APA Style Central.
- Academic Writer Tutorial by Pfeiffer Library Last Updated May 22, 2023 43526 views this year
If you know enough about your research topic that you believe a particular outcome may occur as a result of the study, you can include a hypothesis (thesis statement) in your proposal. A hypothesis is a prediction that you believe will be the outcome of your study. It explains what you think the relationship will be between the independent and dependent variable (Al-Riyami, 2008). It is ok if the hypothesis in your proposal turns out to be incorrect, because it is only a prediction! If you are writing a proposal in the humanities, you may be writing a thesis statement instead of a hypothesis. A thesis presents the main argument of your research project and leads to corresponding evidence to support your argument.
Hypotheses vs. Theories
Hypotheses are different from theories in that theories represent general principles and sets of rules that explain different phenomena. They typically represent large areas of study because they are applicable to anything in a particular field. Hypotheses focus on specific areas within a field and are educated guesses, meaning that they have the potential to be proven wrong (Academic Writer, n.d.). Because of this, hypotheses can also be formed from theories.
For more information on writing effective thesis statements, you can view our guide on writing thesis statements below.
In a research proposal, you must thoroughly explain how you will conduct your study. This includes things such as (Al-Riyami, 2008):
- Research design: What research approach will your study take? Will it be quantitative or qualitative?
- Research subjects/participants: Who will be participating in your study? Does your study require human participants? How will you determine who to study?
- Sample size: How many participants will your study require? If you are not using human participants, how much of the sample will you be studying?
- Timeline: A proposed list of the general tasks and events that you plan to complete the study. This will include a time frame for each task/event and the order in which they will be completed.
- Interventions: If you plan on using anything on human participants for the study, you must include information it here. This is especially important if you plan on using any substances on human subjects.
- Ethical issues: Are there any potential ethical issues surrounding this study?
- Potential limitations: Are there any limitations that could skew the data and findings from your study?
- Appendixes: If you need to present any consent forms, interview questions, surveys, questionnaires, or other items that will be used in your study, you should include samples of each item with an appendix to reference them. If you are using a copyrighted document, you may need written permission from the original creator to use it in your study. A copy of the written permission should be included in your proposal.
- Setting: Where will you be conducting the study?
- Study instruments: What measuring tools or computer software will you be using to collect data? How will you collect the data?
- How you will analyze the data: What strategies or tools will you use to analyze the data you collect?
- Quality control: Will you have precautions in place to ensure that the study is conducted consistently and that outside factors will not skew the data?
- Budget: What type of funding will you need for your study? This will include the funds needed to afford measuring tools, software, etc.
- How you will share the study's findings: What will you plan to do with the findings?
- Significance of the study: How will your study expand on existing knowledge of the subject area?
For more information on research methodologies, you can view our guide on research methods and methodologies below.
- Research Methodologies by Pfeiffer Library Last Updated Aug 2, 2022 51115 views this year
- << Previous: Welcome
- Next: Structure of a Research Proposal >>
- Last Updated: May 22, 2023 10:46 AM
- URL: https://library.tiffin.edu/writingaresearchproposal
How to write a Strong Research Proposal

- Kenyatta University
Discover the world's research
- 25+ million members
- 160+ million publication pages
- 2.3+ billion citations
- Farisani Thomas Nephawe
- Sri Sahbany
- Samsul Hadi

- John W Creswell
- Kay Muir-Leresche
- Richard Coe
- Susan D Hainsworth
- S J Silverman
- Recruit researchers
- Join for free
- Login Email Tip: Most researchers use their institutional email address as their ResearchGate login Password Forgot password? Keep me logged in Log in or Continue with Google Welcome back! Please log in. Email · Hint Tip: Most researchers use their institutional email address as their ResearchGate login Password Forgot password? Keep me logged in Log in or Continue with Google No account? Sign up

How to write a Research Proposal: Components of a research proposal
Components of a research proposal.
- Useful videos
- Common mistakes to avoid
- Sage Research Methods LibGuide This link opens in a new window
- Managing sources
- Request a literature search
- Research proposal - examples
- Creating a Gantt chart
- Free Apps for Research
- Academic writing
Research proposals differ in terms of their presentation depending on what each University department requires. In other words, there is no set template for a research proposal. Please contact your lecturer regarding the format you are expected to use for your research proposal.Thus, the components of a research proposal include, but are not limited to those mentioned in this guide.
1. The title
Try to come up with a title that is unique and at the same time easy to remember. It should also make a lasting impression to the reader and make them want to come back and read your proposal. The title must also capture the main concepts of the study . As the research process is lengthy, it is important that you choose a topic that you are so curious about that you remain motivated for the duration of the research process. Select a topic that you will be able to complete within the time frame that you have for your research.
3. The background
The background to the topic of your intended research must be clear and precise. It must not only include an in-depth explanation of the key points of your subject but also all the developments in the field as well as their timelines . The researcher must also explain the compelling interest in the research issue as well as the personal interest (if any) in the topic. This section must also indicate the specific area within which the topic falls in your particular field of study or subject . Aslo, how will the proposed study contribute to a particular field? In other words, the impact and the significance in a subject area must be clearly outlined. The target audience must also be clearly described.
5. Objectives of the research
It is important that the objectives are in alignment with the research questions. The objectives must indicate what the aim of the research study is. In fact, objectives give you a clear indication of the steps that you will take to achieve the aim of the research. The objectives must be specific, measurable, achievable, relevant and time-bound.
7. Literature review
Collect and present relevant literature on your topic of choice. It is important to include all the main authors or experts in a particular field. Depending on your field of study or topic, ensure that you include recent literature as well as literature that presents counterarguments to the topic. The justification for the study needs to based on existing literature. Click here for more information on how to write a literature review.
8. Limitations and delimitations of the study
The researcher must indicate the limitations of the study which are what the researcher cannot do or factors that are beyond the researcher's control, as well as delimitations that the researcher chooses not to address for the purposes of the study. Delimitations are boundaries that the researcher has set for the study. The r easons both for limitations and delimitations must be discussed in this section.
10. Work plan
Your schedule for the research must be stated clearly including the projected timelines for the various stages of your study.
11. Bibliography
All the sources that you have used for your proposal must be listed in alphabetical order using a referencing style that your lecturer has prescribed for your subject field.
Click here for more information on the various reference styles.
2. Introduction to the research
This section of the proposal must provide a broad overview of the topic. The jargon and key terms used in the particular topic must also be thoroughly explained in order to avoid confusion. The interest of the researcher in the particular topic must also be clearly outlined while at the same time mentioning, albeit briefly at this point, a critical review of the main literature that covers the topic. The researcher must also provide the aim of the research by clearly and concisely stating the problem, as well as the research questions to be dealt with. This section must also indicate what the research study will not be covering .
4. The research questions
The research questions must state clearly what your proposed study is meant to address or answer. Ensure that you use simple language that is easy to understand, while being cognisant of the level of your intended audience .
6. Research methodology / research methods
This section outlines the approach which the researcher will follow in order to address the research problem and to answer all the research questions from the researcher. The research design must be clearly defined, e.g., is the research Descriptive, Correlational, Causal-Comparative/Quasi-Experimental, Experimental, Diagnostic or Explanatory.
State clearly
- how the research will be conducted in terms of the theoretical resources that will be used
- the theoretical framework for conducting the research, which is the theoretical approach drawn from your literature review to support your research study
- proposed research method(s)
- a comparison of the advantages, limitations and suitability of the available approaches and methods for conducting your research
- participants, instruments, procedure, analysis, etc.
Research design
Selecting the approach to use
Research approach
Research design and methodology
Importance of research
Attributes of a good research scholar
Summary of different research methodologies

9. Significance of the research
The researcher must provide justification for the need to conduct the study. What is the gap that the study will fill, and what is its contribution to the existing body of knowledge? The originality and importance of the research which will be level appropriate, must be clearly described, for instance, the required level of originality for a fourth year research project is different to that of a doctoral candidate.
The impact of the study for the subject field must be indicated. In other words, how will the research improve the field, who will it impact, how will it make changes in your industy or field etc.? Lastly, the proposed resaerch must be relatable , interesting and engaging .
- << Previous: Home
- Next: Books >>
- Last Updated: Sep 25, 2024 3:09 PM
- URL: https://libguides.unisa.ac.za/research_proposal
Research Administration
- SURF Application Instructions
- Export Controls and Research Compliance
- Uniform Guidance
- Frequently Asked Questions
- Frequently Required Information
- Animal Subjects and IRB
- Streamlyne Information
Pre-Award Resources
- Funding Agencies
- Funding Opportunities With Institutional Limits
- Funding Resources
Post-Award Resources
- Award Setup
- Award Management
- Award Closeout
- Effort Certification
Proposal Characteristics
1. The need for the proposed activity is clearly established, preferably with data.
2. The most important ideas are highlighted and repeated in several places.
3. Appropriate detail is provided in all portions of the proposal such as in the objectives and a detailed schedule of activities for the project or project schedule.
