

68 Best Chemistry Experiments: Learn About Chemical Reactions
Whether you’re a student eager to explore the wonders of chemical reactions or a teacher seeking to inspire and engage your students, we’ve compiled a curated list of the top 68 chemistry experiments so you can learn about chemical reactions.
While the theories and laws governing chemistry can sometimes feel abstract, experiments bridge the gap between these concepts and their tangible manifestations. These experiments provide hands-on experiences illuminating the intricacies of chemical reactions, molecular structures, and elemental properties.
1. Covalent Bonds

By engaging in activities that demonstrate the formation and properties of covalent bonds, students can grasp the significance of these bonds in holding atoms together and shaping the world around us.
Learn more: Covalent Bonds
2. Sulfuric Acid and Sugar Demonstration
Through this experiment, students can develop a deeper understanding of chemical properties, appreciate the power of chemical reactions, and ignite their passion for scientific exploration.
3. Make Hot Ice at Home
Making hot ice at home is a fascinating chemistry experiment that allows students to witness the captivating transformation of a liquid into a solid with a surprising twist.
4. Make a Bouncing Polymer Ball

This hands-on activity not only allows students to explore the fascinating properties of polymers but also encourages experimentation and creativity.
Learn more: Thought Co
5. Diffusion Watercolor Art

This experiment offers a wonderful opportunity for students to explore the properties of pigments, observe how they interact with water, and discover the mesmerizing patterns and textures that emerge.
Learn more: Diffusion Watercolor Art
6. Exploding Baggie

The exploding baggie experiment is a captivating and dynamic demonstration that students should engage in with caution and under the supervision of a qualified instructor.
Learn more: Exploding Baggie
7. Color Changing Chemistry Clock

This experiment not only engages students in the world of chemical kinetics but also introduces them to the concept of a chemical clock, where the color change acts as a timekeeping mechanism.
Learn more: Color Changing Chemistry Clock
8. Pipe Cleaner Crystal Trees

By adjusting the concentration of the Borax solution or experimenting with different pipe cleaner arrangements, students can customize their crystal trees and observe how it affects the growth patterns.
Learn more: Pipe Cleaner Crystal Trees
9. How To Make Ice Sculptures

Through this experiment, students gain a deeper understanding of the physical and chemical changes that occur when water freezes and melts.
Learn more: Ice Sculpture
10. How to Make Paper

Through this hands-on activity, students gain a deeper understanding of the properties of cellulose fibers and the transformative power of chemical reactions.
Learn more: How to Make Paper
11. Color Changing Chemistry
Color changing chemistry is an enchanting experiment that offers a captivating blend of science and art. Students should embark on this colorful journey to witness the mesmerizing transformations of chemicals and explore the principles of chemical reactions.
12. Gassy Banana
The gassy banana experiment is a fun and interactive way for students to explore the principles of chemical reactions and gas production.
Learn more: Gassy Banana
13. Gingerbread Man Chemistry Experiment

This hands-on activity not only introduces students to the concepts of chemical leavening and heat-induced reactions but also allows for creativity in decorating and personalizing their gingerbread creations.
Learn more: Gingerbread Man Chemistry Experiment
14. Make Amortentia Potion

While the love potion is fictional, this activity offers a chance to explore the art of potion-making and the chemistry behind it.
Learn more: How to Make Amortentia Potion
15. Strawberry DNA Extraction
This hands-on experiment offers a unique opportunity to observe DNA, the building blocks of life, up close and learn about its structure and properties.
16. Melting Snowman

The melting snowman experiment is a fun and whimsical activity that allows students to explore the principles of heat transfer and phase changes.
Learn more: Melting Snowman
17. Acid Base Cabbage Juice

The acid-base cabbage juice experiment is an engaging and colorful activity that allows students to explore the pH scale and the properties of acids and bases.
By extracting the purple pigment from red cabbage leaves and creating cabbage juice, students can use this natural indicator to identify and differentiate between acidic and basic substances.
Learn more: Acid Base Cabbage Juice
18. Magic Milk

The magic milk experiment is a mesmerizing and educational activity that allows students to explore the concepts of surface tension and chemical reactions.
By adding drops of different food colors to a dish of milk and then introducing a small amount of dish soap, students can witness a captivating display of swirling colors and patterns.
Learn more: Magic Milk
19. Melting Ice with Salt and Water

Through this hands-on activity, students can gain a deeper understanding of the science behind de-icing and how different substances can influence the physical properties of water.
Learn more: Melting Ice with Salt and Water
20. Barking Dog Chemistry Demonstration

The barking dog chemistry demonstration is an exciting and visually captivating experiment that showcases the principles of combustion and gas production.
21. How to Make Egg Geodes

Making egg geodes is a fascinating and creative chemistry experiment that students should try. By using common materials like eggshells, salt, and food coloring, students can create their own beautiful geode-like crystals.
Learn more: How to Make Egg Geodes
22. Make Sherbet

This experiment not only engages the taste buds but also introduces concepts of acidity, solubility, and the chemical reactions that occur when the sherbet comes into contact with moisture.
Learn more: Make Sherbet
23. Hatch a Baking Soda Dinosaur Egg

As the baking soda dries and hardens around the toy, it forms a “shell” resembling a dinosaur egg. To hatch the egg, students can pour vinegar onto the shell, causing a chemical reaction that produces carbon dioxide gas.
Learn more: Steam Powered Family
24. Chromatography Flowers

By analyzing the resulting patterns, students can gain insights into the different pigments present in flowers and the science behind their colors.
Learn more: Chromatography Flowers
25. Turn Juice Into Solid