4. Collaboration with all interested groups in planning of the proposed project is evident in the proposal. The commitment of all involved parties, including project staff and consultants, is evident, from letters of commitment in the appendix and cost sharing stated in both the proposal narrative’s and budget.
5. The budget and the proposal narrative are consistent. The uses of money are indicated clearly in the proposal narrative as well as in the budget.
6. All of the major matters indicated in the proposal guidelines and directions are followed carefully and addressed clearly in the proposal. The length is also consistent with the funding agency’s proposal guidelines and expectations.
7. The agreement of all project staff and consultants to participate in the project was acquired and indicated in the proposal.
8. All governmental procedures are followed with regard to matters such as civil rights compliance and protection of human subjects.
9. Appendices are used appropriately for detailed and lengthy materials that reviewers may not want to read but are useful as evidence of careful planning, previous experience, etc.
10. The budget explanations provide an adequate basis for the figures used in building the budget.
11. If appropriate, a clear statement of commitment to continue the project after external funding is including.
12. The proposal speaks to the readers, helping them understand the problems and the project. Summarized statements and headings are used to lead the reader. The writing style is clear and concise.
- Office of Research and Sponsored Programs
- Ottenheimer Library 512 2801 S. University Avenue Little Rock , AR 72204
- Phone: 501-916-3474 Email: [email protected]
- More contact information
Interact with Us
Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, generate accurate citations for free.
- Knowledge Base
- Starting the research process
- Writing Strong Research Questions | Criteria & Examples
Writing Strong Research Questions | Criteria & Examples
Published on October 26, 2022 by Shona McCombes . Revised on November 21, 2023.
A research question pinpoints exactly what you want to find out in your work. A good research question is essential to guide your research paper , dissertation , or thesis .
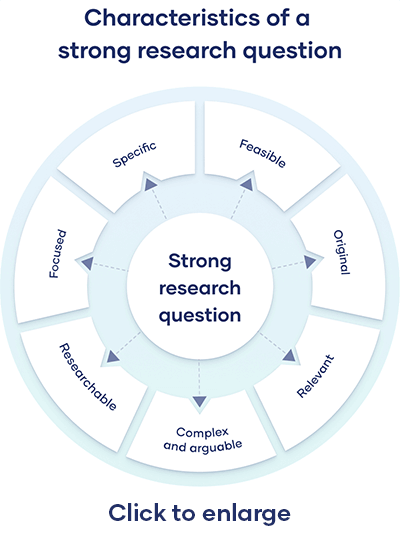
All research questions should be:
- Focused on a single problem or issue
- Researchable using primary and/or secondary sources
- Feasible to answer within the timeframe and practical constraints
- Specific enough to answer thoroughly
- Complex enough to develop the answer over the space of a paper or thesis
- Relevant to your field of study and/or society more broadly
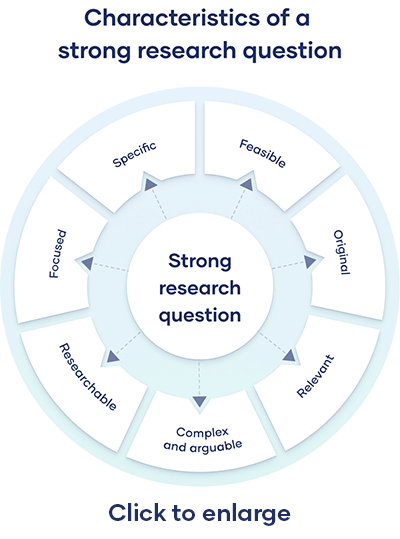
Table of contents
How to write a research question, what makes a strong research question, using sub-questions to strengthen your main research question, research questions quiz, other interesting articles, frequently asked questions about research questions.
You can follow these steps to develop a strong research question:
- Choose your topic
- Do some preliminary reading about the current state of the field
- Narrow your focus to a specific niche
- Identify the research problem that you will address
The way you frame your question depends on what your research aims to achieve. The table below shows some examples of how you might formulate questions for different purposes.
| Research question formulations | |
|---|---|
| Describing and exploring | |
| Explaining and testing | |
| Evaluating and acting | is X |
Using your research problem to develop your research question
| Example research problem | Example research question(s) |
|---|---|
| Teachers at the school do not have the skills to recognize or properly guide gifted children in the classroom. | What practical techniques can teachers use to better identify and guide gifted children? |
| Young people increasingly engage in the “gig economy,” rather than traditional full-time employment. However, it is unclear why they choose to do so. | What are the main factors influencing young people’s decisions to engage in the gig economy? |
Note that while most research questions can be answered with various types of research , the way you frame your question should help determine your choices.
Here's why students love Scribbr's proofreading services
Discover proofreading & editing
Research questions anchor your whole project, so it’s important to spend some time refining them. The criteria below can help you evaluate the strength of your research question.
Focused and researchable
| Criteria | Explanation |
|---|---|
| Focused on a single topic | Your central research question should work together with your research problem to keep your work focused. If you have multiple questions, they should all clearly tie back to your central aim. |
| Answerable using | Your question must be answerable using and/or , or by reading scholarly sources on the to develop your argument. If such data is impossible to access, you likely need to rethink your question. |
| Not based on value judgements | Avoid subjective words like , , and . These do not give clear criteria for answering the question. |
Feasible and specific
| Criteria | Explanation |
|---|---|
| Answerable within practical constraints | Make sure you have enough time and resources to do all research required to answer your question. If it seems you will not be able to gain access to the data you need, consider narrowing down your question to be more specific. |
| Uses specific, well-defined concepts | All the terms you use in the research question should have clear meanings. Avoid vague language, jargon, and too-broad ideas. |
| Does not demand a conclusive solution, policy, or course of action | Research is about informing, not instructing. Even if your project is focused on a practical problem, it should aim to improve understanding rather than demand a ready-made solution. If ready-made solutions are necessary, consider conducting instead. Action research is a research method that aims to simultaneously investigate an issue as it is solved. In other words, as its name suggests, action research conducts research and takes action at the same time. |
Complex and arguable
| Criteria | Explanation |
|---|---|
| Cannot be answered with or | Closed-ended, / questions are too simple to work as good research questions—they don’t provide enough for robust investigation and discussion. |
| Cannot be answered with easily-found facts | If you can answer the question through a single Google search, book, or article, it is probably not complex enough. A good research question requires original data, synthesis of multiple sources, and original interpretation and argumentation prior to providing an answer. |
Relevant and original
| Criteria | Explanation |
|---|---|
| Addresses a relevant problem | Your research question should be developed based on initial reading around your . It should focus on addressing a problem or gap in the existing knowledge in your field or discipline. |
| Contributes to a timely social or academic debate | The question should aim to contribute to an existing and current debate in your field or in society at large. It should produce knowledge that future researchers or practitioners can later build on. |
| Has not already been answered | You don’t have to ask something that nobody has ever thought of before, but your question should have some aspect of originality. For example, you can focus on a specific location, or explore a new angle. |
Chances are that your main research question likely can’t be answered all at once. That’s why sub-questions are important: they allow you to answer your main question in a step-by-step manner.
Good sub-questions should be:
- Less complex than the main question
- Focused only on 1 type of research
- Presented in a logical order
Here are a few examples of descriptive and framing questions:
- Descriptive: According to current government arguments, how should a European bank tax be implemented?
- Descriptive: Which countries have a bank tax/levy on financial transactions?
- Framing: How should a bank tax/levy on financial transactions look at a European level?
Keep in mind that sub-questions are by no means mandatory. They should only be asked if you need the findings to answer your main question. If your main question is simple enough to stand on its own, it’s okay to skip the sub-question part. As a rule of thumb, the more complex your subject, the more sub-questions you’ll need.
Try to limit yourself to 4 or 5 sub-questions, maximum. If you feel you need more than this, it may be indication that your main research question is not sufficiently specific. In this case, it’s is better to revisit your problem statement and try to tighten your main question up.
Receive feedback on language, structure, and formatting
Professional editors proofread and edit your paper by focusing on:
- Academic style
- Vague sentences
- Style consistency
See an example

If you want to know more about the research process , methodology , research bias , or statistics , make sure to check out some of our other articles with explanations and examples.