Turning juice into a solid through gelification is an engaging and educational chemistry experiment that students should try. By exploring the transformation of a liquid into a solid, students can gain insights of chemical reactions and molecular interactions.
Learn more: Turn Juice into Solid
26. Bouncy Balls
Making bouncy balls allows students to explore the fascinating properties of polymers, such as their ability to stretch and rebound.
27. Make a Lemon Battery
Creating a lemon battery is a captivating and hands-on experiment that allows students to explore the fundamentals of electricity and chemical reactions.
28. Mentos and Soda Project
The Mentos and soda project is a thrilling and explosive experiment that students should try. By dropping Mentos candies into a bottle of carbonated soda, an exciting eruption occurs.
29. Alkali Metal in Water
The reaction of alkali metals with water is a fascinating and visually captivating chemistry demonstration.
30. Rainbow Flame
The rainbow flame experiment is a captivating and visually stunning chemistry demonstration that students should explore.
31. Sugar Yeast Experiment
This experiment not only introduces students to the concept of fermentation but also allows them to witness the effects of a living organism, yeast, on the sugar substrate.
32. The Thermite Reaction
The thermite reaction is a highly energetic and visually striking chemical reaction that students can explore with caution and under proper supervision.
This experiment showcases the principles of exothermic reactions, oxidation-reduction, and the high temperatures that can be achieved through chemical reactions.
33. Polishing Pennies
Polishing pennies is a simple and enjoyable chemistry experiment that allows students to explore the concepts of oxidation and cleaning methods.
34. Elephant Toothpaste
The elephant toothpaste experiment is a thrilling and visually captivating chemistry demonstration that students should try with caution and under the guidance of a knowledgeable instructor.
35. Magic Potion
Creating a magic potion is an exciting and imaginative activity that allows students to explore their creativity while learning about the principles of chemistry.
36. Color Changing Acid-Base Experiment

Through the color changing acid-base experiment, students can gain a deeper understanding of chemical reactions and the role of pH in our daily lives.
Learn more: Color Changing Acid-Base Experiment
37. Fill up a Balloon
Filling up a balloon is a simple and enjoyable physics experiment that demonstrates the properties of air pressure. By blowing air into a balloon, you can observe how the balloon expands and becomes inflated.
38. Jello and Vinegar

The combination of Jello and vinegar is a fascinating and tasty chemistry experiment that demonstrates the effects of acid on a gelatin-based substance.
Learn more: Jello and Vinegar
39. Vinegar and Steel Wool Reaction

This experiment not only provides a visual demonstration of the oxidation process but also introduces students to the concept of corrosion and the role of acids in accelerating the process.
Learn more: Vinegar and Steel Wool Reaction
40. Dancing Rice

The dancing rice experiment is a captivating and educational demonstration that showcases the principles of density and buoyancy.
By pouring a small amount of uncooked rice into a clear container filled with water, students can witness the rice grains moving and “dancing” in the water.
Learn more: Dancing Rice
41. Soil Testing Garden Science

Soil testing is a valuable and informative experiment that allows students to assess the composition and properties of soil.
By collecting soil samples from different locations and analyzing them, students can gain insights into the nutrient content, pH level, and texture of the soil.
Learn more: Soil Testing Garden Science
42. Heat Sensitive Color Changing Slime

Creating heat-sensitive color-changing slime is a captivating and playful chemistry experiment that students should try.
Learn more: Left Brain Craft Brain
43. Experimenting with Viscosity

Experimenting with viscosity is an engaging and hands-on activity that allows students to explore the flow properties of liquids.
Viscosity refers to a liquid’s resistance to flow, and this experiment enables students to investigate how different factors affect viscosity.
Learn more: Experimenting with Viscosity
44. Rock Candy Science

Rock candy science is a delightful and educational chemistry experiment that students should try. By growing their own rock candy crystals, students can learn about crystal formation and explore the principles of solubility and saturation.
Learn more: Rock Candy Science
45. Baking Soda vs Baking Powder

Baking soda and baking powder have distinct properties that influence the leavening process in different ways.
This hands-on experiment provides a practical understanding of how these ingredients interact with acids and moisture to create carbon dioxide gas.
46. Endothermic and Exothermic Reactions Experiment

The endothermic and exothermic reactions experiment is an exciting and informative chemistry exploration that students should try.
By observing and comparing the heat changes in different reactions, students can gain a deeper understanding of energy transfer and the concepts of endothermic and exothermic processes.
Learn more: Education.com
47. Diaper Chemistry

By dissecting a diaper and examining its components, students can uncover the chemical processes that make diapers so effective at absorbing and retaining liquids.
Learn more: Diaper Chemistry
48. Candle Chemical Reaction
The “Flame out” experiment is an intriguing and educational chemistry demonstration that students should try. By exploring the effects of a chemical reaction on a burning candle, students can witness the captivating moment when the flame is extinguished.
49. Make Curds and Whey

This experiment not only introduces students to the concept of acid-base reactions but also offers an opportunity to explore the science behind cheese-making.
Learn more: Tinkerlab
50. Grow Crystals Overnight

By creating a supersaturated solution using substances like epsom salt, sugar, or borax, students can observe the fascinating process of crystal growth. This experiment allows students to explore the principles of solubility, saturation, and nucleation.
Learn more: Grow Crystals Overnight
51. Measure Electrolytes in Sports Drinks
The “Measure Electrolytes in Sports Drinks” experiment is an informative and practical chemistry activity that students should try.
By using simple tools like a multimeter or conductivity probe, students can measure the electrical conductivity of different sports drinks to determine their electrolyte content.
52. Oxygen and Fire Experiment
The oxygen and fire experiment is a captivating and educational chemistry demonstration that students should try. By observing the effects of oxygen on a controlled fire, students can witness the essential role of oxygen in supporting combustion.
53. Electrolysis Of Water

The electrolysis of water experiment is a captivating and educational chemistry demonstration that students should try.
Learn more: Electrolysis Of Water
54. Expanding Ivory Soap

The expanding Ivory Soap experiment is a fun and interactive chemistry activity that students should try. By placing a bar of Ivory soap in a microwave, students can witness the remarkable expansion of the soap as it heats up.
Learn more: Little Bins Little Hands
55. Glowing Fireworks