Methodology
- Sampling methods
- Simple random sampling
- Stratified sampling
- Cluster sampling
- Likert scales
- Reproducibility
Statistics
- Null hypothesis
- Statistical power
- Probability distribution
- Effect size
- Poisson distribution
Research bias
- Optimism bias
- Cognitive bias
- Implicit bias
- Hawthorne effect
- Anchoring bias
- Explicit bias
The way you present your research problem in your introduction varies depending on the nature of your research paper . A research paper that presents a sustained argument will usually encapsulate this argument in a thesis statement .
A research paper designed to present the results of empirical research tends to present a research question that it seeks to answer. It may also include a hypothesis —a prediction that will be confirmed or disproved by your research.
As you cannot possibly read every source related to your topic, it’s important to evaluate sources to assess their relevance. Use preliminary evaluation to determine whether a source is worth examining in more depth.
This involves:
- Reading abstracts , prefaces, introductions , and conclusions
- Looking at the table of contents to determine the scope of the work
- Consulting the index for key terms or the names of important scholars
A research hypothesis is your proposed answer to your research question. The research hypothesis usually includes an explanation (“ x affects y because …”).
A statistical hypothesis, on the other hand, is a mathematical statement about a population parameter. Statistical hypotheses always come in pairs: the null and alternative hypotheses . In a well-designed study , the statistical hypotheses correspond logically to the research hypothesis.
Formulating a main research question can be a difficult task. Overall, your question should contribute to solving the problem that you have defined in your problem statement .
However, it should also fulfill criteria in three main areas:
- Researchability
- Feasibility and specificity
- Relevance and originality
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the “Cite this Scribbr article” button to automatically add the citation to our free Citation Generator.
McCombes, S. (2023, November 21). Writing Strong Research Questions | Criteria & Examples. Scribbr. Retrieved September 25, 2024, from https://www.scribbr.com/research-process/research-questions/
Is this article helpful?
Shona McCombes
Other students also liked, how to define a research problem | ideas & examples, how to write a problem statement | guide & examples, 10 research question examples to guide your research project, "i thought ai proofreading was useless but..".
I've been using Scribbr for years now and I know it's a service that won't disappoint. It does a good job spotting mistakes”
Literature Searching

Characteristics of a good research question
The first step in a literature search is to construct a well-defined question. This helps in ensuring a comprehensive and efficient search of the available literature for relevant publications on your topic. The well-constructed research question provides guidance for determining search terms and search strategy parameters.
A good or well-constructed research question is:
- Original and of interest to the researcher and the outside world
- It is clear and focused: it provides enough specifics that it is easy to understand its purpose and it is narrow enough that it can be answered. If the question is too broad it may not be possible to answer it thoroughly. If it is too narrow you may not find enough resources or information to develop a strong argument or research hypothesis.
- The question concept is researchable in terms of time and access to a suitable amount of quality research resources.
- It is analytical rather than descriptive. The research question should allow you to produce an analysis of an issue or problem rather than a simple description of it. In other words, it is not answerable with a simple “yes” or “no” but requires a synthesis and analysis of ideas and sources.
- The results are potentially important and may change current ideas and/or practice
- And there is the potential to develop further projects with similar themes
The question you ask should be developed for the discipline you are studying. A question appropriate for Physical Therapy, for instance, is different from an appropriate one in Sociology, Political Science or Microbiology .
The well-constructed question provides guidance for determining search terms and search strategy parameters. The process of developing a good question to research involves taking your topic and breaking each aspect of it down into its component parts.
One well-established way that can be used both for creating research questions and developing strategies is known as PICO(T). The PICO framework was designed primarily for questions that include clinical interventions and comparisons, however other types of questions may also be able to follow its principles. If the PICO framework does not precisely fit your question, using its principles can help you to think about what you want to explore even if you do not end up with a true PICO question.
References/Additional Resources
Fandino W. (2019). Formulating a good research question: Pearls and pitfalls. Indian journal of anaesthesia , 63 (8), 611–616.
Vandenbroucke, J. P., & Pearce, N. (2018). From ideas to studies: how to get ideas and sharpen them into research questions . Clinical epidemiology , 10 , 253–264.
Ratan, S. K., Anand, T., & Ratan, J. (2019). Formulation of Research Question - Stepwise Approach . Journal of Indian Association of Pediatric Surgeons , 24 (1), 15–20.
Lipowski, E.E. (2008). Developing great research questions. American Journal of Health-System Pharmacy, 65(17) , 1667–1670.
FINER Criteria
Another set of criteria for developing a research question was proposed by Hulley (2013) and is known as the FINER criteria.
FINER stands for:
Feasible – Writing a feasible research question means that it CAN be answered under objective aspects like time, scope, resources, expertise, or funding. Good questions must be amenable to the formulation of clear hypotheses.
Interesting – The question or topic should be of interest to the researcher and the outside world. It should have a clinical and/or educational significance – the “so what?” factor.
Novel – In scientific literature, novelty defines itself by being an answer to an existing gap in knowledge. Filling one of these gaps is highly rewarding for any researcher as it may represent a real difference in peoples’ lives.
Good research leads to new information. An investigation which simply reiterates what is previously proven is not worth the effort and cost. A question doesn’t have to be completely original. It may ask whether an earlier observation could be replicated, whether the results in one population also apply to others, or whether enhanced measurement methods can make clear the relationship between two variables.
Ethical – In empirical research, ethics is an absolute MUST. Make sure that safety and confidentiality measures are addressed, and according to the necessary IRB protocols.
Relevant – An idea that is considered relevant in the healthcare community has better chances to be discussed upon by a larger number of researchers and recognized experts, leading to innovation and rapid information dissemination.
The results could potentially be important and may change current ideas and/or practice.
Cummings, S.R., Browner, W.S., & Hulley, S.B. (2013). Conceiving the research question and developing the study plan. In: Designing clinical research (Hulley, S. R. Cummings, W. S. Browner, D. Grady, & T. B. Newman, Eds.; Fourth edition.). Wolters Kluwer/Lippincott Williams & Wilkins. Pp. 14-22.
- << Previous: Major Steps in a Literature Search
- Next: Types of Research Questions >>
Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, automatically generate references for free.
- Knowledge Base
- Research process
- How to Write a Research Proposal | Examples & Templates
How to Write a Research Proposal | Examples & Templates
Published on 30 October 2022 by Shona McCombes and Tegan George. Revised on 13 June 2023.
A research proposal describes what you will investigate, why it’s important, and how you will conduct your research.
The format of a research proposal varies between fields, but most proposals will contain at least these elements:
Introduction
Literature review.
- Research design
Reference list
While the sections may vary, the overall objective is always the same. A research proposal serves as a blueprint and guide for your research plan, helping you get organised and feel confident in the path forward you choose to take.
Table of contents
Research proposal purpose, research proposal examples, research design and methods, contribution to knowledge, research schedule, frequently asked questions.
Academics often have to write research proposals to get funding for their projects. As a student, you might have to write a research proposal as part of a grad school application , or prior to starting your thesis or dissertation .
In addition to helping you figure out what your research can look like, a proposal can also serve to demonstrate why your project is worth pursuing to a funder, educational institution, or supervisor.
| Show your reader why your project is interesting, original, and important. | |
| Demonstrate your comfort and familiarity with your field. Show that you understand the current state of research on your topic. | |
| Make a case for your . Demonstrate that you have carefully thought about the data, tools, and procedures necessary to conduct your research. | |
| Confirm that your project is feasible within the timeline of your program or funding deadline. |
Research proposal length
The length of a research proposal can vary quite a bit. A bachelor’s or master’s thesis proposal can be just a few pages, while proposals for PhD dissertations or research funding are usually much longer and more detailed. Your supervisor can help you determine the best length for your work.
One trick to get started is to think of your proposal’s structure as a shorter version of your thesis or dissertation , only without the results , conclusion and discussion sections.
Download our research proposal template
Prevent plagiarism, run a free check.
Writing a research proposal can be quite challenging, but a good starting point could be to look at some examples. We’ve included a few for you below.
- Example research proposal #1: ‘A Conceptual Framework for Scheduling Constraint Management’
- Example research proposal #2: ‘ Medical Students as Mediators of Change in Tobacco Use’
Like your dissertation or thesis, the proposal will usually have a title page that includes:
- The proposed title of your project
- Your supervisor’s name
- Your institution and department
The first part of your proposal is the initial pitch for your project. Make sure it succinctly explains what you want to do and why.
Your introduction should:
- Introduce your topic
- Give necessary background and context
- Outline your problem statement and research questions
To guide your introduction , include information about:
- Who could have an interest in the topic (e.g., scientists, policymakers)
- How much is already known about the topic
- What is missing from this current knowledge
- What new insights your research will contribute
- Why you believe this research is worth doing
As you get started, it’s important to demonstrate that you’re familiar with the most important research on your topic. A strong literature review shows your reader that your project has a solid foundation in existing knowledge or theory. It also shows that you’re not simply repeating what other people have already done or said, but rather using existing research as a jumping-off point for your own.