This experiment not only introduces students to the principles of pyrotechnics and combustion but also encourages observation, critical thinking, and an appreciation for the physics and chemistry behind.
Learn more: Glowing Fireworks
56. Colorful Polymer Chemistry

Colorful polymer chemistry is an exciting and vibrant experiment that students should try to explore polymers and colorants.
By combining different types of polymers with various colorants, such as food coloring or pigments, students can create a kaleidoscope of colors in their polymer creations.
Learn more: Colorful Polymer Chemistry
57. Sulfur Hexafluoride- Deep Voice Gas
This experiment provides a firsthand experience of how the density and composition of gases can influence sound transmission.
It encourages scientific curiosity, observation, and a sense of wonder as students witness the surprising transformation of their voices.
58. Liquid Nitrogen Ice Cream

Liquid nitrogen ice cream is a thrilling and delicious chemistry experiment that students should try. By combining cream, sugar, and flavorings with liquid nitrogen, students can create ice cream with a unique and creamy texture.
59. White Smoke Chemistry Demonstration

The White Smoke Chemistry Demonstration provides an engaging and visually captivating experience for students to explore chemical reactions and gases. By combining hydrochloric acid and ammonia solutions, students can witness the mesmerizing formation of white smoke.
60. Nitrogen Triiodide Chemistry Demonstration

The nitrogen triiodide chemistry demonstration is a remarkable and attention-grabbing experiment that students should try under the guidance of a knowledgeable instructor.
By reacting iodine crystals with concentrated ammonia, students can precipitate nitrogen triiodide (NI3), a highly sensitive compound.
61. Make a Plastic- Milk And Vinegar Reaction Experiment

Through the “Make a Plastic – Milk and Vinegar Reaction” experiment, students can gain a deeper understanding of the chemistry behind plastics, environmental sustainability, and the potential of biodegradable materials.
Learn more: Rookie Parenting
62. Eno and Water Experiment
This experiment not only introduces students to acid-base reactions but also engages their senses as they witness the visible and audible effects of the reaction.
63. The Eternal Kettle Experiment
By filling a kettle with alcohol and igniting it, students can investigate the behavior of the alcohol flame and its sustainability.
64. Coke and Chlorine Bombs
Engaging in this experiment allows students to experience the wonders of chemistry firsthand, making it an ideal choice to ignite their curiosity and passion for scientific exploration.
65. Set your Hand on Fire
This experiment showcases the fascinating nature of combustion and the science behind fire.
By carefully following proper procedures and safety guidelines, students can witness firsthand how the sanitizer’s high alcohol content interacts with an open flame, resulting in a brief but captivating display of controlled combustion.
66. Instant Ice Experiments
The Instant Ice Experiment offers an engaging and captivating opportunity for students to explore the wonders of chemistry and phase changes.
By using simple household ingredients, students can witness the fascinating phenomenon of rapid ice formation in just a matter of seconds.
67. Coke Cans in Acid and Base
Engaging in this experiment allows students to gain a deeper understanding of the chemical properties of substances and the importance of safety protocols in scientific investigations.
68. Color Changing Invisible Ink

The Color Changing Invisible Ink experiment offers an intriguing and fun opportunity for students to explore chemistry and learn about the concept of chemical reactions.
Learn more: Research Parent
Similar Posts:
- Top 100 Fine Motor Skills Activities for Toddlers and Preschoolers
- 37 Water Science Experiments: Fun & Easy
- Top 58 Creative Art Activities for Kids and Preschoolers
Leave a Comment Cancel reply
Save my name and email in this browser for the next time I comment.
- Grades 6-12
- School Leaders
Free printable to elevate your AI game 🤖
Every product is independently selected by (obsessive) editors. Things you buy through our links may earn us a commission.
45 Cool Chemistry Experiments, Demos, and Science Fair Projects
Don’t forget your safety equipment!

Bunsen burners, colorful chemicals, and the possibility of a (controlled) explosion or two? Everybody loves chemistry experiments! We’ve rounded up the best activities, demos, and chemistry science fair projects for kids and teens. Try them in the classroom or at home.
Easy Chemistry Experiments and Activities for All Ages
Chemistry science fair projects.
These chemistry experiments and activities are all easy to do using simple supplies you probably already have. Families can try them at home, or teachers and students can do them together in the classroom.
Mix up some magic milk
Kids love this colorful experiment, which explores the concept of surface tension. This is one of our favorite chemistry experiments to try at home, since the supplies are so basic and the results are so cool!

Taste the Rainbow
Teach your students about diffusion while creating a beautiful and tasty rainbow. You’ll definitely want to have extra Skittles on hand so your class can enjoy a few as well!
Learn more: Skittles Diffusion

Crystallize sweet treats
Crystal science experiments teach kids about supersaturated solutions. This one is easy to do at home, and the results are absolutely delicious!
Learn more: Candy Crystals
Make elephant-sized toothpaste
This fun project uses yeast and a hydrogen peroxide solution to create overflowing “elephant toothpaste.” You can also add an extra fun layer by having kids create toothpaste wrappers for their plastic bottles.
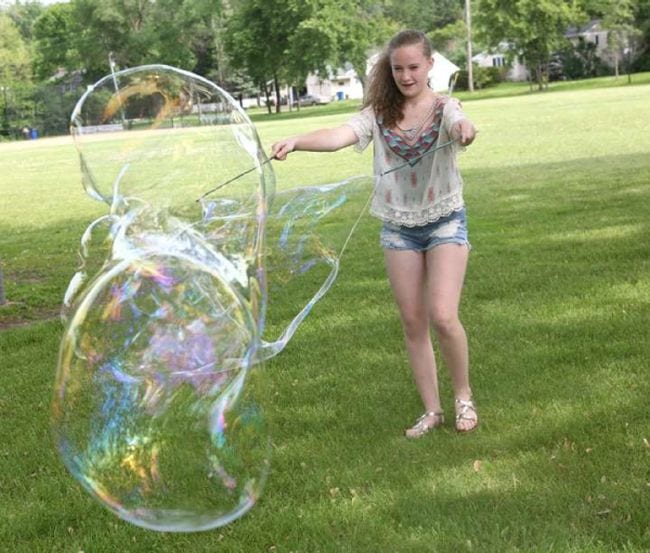
Blow the biggest bubbles you can
Add a few simple ingredients to dish soap solution to create the largest bubbles you’ve ever seen! Kids learn about surface tension as they engineer these bubble-blowing wands.
Learn more: Giant Soap Bubbles

Demonstrate the “magic” leakproof bag
So simple and so amazing! All you need is a zip-top plastic bag, sharp pencils, and some water to blow your kids’ minds. Once they’re suitably impressed, teach them how the “trick” works by explaining the chemistry of polymers.
Learn more: Leakproof Bag

Use apple slices to learn about oxidation
Have students make predictions about what will happen to apple slices when immersed in different liquids, then put those predictions to the test! Finally, have them record their observations.
Learn more: Apple Oxidation
Float a marker man
Their eyes will pop out of their heads when you “levitate” a stick figure right off the table. This experiment works due to the insolubility of dry-erase marker ink in water, combined with the lighter density of the ink.
Learn more: Floating Marker Man

Discover density with hot and cold water
There are a lot of easy science experiments you can do with density. This one is extremely simple, involving only hot and cold water and food coloring, but the visuals make it appealing and fun.
Learn more: Layered Water

Layer more liquids
This density demo is a little more complicated, but the effects are spectacular. Slowly layer liquids like honey, dish soap, water, and rubbing alcohol in a glass. Kids will be amazed when the liquids float one on top of the other like magic (except it is really science).
Learn more: Layered Liquids

Grow a carbon sugar snake
Easy science experiments can still have impressive results. This eye-popping chemical reaction demonstration only requires simple supplies like sugar, baking soda, and sand.
Learn more: Carbon Sugar Snake

Make homemade bouncy balls
These homemade bouncy balls are easy to make since all you need is glue, food coloring, borax powder, cornstarch, and warm water. You’ll want to store them inside a container like a plastic egg because they will flatten out over time.
Learn more: Make Your Own Bouncy Balls

Create eggshell chalk
Eggshells contain calcium, the same material that makes chalk. Grind them up and mix them with flour, water, and food coloring to make your very own sidewalk chalk.
Learn more: Eggshell Chalk

Make naked eggs
This is so cool! Use vinegar to dissolve the calcium carbonate in an eggshell to discover the membrane underneath that holds the egg together. Then, use the “naked” egg for another easy science experiment that demonstrates osmosis .
Learn more: Naked Egg Experiment
Turn milk into plastic
This sounds a lot more complicated than it is, but don’t be afraid to give it a try. Use simple kitchen supplies to create plastic polymers from plain old milk. Sculpt them into cool shapes when you’re done.

Test pH using cabbage
Teach kids about acids and bases without needing pH test strips. Simply boil some red cabbage and use the resulting water to test various substances—acids turn red and bases turn green.
Learn more: Cabbage pH

Clean some old coins
Use common household items to make old oxidized coins clean and shiny again in this simple chemistry experiment. Ask kids to predict (hypothesize) which will work best, then expand the learning by doing some research to explain the results.
Learn more: Cleaning Coins
Blow up a balloon (without blowing)
Chances are good you probably did easy science experiments like this when you were in school yourself. This well-known activity demonstrates the reactions between acids and bases. Fill a bottle with vinegar and a balloon with baking soda. Fit the balloon over the top, shake the baking soda down into the vinegar, and watch the balloon inflate.
Learn more: Balloon Experiments
Assemble a DIY lava lamp
This 1970s trend is back—as an easy science experiment! This activity combines acid/base reactions with density for a totally groovy result.

Explore how sugary drinks affect teeth
The calcium content of eggshells makes them a great stand-in for teeth. Use eggs to explore how soda and juice can stain teeth and wear down the enamel. Expand your learning by trying different toothpaste and toothbrush combinations to see how effective they are.
Learn more: Sugar and Teeth Experiment
Mummify a hot dog
If your kids are fascinated by the Egyptians, they’ll love learning to mummify a hot dog. No need for canopic jars ; just grab some baking soda and get started.
Extinguish flames with carbon dioxide
This is a fiery twist on acid-base experiments. Light a candle and talk about what fire needs in order to survive. Then, create an acid-base reaction and “pour” the carbon dioxide to extinguish the flame. The CO2 gas acts like a liquid, suffocating the fire.

Send secret messages with invisible ink
Turn your kids into secret agents! Write messages with a paintbrush dipped in lemon juice, then hold the paper over a heat source and watch the invisible become visible as oxidation goes to work.
Learn more: Invisible Ink
Set popcorn dancing
This is a fun version of the classic baking soda and vinegar experiment, perfect for the younger crowd. The bubbly mixture causes popcorn to dance around in the water.
Learn more: Dancing Popcorn Experiment
Shoot a soda geyser sky-high
You’ve always wondered if this really works, so it’s time to find out for yourself! Kids will marvel at the chemical reaction that sends diet soda shooting high in the air when Mentos are added.
Learn more: Mentos and Coke Experiment
All of these chemistry experiments are perfect for using the scientific method. Form a hypothesis, alter the variables, and then observe the results! You can simplify these projects for younger kids, or add more complexity for older students.

Break apart covalent bonds
Difficulty: Medium / Materials: Medium
Break the covalent bond of H 2 O into H and O with this simple experiment. You only need simple supplies for this one. Turn it into a science fair project by changing up the variables—does the temperature of the water matter? What happens if you try this with other liquids?
Learn more: Breaking Covalent Bonds
Measure the calories in various foods
Are the calorie counts on your favorite snacks accurate? Build your own calorimeter and find out! This kit from Home Science Tools has all the supplies you’ll need.