In this section, share exactly how your project will contribute to ongoing conversations in the field by:
- Comparing and contrasting the main theories, methods, and debates
- Examining the strengths and weaknesses of different approaches
- Explaining how will you build on, challenge, or synthesise prior scholarship
Following the literature review, restate your main objectives . This brings the focus back to your own project. Next, your research design or methodology section will describe your overall approach, and the practical steps you will take to answer your research questions.
| ? or ? , , or research design? | |
| , )? ? | |
| , , , )? | |
| ? |
To finish your proposal on a strong note, explore the potential implications of your research for your field. Emphasise again what you aim to contribute and why it matters.
For example, your results might have implications for:
- Improving best practices
- Informing policymaking decisions
- Strengthening a theory or model
- Challenging popular or scientific beliefs
- Creating a basis for future research
Last but not least, your research proposal must include correct citations for every source you have used, compiled in a reference list . To create citations quickly and easily, you can use our free APA citation generator .
Some institutions or funders require a detailed timeline of the project, asking you to forecast what you will do at each stage and how long it may take. While not always required, be sure to check the requirements of your project.
Here’s an example schedule to help you get started. You can also download a template at the button below.
Download our research schedule template
| Research phase | Objectives | Deadline |
|---|---|---|
| 1. Background research and literature review | 20th January | |
| 2. Research design planning | and data analysis methods | 13th February |
| 3. Data collection and preparation | with selected participants and code interviews | 24th March |
| 4. Data analysis | of interview transcripts | 22nd April |
| 5. Writing | 17th June | |
| 6. Revision | final work | 28th July |
If you are applying for research funding, chances are you will have to include a detailed budget. This shows your estimates of how much each part of your project will cost.
Make sure to check what type of costs the funding body will agree to cover. For each item, include:
- Cost : exactly how much money do you need?
- Justification : why is this cost necessary to complete the research?
- Source : how did you calculate the amount?
To determine your budget, think about:
- Travel costs : do you need to go somewhere to collect your data? How will you get there, and how much time will you need? What will you do there (e.g., interviews, archival research)?
- Materials : do you need access to any tools or technologies?
- Help : do you need to hire any research assistants for the project? What will they do, and how much will you pay them?
Once you’ve decided on your research objectives , you need to explain them in your paper, at the end of your problem statement.
Keep your research objectives clear and concise, and use appropriate verbs to accurately convey the work that you will carry out for each one.
I will compare …
A research aim is a broad statement indicating the general purpose of your research project. It should appear in your introduction at the end of your problem statement , before your research objectives.
Research objectives are more specific than your research aim. They indicate the specific ways you’ll address the overarching aim.
A PhD, which is short for philosophiae doctor (doctor of philosophy in Latin), is the highest university degree that can be obtained. In a PhD, students spend 3–5 years writing a dissertation , which aims to make a significant, original contribution to current knowledge.
A PhD is intended to prepare students for a career as a researcher, whether that be in academia, the public sector, or the private sector.
A master’s is a 1- or 2-year graduate degree that can prepare you for a variety of careers.
All master’s involve graduate-level coursework. Some are research-intensive and intend to prepare students for further study in a PhD; these usually require their students to write a master’s thesis . Others focus on professional training for a specific career.
Critical thinking refers to the ability to evaluate information and to be aware of biases or assumptions, including your own.
Like information literacy , it involves evaluating arguments, identifying and solving problems in an objective and systematic way, and clearly communicating your ideas.
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the ‘Cite this Scribbr article’ button to automatically add the citation to our free Reference Generator.
McCombes, S. & George, T. (2023, June 13). How to Write a Research Proposal | Examples & Templates. Scribbr. Retrieved 23 September 2024, from https://www.scribbr.co.uk/the-research-process/research-proposal-explained/
Is this article helpful?
Shona McCombes
Other students also liked, what is a research methodology | steps & tips, what is a literature review | guide, template, & examples, how to write a results section | tips & examples.
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
The PMC website is updating on October 15, 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Indian J Anaesth
- v.60(9); 2016 Sep
How to write a research proposal?
Department of Anaesthesiology, Bangalore Medical College and Research Institute, Bengaluru, Karnataka, India
Devika Rani Duggappa
Writing the proposal of a research work in the present era is a challenging task due to the constantly evolving trends in the qualitative research design and the need to incorporate medical advances into the methodology. The proposal is a detailed plan or ‘blueprint’ for the intended study, and once it is completed, the research project should flow smoothly. Even today, many of the proposals at post-graduate evaluation committees and application proposals for funding are substandard. A search was conducted with keywords such as research proposal, writing proposal and qualitative using search engines, namely, PubMed and Google Scholar, and an attempt has been made to provide broad guidelines for writing a scientifically appropriate research proposal.
INTRODUCTION
A clean, well-thought-out proposal forms the backbone for the research itself and hence becomes the most important step in the process of conduct of research.[ 1 ] The objective of preparing a research proposal would be to obtain approvals from various committees including ethics committee [details under ‘Research methodology II’ section [ Table 1 ] in this issue of IJA) and to request for grants. However, there are very few universally accepted guidelines for preparation of a good quality research proposal. A search was performed with keywords such as research proposal, funding, qualitative and writing proposals using search engines, namely, PubMed, Google Scholar and Scopus.
Five ‘C’s while writing a literature review

BASIC REQUIREMENTS OF A RESEARCH PROPOSAL
A proposal needs to show how your work fits into what is already known about the topic and what new paradigm will it add to the literature, while specifying the question that the research will answer, establishing its significance, and the implications of the answer.[ 2 ] The proposal must be capable of convincing the evaluation committee about the credibility, achievability, practicality and reproducibility (repeatability) of the research design.[ 3 ] Four categories of audience with different expectations may be present in the evaluation committees, namely academic colleagues, policy-makers, practitioners and lay audiences who evaluate the research proposal. Tips for preparation of a good research proposal include; ‘be practical, be persuasive, make broader links, aim for crystal clarity and plan before you write’. A researcher must be balanced, with a realistic understanding of what can be achieved. Being persuasive implies that researcher must be able to convince other researchers, research funding agencies, educational institutions and supervisors that the research is worth getting approval. The aim of the researcher should be clearly stated in simple language that describes the research in a way that non-specialists can comprehend, without use of jargons. The proposal must not only demonstrate that it is based on an intelligent understanding of the existing literature but also show that the writer has thought about the time needed to conduct each stage of the research.[ 4 , 5 ]
CONTENTS OF A RESEARCH PROPOSAL
The contents or formats of a research proposal vary depending on the requirements of evaluation committee and are generally provided by the evaluation committee or the institution.
In general, a cover page should contain the (i) title of the proposal, (ii) name and affiliation of the researcher (principal investigator) and co-investigators, (iii) institutional affiliation (degree of the investigator and the name of institution where the study will be performed), details of contact such as phone numbers, E-mail id's and lines for signatures of investigators.
The main contents of the proposal may be presented under the following headings: (i) introduction, (ii) review of literature, (iii) aims and objectives, (iv) research design and methods, (v) ethical considerations, (vi) budget, (vii) appendices and (viii) citations.[ 4 ]
Introduction
It is also sometimes termed as ‘need for study’ or ‘abstract’. Introduction is an initial pitch of an idea; it sets the scene and puts the research in context.[ 6 ] The introduction should be designed to create interest in the reader about the topic and proposal. It should convey to the reader, what you want to do, what necessitates the study and your passion for the topic.[ 7 ] Some questions that can be used to assess the significance of the study are: (i) Who has an interest in the domain of inquiry? (ii) What do we already know about the topic? (iii) What has not been answered adequately in previous research and practice? (iv) How will this research add to knowledge, practice and policy in this area? Some of the evaluation committees, expect the last two questions, elaborated under a separate heading of ‘background and significance’.[ 8 ] Introduction should also contain the hypothesis behind the research design. If hypothesis cannot be constructed, the line of inquiry to be used in the research must be indicated.
Review of literature
It refers to all sources of scientific evidence pertaining to the topic in interest. In the present era of digitalisation and easy accessibility, there is an enormous amount of relevant data available, making it a challenge for the researcher to include all of it in his/her review.[ 9 ] It is crucial to structure this section intelligently so that the reader can grasp the argument related to your study in relation to that of other researchers, while still demonstrating to your readers that your work is original and innovative. It is preferable to summarise each article in a paragraph, highlighting the details pertinent to the topic of interest. The progression of review can move from the more general to the more focused studies, or a historical progression can be used to develop the story, without making it exhaustive.[ 1 ] Literature should include supporting data, disagreements and controversies. Five ‘C's may be kept in mind while writing a literature review[ 10 ] [ Table 1 ].
Aims and objectives
The research purpose (or goal or aim) gives a broad indication of what the researcher wishes to achieve in the research. The hypothesis to be tested can be the aim of the study. The objectives related to parameters or tools used to achieve the aim are generally categorised as primary and secondary objectives.
Research design and method
The objective here is to convince the reader that the overall research design and methods of analysis will correctly address the research problem and to impress upon the reader that the methodology/sources chosen are appropriate for the specific topic. It should be unmistakably tied to the specific aims of your study.
In this section, the methods and sources used to conduct the research must be discussed, including specific references to sites, databases, key texts or authors that will be indispensable to the project. There should be specific mention about the methodological approaches to be undertaken to gather information, about the techniques to be used to analyse it and about the tests of external validity to which researcher is committed.[ 10 , 11 ]
The components of this section include the following:[ 4 ]
Population and sample
Population refers to all the elements (individuals, objects or substances) that meet certain criteria for inclusion in a given universe,[ 12 ] and sample refers to subset of population which meets the inclusion criteria for enrolment into the study. The inclusion and exclusion criteria should be clearly defined. The details pertaining to sample size are discussed in the article “Sample size calculation: Basic priniciples” published in this issue of IJA.
Data collection
The researcher is expected to give a detailed account of the methodology adopted for collection of data, which include the time frame required for the research. The methodology should be tested for its validity and ensure that, in pursuit of achieving the results, the participant's life is not jeopardised. The author should anticipate and acknowledge any potential barrier and pitfall in carrying out the research design and explain plans to address them, thereby avoiding lacunae due to incomplete data collection. If the researcher is planning to acquire data through interviews or questionnaires, copy of the questions used for the same should be attached as an annexure with the proposal.
Rigor (soundness of the research)
This addresses the strength of the research with respect to its neutrality, consistency and applicability. Rigor must be reflected throughout the proposal.
It refers to the robustness of a research method against bias. The author should convey the measures taken to avoid bias, viz. blinding and randomisation, in an elaborate way, thus ensuring that the result obtained from the adopted method is purely as chance and not influenced by other confounding variables.
Consistency
Consistency considers whether the findings will be consistent if the inquiry was replicated with the same participants and in a similar context. This can be achieved by adopting standard and universally accepted methods and scales.
Applicability
Applicability refers to the degree to which the findings can be applied to different contexts and groups.[ 13 ]
Data analysis
This section deals with the reduction and reconstruction of data and its analysis including sample size calculation. The researcher is expected to explain the steps adopted for coding and sorting the data obtained. Various tests to be used to analyse the data for its robustness, significance should be clearly stated. Author should also mention the names of statistician and suitable software which will be used in due course of data analysis and their contribution to data analysis and sample calculation.[ 9 ]
Ethical considerations
Medical research introduces special moral and ethical problems that are not usually encountered by other researchers during data collection, and hence, the researcher should take special care in ensuring that ethical standards are met. Ethical considerations refer to the protection of the participants' rights (right to self-determination, right to privacy, right to autonomy and confidentiality, right to fair treatment and right to protection from discomfort and harm), obtaining informed consent and the institutional review process (ethical approval). The researcher needs to provide adequate information on each of these aspects.
Informed consent needs to be obtained from the participants (details discussed in further chapters), as well as the research site and the relevant authorities.
When the researcher prepares a research budget, he/she should predict and cost all aspects of the research and then add an additional allowance for unpredictable disasters, delays and rising costs. All items in the budget should be justified.
Appendices are documents that support the proposal and application. The appendices will be specific for each proposal but documents that are usually required include informed consent form, supporting documents, questionnaires, measurement tools and patient information of the study in layman's language.
As with any scholarly research paper, you must cite the sources you used in composing your proposal. Although the words ‘references and bibliography’ are different, they are used interchangeably. It refers to all references cited in the research proposal.
Successful, qualitative research proposals should communicate the researcher's knowledge of the field and method and convey the emergent nature of the qualitative design. The proposal should follow a discernible logic from the introduction to presentation of the appendices.
Financial support and sponsorship
Conflicts of interest.
There are no conflicts of interest.
writing@chalmers
Basic features of a proposal.
There are some basic features that all proposals are likely to contain, which are summarised in the table below (based on information in Björk, Björk & Räisänen, 1996). However, the information provided is only a guideline – you may need to adjust according to your document’s purpose and your discipline’s preferences.
Academic Proposals
An academic proposal, such as an application for PhD studies, usually looks a bit different from a business proposal. The following sections are commonly found in an academic proposal:
- Introduction and Theoretical Framework
- Statement of the Problem
- Purpose of the Study
- Review of the Literature
- Research Questions and/or Hypotheses
- The Design – Methods and Procedures
- Limitations and Delimitations
- Significance of the Study
You will find information on all the sections here: The Elements of a Proposal by Frank Pajares
N.B. Information regarding features of other proposal types will be available here in the future.
Reference : Björk,L., Björk, M. & C. Räisänen (1996). Academic Writing, A University Writing Course . Studentlitteratur.
Redirect Notice
Inclusion across the lifespan in human subjects research.
Learn about the Inclusion Across the Lifespan policy and how to comply with this policy in applications and progress reports. All human subjects research supported by NIH must include participants of all ages, including children and older adults, unless there are scientific or ethical reasons not to include them.
The purpose of the Inclusion Across the Lifespan Policy is to ensure individuals are included in clinical research in a manner appropriate to the scientific question under study so that the knowledge gained from NIH-funded research is applicable to all those affected by the researched diseases/conditions. The policy expands the Inclusion of Children in Clinical Research Policy to include individuals of all ages, including children and older adults . The policy also requires that the age at enrollment of each participant be collected in progress reports.
Implementation
The Inclusion Across the Lifespan policy is now in effect, and applies to all grant applications submitted for due dates on or after January 25, 2019 . The policy also applies to solicitations for Research & Development contracts issued January 25, 2019 or later, and intramural studies submitted on/after this date. Ongoing, non-competing awards will be expected to comply with the policy at the submission of a competing renewal application. Research that was submitted before January 25, 2019 continues to be subject to the Inclusion of Children in Clinical Research Policy .
Applications & Proposals
Applications and proposals involving human subjects research must address plans for including individuals across the lifespan in the PHS Human Subjects and Clinical Trial Information Form. Any age-related exclusions must include a rationale and justification based on a scientific or ethical basis. Refer to the PHS Human Subjects and Clinical Trial Information Form Instructions for complete guidance on what to address.
Peer Review
Scientific Review Groups will assess each application/proposal as being "acceptable" or "unacceptable" with regard to the age-appropriate inclusion or exclusion of individuals in the research project. For additional information on review considerations, refer to the Guidelines for the Review of Inclusion in Clinical Research . For information regarding the coding used to rate inclusion during peer review, see the list of NIH Peer Review Inclusion Codes .
Progress Reports
NIH recipients/offerors must submit individual-level data on participant age at enrollment in progress reports. Age at enrollment must be provided along with information on sex or gender, race, and ethnicity in the Inclusion Enrollment Report. Units for reporting age at enrollment range from minutes to years.