Detect latent fingerprints
Forensic science is engrossing and can lead to important career opportunities too. Explore the chemistry needed to detect latent (invisible) fingerprints, just like they do for crime scenes!
Learn more: Fingerprints Project
Use Alka-Seltzer to explore reaction rate
Difficulty: Easy / Materials: Easy
Tweak this basic concept to create a variety of high school chemistry science fair projects. Change the temperature, surface area, pressure, and more to see how reaction rates change.
Determine whether sports drinks really have more electrolytes than other beverages
Difficulty: Medium / Materials: Advanced
Are those pricey sports drinks really worth it? Try this experiment to find out. You’ll need some special equipment for this one; buy a complete kit at Home Science Tools .
Turn flames into a rainbow
You’ll need to get your hands on a few different chemicals for this experiment, but the wow factor will make it worth the effort. Make it a science project by seeing if different materials, air temperature, or other factors change the results.
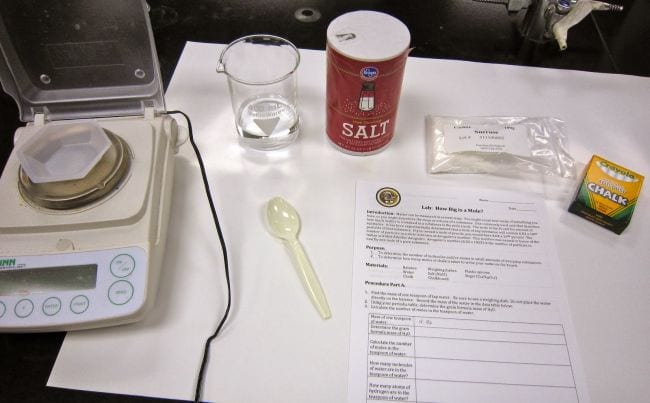
Discover the size of a mole
The mole is a key concept in chemistry, so it’s important to ensure students really understand it. This experiment uses simple materials like salt and chalk to make an abstract concept more concrete. Make it a project by applying the same procedure to a variety of substances, or determining whether outside variables have an effect on the results.
Learn more: How Big Is a Mole?

Cook up candy to learn mole and molecule calculations
This edible experiment lets students make their own peppermint hard candy while they calculate mass, moles, molecules, and formula weights. Tweak the formulas to create different types of candy and make this into a sweet science fair project!
Learn more: Candy Chemistry

Make soap to understand saponification
Take a closer look at an everyday item: soap! Use oils and other ingredients to make your own soap, learning about esters and saponification. Tinker with the formula to find one that fits a particular set of parameters.
Learn more: Saponification
Uncover the secrets of evaporation
Explore the factors that affect evaporation, then come up with ways to slow them down or speed them up for a simple science fair project.
Learn more: Evaporation
More Chemistry Experiment Science Fair Ideas
These questions and prompts can spark ideas for unique chemistry experiments:
- Compare the properties of sugar and artificial sweeteners.
- Explore the impact of temperature, concentration, and seeding on crystal growth.
- Test various antacids on the market to find the most effective product.
- What is the optimum temperature for yeast production when baking bread from scratch?
- Compare the vitamin C content of various fruits and vegetables.
- How does temperature affect enzyme-catalyzed reactions?
- Investigate the effects of pH on an acid-base chemical reaction.
- Devise a new natural way to test pH levels (such as cabbage leaves).
- What’s the best way to slow down metal oxidation (the form of rust)?
- How do changes in ingredients and method affect the results of a baking recipe?
Like these chemistry experiments? Don’t miss STEM Activities for Kids of All Ages and Interests .
Plus, get all the latest teaching news and ideas when you sign up for our free newsletters.

You Might Also Like

70 Best High School Science Fair Projects in Every Subject
Fire up the Bunsen burners! Continue Reading
Copyright © 2024. All rights reserved. 5335 Gate Parkway, Jacksonville, FL 32256

13 Awesome Chemical Reaction Experiments You Can Do At Home
Categories Activities & Ideas
Kids love to play mad scientist. And what better way to get kids excited about chemistry than with a few awesome chemical reaction experiments you can do at home?
Chemistry is an important branch of science that plays a big role in our everyday lives – so it’s definitely a subject children should learn about.

Luckily, chemistry is like performing magic – it’s fun to learn and do!
So, with that said, here are some awesome chemical experiments that can be done at home, all just as fun as they are educational.
Just remember: For health and safety, just make sure to supervise your child while doing the following experiments. If you’re a teacher, conduct these experiments in class or have the child’s parents supervise them at home.
1. Mentos & Diet Soda Chemical Volcano Eruption
Mixing mentos and diet soda is a classic experiment that produces a large chemical reaction that’s ideal for showing kids what chemistry is capable of. It’s safe and non-toxic, but the experiment can get messy, so it’s best to do it outdoors!
For this chemical reaction experiment, all you’ll need are a two-liter bottle of diet soda, a test tube or sheet of paper, an index card, and Mentos candies. You can use normal soda instead of diet soda, as well as M&Ms or Skittles instead of Mentos.
2. Baking Soda And Vinegar Balloon Experiment
If you have never seen a self-inflating balloon, this awesome chemical reaction experiment is sure to amaze the kids. The best thing is that baking soda and vinegar are common household items, so this experiment is easy enough to do at home.
All it involves is mixing baking soda and vinegar in a bottle and placing a balloon on the open end. The chemical reaction between baking soda and vinegar will release carbon dioxide gas that will inflate the balloon.
3. Green Flames
Copper sulfate is another item that can be easily found in most homes, but what’s special about it is that it can turn flames green. This chemical reaction experiment is best done in a fireplace or with a small controlled flame.
This is another awesome experiment that’s simple to do. Simply sprinkle copper sulfate onto a flame to see the color turn blue. You can also use alcohol or an alcohol-based fuel before lighting to achieve a brighter green color.
4. Fireproof Balloon
If you have more balloons left over from the baking soda and vinegar balloon experiment shown above, creating a fireproof balloon is another awesome chemical reaction experiment that’s easy to do and sure to impress.
For this experiment, all you need are two balloons, 50 milliliters of water, a syringe, and a candle or lighter. Simply blow two balloons up, one with 50ml of water added using the syringe. Hold each balloon over a candle to see which one bursts!
5. Foaming Volcano At Home Experiment
The foaming volcano experiment is similar to the baking soda and vinegar experiment except that it’s less explosive and involves different items. It uses baking soda, liquid soap, red food coloring, and acetic acid to create a foaming chemical reaction.
All it involves is using plasticine to create a volcano, then filling the inside with baking soda, a few drops of food coloring, and liquid soap. The last step is adding the acetic acid to watch the volcano erupt. Perform the experiment outside or in a tray to avoid mess!
6. Ice Cream In A Bag
Teach children that cooking also involves chemistry! This ice cream in a bag chemical reaction experiment is a great way to teach kids about freezing point depression, creating a tasty treat that they can also eat afterward.
The main items you’ll need for this experiment include zipper-top bags, ice, sodium chloride as table salt or rock salt, and ingredients for the ice cream, which are sugar, milk, whipping cream, and vanilla flavoring.
7. Elephant Toothpaste
This elephant toothpaste experiment is another simple chemical reaction that you can do at home using common household items. All you’ll need are an empty bottle (plastic or glass), 6% hydrogen peroxide, dry yeast, dish soap, food coloring, and warm water.
Similar to the foaming volcano, this elephant toothpaste experiment creates a chemical reaction that will see the bottle overflow with a foamy liquid. To make it extra fun, conduct the experiment with different food coloring!