Policy Notices
| : NIH Policy and Guidelines on the Inclusion of Individuals Across the Lifespan as Participants in Research Involving Human Subjects | This policy revises previous policy and guidelines regarding the inclusion of children in research. Changes to the policy include (1) the applicability of the policy to individuals of all ages, (2) clarification of potentially acceptable reasons for excluding participants on age, and (3) a requirement to provide data on participant age at enrollment in progress reports. | December 19, 2017 |
| : Inclusion of Children in Clinical Research: Change in NIH Definition | For the purposes of inclusion policy, a child is defined as individuals under 18 years old. Applicants/offerors for NIH funding are still expected to justify the age range of the proposed participants in their clinical research. | October 13, 2015 |
| NIH Policy and Guidelines on The Inclusion of Children as Participants in Research Involving Human Subjects | The goal of this policy is to increase the participation of children in research so that adequate data will be developed to support the treatment modalities for disorders and conditions that affect adults and may also affect children. | March 6, 1998 |
| Infographic that walks through the elements of the existing dataset or resource definition to help users understand whether how it applies to their research. | August 2, 2024 | |
| Report on the representation of participants in human subjects studies from fiscal years 2018-2021 for FY2018 projects associated with the listed Research, Condition, and Disease Categorization (RCDC) categories. | October 31, 2023 | |
| This document describes several mock studies as examples of how to consider the Inclusion Across the Lifespan policy in study design and eligibility criteria. Examples include commentary on scientific and ethical reasons that may be acceptable or unacceptable for age-based exclusion. | September 09, 2023 | |
| This one-page resource highlights allowable costs for NIH grants that can be utilized to enhance inclusion through recruitment and retention activities. Allowable costs listed in the NIH Grants Policy Statement are provided with examples of inclusion-related activities. | August 10, 2023 | |
| April 20, 2022 | ||
| NIH’s Inclusion Policy Officer Dawn Corbett covers inclusion plans during peer review and post-award in Part 2 of this NIH All About Grants podcast miniseries. | April 20, 2022 | |
| Using the Participant-level Data Template | For research that falls under the Inclusion Across the Lifespan policy, submission of individual-level data is required in progress reports. This tip sheet serves as a quick guide for using the participant-level data template in the Human Subjects System to populate data in the cumulative (actual) enrollment table. | January 20, 2022 |
| : Recruitment and Retention | Document listing resources on recruitment and retention of women, racial and ethnic minorities, and individuals across the lifespan. Resources include toolkits, articles, and more. | May 9, 2022 |
| : Including Diverse Populations in NIH-funded Clinical Research | Video presentation by the NIH Inclusion Policy Officer for the NIH Grants Conference PreCon event, Human Subjects Research: Policies, Clinical Trials, & Inclusion, in December 2022. The presentation explains NIH inclusion policies and requirements for applicants and recipients. | January 27, 2023 |
| - (PDF - 1.1 MB) | Report summarizing the presentations and discussions that took place during the Inclusion Across the Lifespan II Workshop on September 2, 2020. | December 10, 2020 |
| : Some Thoughts Following the NIH Inclusion Across the Lifespan II Workshop | Blog post by NIH's Deputy Director of Extramural Research, Dr. Mike Lauer, highlighting the Inclusion Across the Lifespan II Workshop. | December 10, 2020 |
| Entering Inclusion Data Using the Participant Level Data Template | This video tutorial demonstrates how to enter inclusion data using the Participant Level Data Template in the Human Subjects System (HSS). | February 26, 2020 |
| Guidance for Applying the Inclusion Across the Lifespan Policy | At-a-glance guidance for complying with the policy in applications and progress reports. | May 3, 2019 |
| The Inclusion Across the Lifespan Policy | The "All About Grants" podcast featuring an interview with the NIH Inclusion Policy Officer about the Inclusion Across the Lifespan policy. | August 27, 2018 |
| HSS overview and training information | As of June 9, 2018, the Human Subjects System (HSS) replaced the Inclusion Management System (IMS). Similar to IMS, HSS is used by NIH staff, grant applicants, and recipients to manage human subjects information, including inclusion information. | May 25, 2018 |
| The Inclusion Across the Lifespan Policy | Blog post by Dr. Mike Lauer, Deputy Director of Extramural Research, and Dawn Corbett, NIH Inclusion Policy Officer, titled Understanding Age in the NIH Portfolio: Implementation of the NIH Inclusion Across the Lifespan Policy | November 13, 2018 |
| Inclusion Across the Lifespan | Summary report from the Inclusion Across the Lifespan workshop held June 1-2, 2017 | July, 2017 |
Upcoming Events
DHSR One pager of resources for external users
- Human Subjects Research
- National Institute on Aging
- National Institute of Child Health and Human Development
- For NIH Staff
Have additional questions? Contact your program officer or the Inclusion policy team: [email protected]
Application of Pharmacometrics in Advancing the Clinical Research of Antibody-Drug Conjugates: Principles and Modeling Strategies
- Review Article
- Published: 26 September 2024
Cite this article

- Xiuqi Li 1 ,
- Dan Liu 2 ,
- Shupeng Liu 1 ,
- Mengyang Yu 1 ,
- Xiaofei Wu 1 &
- Hongyun Wang ORCID: orcid.org/0000-0002-9140-0127 1
Antibody-drug conjugates (ADCs) have become a pivotal area in the research and development of antitumor drugs. They provide innovative possibilities for tumor therapy by integrating the tumor-targeting capabilities of monoclonal antibodies with the cytotoxic effect of small molecule drugs. Pharmacometrics, an important discipline, facilitates comprehensive understanding of the pharmacokinetic characteristics of ADCs by integrating clinical trial data through modeling and simulation. However, due to the complex structure of ADCs, their modeling approaches are still unclear. In this review, we analyzed published population pharmacokinetic models for ADCs and classified them into single-analyte, two-analyte, and three-analyte models. We also described the benefits, limitations, and recommendations for each model. Furthermore, we suggested that the development of population pharmacokinetic models for ADCs should be rigorously considered and established based on four key aspects: (1) research objectives; (2) available in vitro and animal data; (3) accessible clinical information; and (4) the capability of bioanalytical methods. This review offered insights to guide the application of pharmacometrics in the clinical research of ADCs, thereby contributing to more effective therapeutic development.
This is a preview of subscription content, log in via an institution to check access.
Access this article
Subscribe and save.
- Get 10 units per month
- Download Article/Chapter or eBook
- 1 Unit = 1 Article or 1 Chapter
- Cancel anytime
Price includes VAT (Russian Federation)
Instant access to the full article PDF.
Rent this article via DeepDyve
Institutional subscriptions

Figure from Gupta et al. [ 14 ], under CCC license

Figure from Garrett et al. [ 11 ], under CC BY 4.0 license

Figure from Li et al. [ 20 ], under CC BY-NC 4.0 license.

Figure from Chudasama et al. [ 22 ], under CCC license.

Dumontet C, Reichert JM, Senter PD, Lambert JM, Beck A. Antibody-drug conjugates come of age in oncology. Nat Rev Drug Discov. 2023;22(8):641–61. https://doi.org/10.1038/s41573-023-00709-2 .
Article CAS PubMed Google Scholar
Deslandes A. Comparative clinical pharmacokinetics of antibody-drug conjugates in first-in-human Phase 1 studies. MAbs. 2014;6(4):859–70. https://doi.org/10.4161/mabs.28965 .
Article PubMed PubMed Central Google Scholar
Fu Z, Li S, Han S, Shi C, Zhang Y. Antibody drug conjugate: the “biological missile” for targeted cancer therapy. Signal Transduct Target Ther. 2022;7(1):93. https://doi.org/10.1038/s41392-022-00947-7 .
Article CAS PubMed PubMed Central Google Scholar
Lin K, Tibbitts J. Pharmacokinetic considerations for antibody drug conjugates. Pharm Res. 2012;29(9):2354–66. https://doi.org/10.1007/s11095-012-0800-y .
Hamblett KJ, Senter PD, Chace DF, et al. Effects of drug loading on the antitumor activity of a monoclonal antibody drug conjugate. Clin Cancer Res. 2004;10(20):7063–70. https://doi.org/10.1158/1078-0432.CCR-04-0789 .
Singh AP, Shin YG, Shah DK. Application of pharmacokinetic-pharmacodynamic modeling and simulation for antibody-drug conjugate development. Pharm Res. 2015;32(11):3508–25. https://doi.org/10.1007/s11095-015-1626-1 .
Marshall SF, Burghaus R, Cosson V, et al. Good practices in model-informed drug discovery and development: practice, application, and documentation. CPT Pharmacometrics Syst Pharmacol. 2016;5(3):93–122. https://doi.org/10.1002/psp4.12049 .
Petersdorf SH, Kopecky KJ, Slovak M, et al. A phase 3 study of gemtuzumab ozogamicin during induction and postconsolidation therapy in younger patients with acute myeloid leukemia. Blood. 2013;121(24):4854–60. https://doi.org/10.1182/blood-2013-01-466706 .
Fostvedt LK, Hibma JE, Masters JC, Vandendries E, Ruiz-Garcia A. Pharmacokinetic/pharmacodynamic modeling to support the re-approval of gemtuzumab ozogamicin. Clin Pharmacol Ther. 2019;106(5):1006–17. https://doi.org/10.1002/cpt.1500 .
Zuo P. Capturing the magic bullet: pharmacokinetic principles and modeling of antibody-drug conjugates. AAPS J. 2020;22(5):105. https://doi.org/10.1208/s12248-020-00475-8 .
Garrett M, Ruiz-Garcia A, Parivar K, et al. Population pharmacokinetics of inotuzumab ozogamicin in relapsed/refractory acute lymphoblastic leukemia and non-Hodgkin lymphoma. J Pharmacokinet Pharmacodyn. 2019;46(3):211–22. https://doi.org/10.1007/s10928-018-9614-9 .