8. Smoke Without Fire Experiment
“There’s no smoke without fire” unless you’re a chemist! This fun smoke-without-fire experiment demonstrates an awesome chemical reaction that creates smoke using a combination of concentrated hydrochloric acid and ammonia.
You will need some science equipment to perform this experiment, which includes three flasks connected by gas pipes. The chemical reaction produces a slow release of white smoke with, you guessed it, no fire or sparks.
9. Liquid Layer Density Tower
Density towers, or density columns, are a great way to show and explain the density levels of different liquids. You can use honey, corn syrup or pancake syrup, liquid dishwashing soap, water (with food coloring if desired), vegetable oil, rubbing alcohol, and lamp oil.
When you have a jar or container, the key is to put the heavier liquids in first, then pour the next liquids in using the side of the container. Even if you don’t get it right the first time, it’s a fun experiment that also creates a colorful display item!
If you and the kids don’t mind getting messy, this slime experiment is a fun chemical reaction that will create a thick, stretchy slime that becomes firmer and less sticky the more it’s played with. You can also add food coloring to create different colors of slime!
For this chemical reaction experiment, you’ll need borax powder, water, glue, and food coloring if you want to add color to the slime. You’ll also need a bowl, jar or measuring cup, and teaspoon for mixing.
11. Ignite A Candle With A Battery
Show your class that it’s possible to light a candle without matches or lighters with this awesome battery experiment! All it requires is an AA battery, chewing gum wrapper, cotton wool, scissors, and a candle.
You might not know this, but the chewing gum wrapper is coated with a layer of aluminum. As a good electrical conductor, the wrapper will generate an electric current when connected to the battery. The aluminum will heat up and light the cotton, which will ignite the candle.
12. DIY Lava Lamp
Lava lamps are awesome, so why not make a DIY one at home using an awesome chemical reaction? Although this DIY lava lamp won’t shine as brightly as a regular lava lamp, it’s still a fun experiment that will teach the kids about density.
The end result is a colorful liquid (use any kind of food coloring you like) with floating bubbles that can make a great bedroom ornament. It’s quick and simple, too, requiring just 30 minutes to create overall.
13. Homemade Science Christmas Ornament
Perfect for the holidays, this homemade science Christmas ornament experiment will get the kids to create glowing crystal icicles that can be hung outside or on the Christmas tree. It’s simple as well, only requiring borax powder, pipe cleaners, and string or twine.
To perform this chemical reaction experiment, you’ll need to add boiling hot water to a glass jar with borax. Twist the pipe cleaner around a pencil or pen to create a winding shape, then lower the pipe cleaner into the mixture with string. Leave until crystals start to form!
So there you have it: a list of awesome chemical experiments you can do at home to teach kids about the fascinating world of chemistry. These experiments are fun, educational, and sure to amaze kids of all ages!
Don’t forget: For health and safety reasons, just make sure to supervise your child while doing the above experiments or, if you’re a teacher, conduct the experiments in class or have the child’s parents supervise them at home.
- Recent Posts
- Homeschooling In High School: Pros And Cons - February 24, 2024
- How Do I Withdraw My Child From School To Homeschool? - February 23, 2024
- How To Not Go Crazy Homeschooling Kids: A Guide For Frazzled Parents - February 22, 2024
Related Posts:

65 Amazing Chemistry Experiments for Kids
Chemistry is so much fun, and we have tons of cool chemistry experiments to share with you. Like our awesome physics experiments , we decided we needed to put together a list of fun chemistry projects kids can do at home or in the classroom. Check out these examples of easy chemical reactions below!

65 Chemistry Experiments You Want To Try
We have divided our chemistry experiments below into chemical reactions, acids and bases, chromatography, solutions, polymers, and crystals. Some chemistry experiments also explore concepts in physics .

Chemical Reactions
A chemical reaction is a process where two or more substances react together to form a new chemical substance. This might look like a gas formed, cooking or baking, milk souring, etc.
Sometimes a physical change occurs, like our popcorn experiment or melting crayons, rather than a chemical change . However, these experiments below are all great examples of chemical change, where a new substance is formed.
CHECK OUT: Examples Of Physical Change and Chemical Change Examples
Can chemical reactions happen safely at home or in the classroom? Absolutely! This is one of the most fun parts of chemistry for kids, and you will find lots of ideas below for safe chemical reactions you can do with your junior scientists.

Acids And Bases
Acids and bases are important for many chemical processes in everyday life. An acid has hydrogen ions and can donate protons. Acids taste sour and have a pH from 0 to 7. Vinegar and citric acid are examples of acids.
Bases are molecules that can accept hydrogen ions. They have a pH higher than seven and can taste bitter. Sodium bicarbonate or baking soda and ammonia are examples of bases. Learn more about the pH scale.
Vinegar and baking soda experiments are classic acid-base reactions. You’ll also find experiments that use an acid such as vinegar or lemon juice. We have so many fun variations that your kids will love to try! Check out these acid-base chemistry experiments below.

Chromatography
Chromatography is a technique that involves the separation of a mixture into its parts so you can see each one individually.

A solution is a mixture of 2 or more solutes dissolved in a solvent up to its solubility limit. It most often refers to liquids, but solutions, gases, and solids are also possible. A solution will have its components evenly distributed throughout the mixture.
Chemistry experiments involving solutions are great for kids. Gather liquids you commonly find in your kitchen, oil, water, detergent, etc., and explore what dissolves.

A polymer is a huge molecule made of many smaller molecules layered together in repeating patterns called monomers. Putty, slime, and cornstarch are all examples of polymers. Learn more about the science of slime polymers .
Making slime is great for at-home chemistry! It’s also a classic middle school science demonstration for the classroom. Here are a few of our favorite slime recipes to get you started.

non-Newtonian Fluids
Learn how to make oobleck ! Explore polymers with a simple cornstarch and water mixture.

A crystal is a solid material with a highly ordered internal structure of atoms, molecules, or ions held together by chemical bonds. Grow crystals and observe them by mixing a super-saturated solution and leaving it for several days to let the crystals form.
Simple to grow and taste-safe, a sugar crystals experiment is more accessible for younger kids, but you can also try growing borax crystals for older kids.
Check out our fun theme variations of growing crystals too!

Chemistry For Preschoolers
Let’s keep it basic for our younger or junior scientists! Chemistry is all about how different materials are put together and what they are made up of, like atoms and molecules.
What can you do with your youngest scientists? While working 1-1 or in a very small group is ideal, you can explore chemistry in a few fun ways that don’t require a lengthy setup or a lot of directions to follow. Do NOT overcomplicate the ideas!
Take, for example, our very first baking soda science experiment (age 3). So simple to set up, but so lovely to watch the amazement on my son’s face.
Check out these fun ways for preschoolers to explore science…
- Make liquid mixtures! Mix water and oil in a jar, let it rest, and observe what happens.
- Make solid mixtures! Mix two solid items and observe the changes!
- Mix a solid and a liquid! Add ice to a drink and observe the changes!
- Make a reaction! Set up a tray with baking soda in small cups and colored vinegar in small cups with pipettes. Mix and observe!
- Make oobleck ! Mix cornstarch and water for a weird and messy science activity.
- Explore characteristics of things! Use new science words to describe how different materials feel. Explore squishy, hard, rough, smooth, wet, etc…
Much of preschool science is about you sharing new experiences with them that are relatable and simple. A sk questions, share new words, and offer verbal prompts to get them to communicate with you about what they see!
Chemistry Science Fair Projects
Science projects are excellent tools for older kids to demonstrate their knowledge of science. They can also be used in various environments, including classrooms, homeschools, and groups.
Kids can take everything they have learned about using the scientific method , stating a hypothesis, choosing variables , and analyzing and presenting data.
Want to turn one of these fun chemistry experiments into a science project? Then you will want to check out these helpful resources.
- Easy Science Fair Projects
- Science Project Tips From A Teacher
- Science Fair Board Ideas
More Helpful Science Resources
Here are a few resources that will help you introduce science more effectively to your kiddos or students and feel confident yourself when presenting materials. You’ll find helpful free printables throughout.
- Best Science Practices (as it relates to the scientific method)
- Science Vocabulary
- 8 Science Books for Kids
- All About Scientists
- Free Science Worksheets
- Science Supplies List
- Science Tools for Kids
- Scientific Method for Kids
- Citizen Science Guide
- Join us in the Club
Printable Science Projects For Kids
If you’re looking to grab all of our printable science projects in one convenient place plus exclusive worksheets and bonuses like a STEAM Project pack, our Science Project Pack is what you need! Over 300+ Pages!
- 90+ classic science activities with journal pages, supply lists, set up and process, and science information. NEW! Activity-specific observation pages!
- Best science practices posters and our original science method process folders for extra alternatives!
- Be a Collector activities pack introduces kids to the world of making collections through the eyes of a scientist. What will they collect first?
- Know the Words Science vocabulary pack includes flashcards, crosswords, and word searches that illuminate keywords in the experiments!
- My science journal writing prompts explore what it means to be a scientist!!
- Bonus STEAM Project Pack: Art meets science with doable projects!
- Bonus Quick Grab Packs for Biology, Earth Science, Chemistry, and Physics

17 Comments
- Pingback: Homemade Sand Slime Recipe for Kids Summer Science Activity
- Pingback: Magic Milk Classic Science Experiment Kids Science
- Pingback: Children to Leaders Foundation | 18 Great Online Resources to Get Your Child into Science
- Pingback: Balloon Baking Soda Vinegar Science Experiment for Kids
- Pingback: Erupting Apple Science and Apple Volcano Chemistry Activity for Kids
- Pingback: Simple Physics Activities Science Experiments STEM Ideas for Kids
- Pingback: How to Get Slime Out of Clothes (2 Methods to Try!)
- Pingback: How to Incorporate Chemistry at Home
- Pingback: Winter Magic Milk Science Project for Snowman Science Activities
- Pingback: Coffee Filter Flowers Science and STEAM Activity for Kids
- Pingback: Crystal Flowers Spring Science Experiment and Craft for Mother Day
- Pingback: Erupting Lemon Volcano Chemistry for Kids Science Activities
- Pingback: Grow Sugar Crystals for Edible Rock Candy Chemistry Experiment
- Pingback: 187Great Online Resources to Get Your Child into Science
- Pingback: The BEST Very Simple Science Experiments for Kids to Try Anywhere
- Pingback: Simple Ways To Take STEAM Outdoors This Summer
- Pingback: Geometric Bubble STEM Activity for Kids Summer Science
Comments are closed.