Lewis Phillips GD, Li G, Dugger DL, et al. Targeting HER2-positive breast cancer with trastuzumab-DM1, an antibody-cytotoxic drug conjugate. Cancer Res. 2008;68(22):9280–90. https://doi.org/10.1158/0008-5472.CAN-08-1776 .
Center for Drug Evaluation and Research (CDR). Clinical pharmacology and biopharmaceutics review for trastuzumab emtansine. Application number: 125427Orig1s000. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2013/125427Orig1s000ClinPharmR.pdf . Accessed 20 Apr 2024.
Gupta M, Lorusso PM, Wang B, et al. Clinical implications of pathophysiological and demographic covariates on the population pharmacokinetics of trastuzumab emtansine, a HER2-targeted antibody-drug conjugate, in patients with HER2-positive metastatic breast cancer. J Clin Pharmacol. 2012;52(5):691–703. https://doi.org/10.1177/0091270011403742 .
DiJoseph JF, Dougher MM, Kalyandrug LB, et al. Antitumor efficacy of a combination of CMC-544 (inotuzumab ozogamicin), a CD22-targeted cytotoxic immunoconjugate of calicheamicin, and rituximab against non-Hodgkin’s B-cell lymphoma. Clin Cancer Res. 2006;12(1):242–9. https://doi.org/10.1158/1078-0432.CCR-05-1905 .
Scott LJ. Brentuximab Vedotin: A Review in CD30-Positive Hodgkin Lymphoma. Drugs. 2017;77(4):435–45. https://doi.org/10.1007/s40265-017-0705-5 .
Center for Drug Evaluation and Research (CDR). Clinical pharmacology and biopharmaceutics review for brentuximab vedotin. Application number: 125388Orig1s000. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2011/125388Orig1s000ClinPharmR.pdf . Accessed 20 Apr 2024.
Suri A, Mould DR, Liu Y, Jang G, Venkatakrishnan K. Population PK and exposure-response relationships for the antibody-drug conjugate brentuximab vedotin in CTCL patients in the phase III ALCANZA study. Clin Pharmacol Ther. 2018;104(5):989–99. https://doi.org/10.1002/cpt.1037 .
Suri A, Mould DR, Song G, et al. Population pharmacokinetic modeling and exposure-response assessment for the antibody-drug conjugate brentuximab vedotin in Hodgkin’s lymphoma in the phase III ECHELON-1 study. Clin Pharmacol Ther. 2019;106(6):1268–79. https://doi.org/10.1002/cpt.1530 .
Li H, Han TH, Hunder NN, Jang G, Zhao B. Population pharmacokinetics of brentuximab vedotin in patients with CD30-expressing hematologic malignancies. J Clin Pharmacol. 2017;57(9):1148–58. https://doi.org/10.1002/jcph.920 .
Lu D, Joshi A, Wang B, et al. An integrated multiple-analyte pharmacokinetic model to characterize trastuzumab emtansine (T-DM1) clearance pathways and to evaluate reduced pharmacokinetic sampling in patients with HER2-positive metastatic breast cancer. Clin Pharmacokinet. 2013;52(8):657–72. https://doi.org/10.1007/s40262-013-0060-y .
Chudasama VL, Schaedeli Stark F, Harrold JM, et al. Semi-mechanistic population pharmacokinetic model of multivalent trastuzumab emtansine in patients with metastatic breast cancer. Clin Pharmacol Ther. 2012;92(4):520–7. https://doi.org/10.1038/clpt.2012.153 .
Dornan D, Bennett F, Chen Y, et al. Therapeutic potential of an anti-CD79b antibody-drug conjugate, anti-CD79b-vc-MMAE, for the treatment of non-Hodgkin lymphoma. Blood. 2009;114(13):2721–9. https://doi.org/10.1182/blood-2009-02-205500 .
Center for Drug Evaluation and Research (CDR). Clinical pharmacology review for polatuzumab vedotin. Application number: 761121Orig1s000. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/761121Orig1s000ClinPharmR.pdf . Accessed 20 Apr 2024.
Lu D, Lu T, Gibiansky L, Li X, Li C, Agarwal P, Shemesh CS, Shi R, Dere RC, Hirata J, Miles D, Chanu P, Girish S, Jin JY. Integrated Two-Analyte Population Pharmacokinetic Model of Polatuzumab Vedotin in Patients With Non-Hodgkin Lymphoma. CPT Pharmacometrics Syst Pharmacol. 2020;9(1):48–59. https://doi.org/10.1002/psp4.12482 .
Breij EC, de Goeij BE, Verploegen S, et al. An antibody-drug conjugate that targets tissue factor exhibits potent therapeutic activity against a broad range of solid tumors. Cancer Res. 2014;74(4):1214–26. https://doi.org/10.1158/0008-5472.CAN-13-2440 .
Center for Drug Evaluation and Research (CDR). Muti-discipline review for tisotumab vedotin. Application number: 761208Orig1s000. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2021/761208Orig1s000MultidisciplineR.pdf . Accessed 20 Apr 2024.
Gibiansky L, Passey C, Voellinger J, et al. Population pharmacokinetic analysis for tisotumab vedotin in patients with locally advanced and/or metastatic solid tumors. CPT Pharmacometrics Syst Pharmacol. 2022;11(10):1358–70. https://doi.org/10.1002/psp4.12850 .
Passey C, Voellinger J, Gibiansky L, et al. Exposure-safety and exposure-efficacy analyses for tisotumab vedotin for patients with locally advanced or metastatic solid tumors. CPT Pharmacometrics Syst Pharmacol. 2023;12(9):1262–73. https://doi.org/10.1002/psp4.13007 .
Tai YT, Mayes PA, Acharya C, et al. Novel anti-B-cell maturation antigen antibody-drug conjugate (GSK2857916) selectively induces killing of multiple myeloma. Blood. 2014;123(20):3128–38. https://doi.org/10.1182/blood-2013-10-535088 .
Rathi C, Collins J, Struemper H, et al. Population pharmacokinetics of belantamab mafodotin, a BCMA-targeting agent in patients with relapsed/refractory multiple myeloma. CPT Pharmacometrics Syst Pharmacol. 2021;10(8):851–63. https://doi.org/10.1002/psp4.12660 .
Collins J, van Noort M, Rathi C, et al. Longitudinal efficacy and safety modeling and simulation framework to aid dose selection of belantamab mafodotin for patients with multiple myeloma. CPT Pharmacometrics Syst Pharmacol. 2023;12(10):1411–24. https://doi.org/10.1002/psp4.1301 .
Zammarchi F, Corbett S, Adams L, et al. ADCT-402, a PBD dimer-containing antibody drug conjugate targeting CD19-expressing malignancies. Blood. 2018;131(10):1094–105. https://doi.org/10.1182/blood-2017-10-813493 .
CDR. Center for drug evaluation and research. Muti-discipline review for loncastuximab tesirine. Application number: 761196Orig1s000. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2021/761196Orig1s000MultidisciplineR.pdf . Accessed 20 Apr 2024.
Hess B, Townsend W, Ai W, et al. Efficacy and safety exposure-response analysis of loncastuximab tesirine in patients with B cell non-Hodgkin lymphoma. AAPS J. 2021;24(1):11. https://doi.org/10.1208/s12248-021-00660-3 .
Kamath AV, Iyer S. Preclinical pharmacokinetic considerations for the development of antibody drug conjugates. Pharm Res. 2015;32(11):3470–9. https://doi.org/10.1007/s11095-014-1584-z .
Xu K, Liu L, Saad OM, et al. Characterization of intact antibody-drug conjugates from plasma/serum in vivo by affinity capture capillary liquid chromatography-mass spectrometry. Anal Biochem. 2011;412(1):56–66. https://doi.org/10.1016/j.ab.2011.01.004 .
Center for Drug Evaluation and Research (CDR). Clinical Pharmacology Considerations for Antibody-Drug Conjugates, Guide for industry. 2024. Available at: https://www.fda.gov/media/155997/download . Accessed 20 Apr 2024.
National Medical Products Administration. Technical guidelines for clinical development of anti-cancer antibody-drug conjugates. 2023. Available at: https://www.cde.org.cn/zdyz/downloadAtt?idCODE=65c496c5321ca27252149c11496a6a53 . Accessed 20 Apr 2024.
Sukumaran S, Zhang C, Leipold DD, et al. Development and translational application of an integrated, mechanistic model of antibody-drug conjugate pharmacokinetics. AAPS J. 2017;19(1):130–40. https://doi.org/10.1208/s12248-016-9993-z .
Chen Y, Samineni D, Mukadam S, et al. Physiologically based pharmacokinetic modeling as a tool to predict drug interaction for antibody-drug conjugates. Clin Pharmacokinet. 2015;54(1):81–93. https://doi.org/10.1007/s40262-014-0182-x .
Samineni D, Ding H, Ma F, et al. Physiologically based pharmacokinetic model-informed drug development for polatuzumab vedotin: label for drug-drug interactions without dedicated clinical trials. J Clin Pharmacol. 2020;60(Suppl 1):S120–31. https://doi.org/10.1002/jcph.1718 .
Liu SN, Li C. Clinical pharmacology strategies in supporting drug development and approval of antibody-drug conjugates in oncology. Cancer Chemother Pharmacol. 2021;87(6):743–65. https://doi.org/10.1007/s00280-021-04250-0 .
Download references
Acknowledgments
The authors thank all the contributors for their invaluable contributions to this review.
Author information
Authors and affiliations.
State Key Laboratory of Complex Severe and Rare Diseases, NMPA Key Laboratory for Clinical Research and Evaluation of Drug, Beijing Key Laboratory of Clinical PK & PD Investigation for Innovative Drugs, Clinical Pharmacology Research Center, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, 100730, China
Xiuqi Li, Shupeng Liu, Mengyang Yu, Xiaofei Wu & Hongyun Wang
College of Pharmacy, Shenyang Pharmaceutical University, Shenyang, Liaoning, 110016, China
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Hongyun Wang .
Ethics declarations
Conflicts of interest.
Xiuqi Li, Dan Liu, Shupeng Liu, Mengyang Yu, Xiaofei Wu, and Hongyun Wang declare they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
This research was supported by National High Level Hospital Clinical Research Funding (2022-PUMCH-B-118).
Author Contributions
XL: Investigation, visualization, writing—original draft. DL: Investigation, data curation. SL: Data curation, formal analysis. MY, XW: Valildation and methodology. HW: Writing—review and editing, supervision, funding acquisition, conceptualization.
Availability of data and material
Data availability is not applicable to this article as no new data were created or analyzed in this study.
Ethics approval
Not applicable.
Consent to participate
Consent for publication, code availability, rights and permissions.
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Reprints and permissions
About this article
Li, X., Liu, D., Liu, S. et al. Application of Pharmacometrics in Advancing the Clinical Research of Antibody-Drug Conjugates: Principles and Modeling Strategies. Clin Pharmacokinet (2024). https://doi.org/10.1007/s40262-024-01423-x
Download citation
Accepted : 03 September 2024
Published : 26 September 2024
DOI : https://doi.org/10.1007/s40262-024-01423-x
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Find a journal
- Publish with us
- Track your research

COMMENTS
Characteristics of a Successful Research Proposal . A successful research proposal: 1. Is innovative 2. Includes specific aims 3. Includes preliminary data 4. Describes approach 5. Indicates the significance of the proposal with regard to the specific award and conveys its impact on science and your personal growth.
The purpose of this article is to take students through a step-by-step process of writing good research proposals by discussing the essential ingredients of a good research proposal. ... and hypotheses as well as their distinguishing characteristics; (c) demonstrate a good understanding of the relevant elements to be considered in the ...
Cohesive ties. Cohesive ties are linguistic devices that link words, phrases and clauses into fluent sentences and paragraphs. These are important in a research proposal as they help you signpost and order your material. They are especially useful in the methodology section to ensure that your process is clearly and logically set out.
So in this guide, we'll outline all the elements of a good research proposal and explain why they're important. The elements of a good research proposal are the title, the introduction, literature review, aims and objectives, methodology, scope of the research, outline and timetable, and bibliography. It's important to include these ...
Research proposal examples. Writing a research proposal can be quite challenging, but a good starting point could be to look at some examples. We've included a few for you below. Example research proposal #1: "A Conceptual Framework for Scheduling Constraint Management".
Literature Review. This key component of the research proposal is the most time-consuming aspect in the preparation of your research proposal. As described in Chapter 5, the literature review provides the background to your study and demonstrates the significance of the proposed research.Specifically, it is a review and synthesis of prior research that is related to the problem you are setting ...
The main purpose of a scientific research proposal is to convince your audience that your project is worthwhile, and that you have the expertise and wherewithal to complete it. The elements of an effective research proposal mirror those of the research process itself, which we'll outline below. Essentially, the research proposal should ...
Research proposal formats vary depending on the size of the planned research, the number of participants, the discipline, the characteristics of the research, etc. The following outline assumes an individual researcher. This is just a SAMPLE; several other ways are equally good and can be successful.
Chapter 4 pp. 76-82: The Insider's Guide to Grantmaking How Foundations Find, Fund, and Manage Effective Programs. Proposals come in all shapes, lengths, and sizes; as program officer, you can review them according to any number of criteria. Nonetheless, it is possible to identify some generic characteristics that are hallmarks of a good ...
It puts the proposal in context. 3. The introduction typically begins with a statement of the research problem in precise and clear terms. 1. The importance of the statement of the research problem 5: The statement of the problem is the essential basis for the construction of a research proposal (research objectives, hypotheses, methodology ...
The title of your proposal may be different from the title of your final research project, but that is completely normal! Your findings may help you come up with a title that is more fitting for the final project. Characteristics of good proposal titles are (Al-Riyami, 2008): Catchy: It catches the reader's attention by peaking their interest.
INTRODUCTION. Scientific research is usually initiated by posing evidenced-based research questions which are then explicitly restated as hypotheses.1,2 The hypotheses provide directions to guide the study, solutions, explanations, and expected results.3,4 Both research questions and hypotheses are essentially formulated based on conventional theories and real-world processes, which allow the ...
Your proposal must be carefully planned and complete. It must be relevant and significant. It must be beneficial and effective. To accomplish those tasks, carefully and concisely answer the following questions in your proposal. Then complement that content with clear, effective writing to win over your reviewers.
Characteristics of a good . research proposal . 1. Adequate background information: ... In the preparation of an effective research proposal in library and information science, a researcher needs ...
characteristics; (c) demonstrate a good understanding of the relevant elements to be considered in the constituent sections of a good research proposal; and (d) comprehend the elements of a research proposal that should feature in the final written ... ter, find it difficult to write good research proposals. Thus, there is the need to explain ...
An interactive workshop on 'The Critical Steps for Successful Research: The Research Proposal and Scientific Writing' was conducted in conjunction with the 64 th Annual Conference of the Indian Pharmaceutical Congress-2012 at Chennai, India. In essence, research is performed to enlighten our understanding of a contemporary issue relevant to the needs of society.
This section of the proposal must provide a broad overview of the topic. The jargon and key terms used in the particular topic must also be thoroughly explained in order to avoid confusion. The interest of the researcher in the particular topic must also be clearly outlined while at the same time mentioning, albeit briefly at this point, a critical review of the main literature that covers the ...
Proposal Characteristics. 1. The need for the proposed activity is clearly established, preferably with data. 2. The most important ideas are highlighted and repeated in several places. 3. Appropriate detail is provided in all portions of the proposal such as in the objectives and a detailed schedule of activities for the project or project ...
A good research question is essential to guide your research paper, dissertation, or thesis. All research questions should be: Focused on a single problem or issue. Researchable using primary and/or secondary sources. Feasible to answer within the timeframe and practical constraints. Specific enough to answer thoroughly.
Characteristics of a good research question. The first step in a literature search is to construct a well-defined question. This helps in ensuring a comprehensive and efficient search of the available literature for relevant publications on your topic. The well-constructed research question provides guidance for determining search terms and ...
Research proposal examples. Writing a research proposal can be quite challenging, but a good starting point could be to look at some examples. We've included a few for you below. Example research proposal #1: 'A Conceptual Framework for Scheduling Constraint Management'.
A proposal needs to show how your work fits into what is already known about the topic and what new paradigm will it add to the literature, while specifying the question that the research will answer, establishing its significance, and the implications of the answer. [2] The proposal must be capable of convincing the evaluation committee about ...
acceptance or refutation each alternative solution. 4. An evaluation of the benefits of your proposal. positive effects of your proposal, for example, cost benefits or sustainability. 5. Possible counter arguments to your proposal. an awareness of possible objections to your proposal that you, in turn, argue against. 6.
Research that was submitted before January 25, 2019 continues to be subject to the Inclusion of Children in Clinical Research Policy. Applications & Proposals. Applications and proposals involving human subjects research must address plans for including individuals across the lifespan in the PHS Human Subjects and Clinical Trial Information Form.
Antibody-drug conjugates (ADCs) have become a pivotal area in the research and development of antitumor drugs. They provide innovative possibilities for tumor therapy by integrating the tumor-targeting capabilities of monoclonal antibodies with the cytotoxic effect of small molecule drugs. Pharmacometrics, an important discipline, facilitates comprehensive understanding of the pharmacokinetic ...