Subscribe to receive a free 5-Day STEM Challenge Guide
~ projects to try now ~.

TOP 10 chemical reactions that you can repeat at home
Safe and spectacular experiments
4. Release of gas
Materials and equipment :
sodium bicarbonate (baking soda);
- Fill a glass 1/3 full of water.
- Add a teaspoon of baking soda and a little vinegar.
- Light a match and gently lower it into the glass, not touching the mixture.
The match goes out.
Sodium bicarbonate (soda) is a compound of the following elements: sodium, hydrogen, carbon, and oxygen.
The reaction between sodium bicarbonate and vinegar forms unstable carbonic acid, which immediately decomposes to water and carbon dioxide. The carbon dioxide gas extinguishes the flame:
NaHCO₃ + CH₃COOH → CH₃COONa + H₂O + CO₂↑
5. Destructive vinegar
- Put an eggshell in a glass.
- Fill the glass half full of vinegar. Examine the contents after 12 hours.
The eggshell dissolves in the vinegar.
Vinegar is an acidic substance. It has the ability to break down several substances, such as the calcium carbonate contained in the eggshell:
CaCO₃ + 2CH₃COOH → Ca(CH₃COO)₂ + CO₂↑ + H₂O
9. Dissolving polystyrene in acetone
small pieces of polystyrene;
rubber gloves.
- Put on the gloves.
- Fill the bowl half full of acetone.
- Lower the small pieces of polystyrene into the bowl.
The pieces of polystyrene disappear. Just like this:
Polystyrene is expanded styrofoam and primarily consists of air. This is why it dissolves so remarkably in acetone.
10. Invisible ink
piece of paper;
cotton swab.
- Squeeze some lemon juice into a glass, add a few drops of water, and mix well.
- Dip a cotton swab into the solution and write on the paper with it. Let the paper dry.
- Hold over a burning candle.
The text appears.
Lemon juice contains acid, which darkens at high temperatures.
More experiments you can find in our chemistry sets for kids .

Dozens of experiments you can do at home
One of the most exciting and ambitious home-chemistry educational projects The Royal Society of Chemistry
Babble Dabble Do
50 Chemistry Projects That Will Amaze Kids!
February 26, 2019 by Ana Dziengel 5 Comments
Chemistry projects feel like magic , do they not? If you think about some of your favorite science projects, the ones you love to try with your kids or the ones that amazed YOU as a kid, more likely than not most of them involved chemistry.
Now I know a lot of us associate chemistry with lab coats, beakers and specialty ingredients but the reality is there are so many chemistry projects you can do using very simple, easy to find ingredients, often found in your own pantry. And since these types of simple chemistry projects use relatively safe ingredients, they are perfect to try with younger kids, ie. preschool and elementary aged children! In fact I think it’s so important for young kids to have a positive association with chemistry from a young age that fosters a love of this branch of science.
When most children are finally exposed to chemistry in school, it is at the high school level where the subject turns complex quickly; hopefully giving kids a chance to have fun at young age mixing up concoctions and watching chemical reactions will help carry their interest through the more complicated days of study ahead.
This post is a GIANT compilation of chemistry projects that would be great for the science fair, classroom demos, or at home science with your kids.
Before we get started let’s talk a little bit about what chemistry is and for parents I also included a section covering How to Do Chemistry Projects at Home. If you are a classroom teacher you can skip this section and head right to the projects here.
What is chemistry?
Chemistry is the branch of science that studies matter (anything that has mass and takes up space) and its properties, and how different substances (especially molecules and their atoms) interact, combine, and change to form new substances.
Here are some important definitions to know when working on chemistry projects:
- Element A substance that cannot be separated into any further substances. There are 120 known elements.
- Atom The smallest particle of an element
- Molecule Groups of atoms held together by a chemical bond.
- Ion An atom or molecule that has an electric charge
While most people think of chemistry purely in terms of chemical reactions, chemistry also covers the study of the states of matter as well as the density of substances.
The five branches of chemistry are:
- Analytical chemistry
- Physical chemistry
- Organic chemistry
- Inorganic chemistry
- Biochemistry
Read more about what each branch covers here.
How to Do Chemistry Projects at Home
Many chemistry projects can be done at home using simple materials and are a great way to foster a love of science in kids! I wholeheartedly believe that a wow factor in a project engages and inspires kids to learn more. If you want to try chemistry projects at home here are some suggestions and precautions:
Safety First
Even though most of the projects in this list use safe, easy to find materials they should be used with safety precautions and under adult supervision. Why? Sometimes the chemical reaction that ensues can irritate the skin or eye, can be harmful if swallowed, or is just plain sticky or messy and adults should be on hand to supervise use . Also be advised that there are a few projects on this list that do use materials that are unsafe for kids to handle. These projects are meant to be demonstrations only and are labeled accordingly.
- Use household items for chemistry The classic chemistry project that never fails to impress is the reaction of baking soda (sodium bicarbonate) and vinegar (look for a number of variations on this classic in our Acids and Bases section) but there are lots of other great ingredients for chemistry to find in your kitchen including sugar, salt, yeast, lemons, dish soap, milk, Kool- Aid, cabbage, gelatin, and food coloring to name a few…before you order any materials online, try some projects with pantry essentials.
- Safety Goggles
- Large plastic beakers
- Prepare for mess Since a lot of chemistry involves reactions and the ensuing mess, be sure to choose a place in your home that you can easily clean up and where you won’t worry about getting dirty. A patio, breakfast area, or the garage are great choices.
- Generous work area Be sure to have a large table available so everyone has plenty of room to work and/or view projects without bumping into each other.
- Access to Water Clean up is always easier with water at the ready! Choose a location near a hose or shop sink.
Managing Messes
- Hose it down Depending on the project I suggest doing super messy chemical reactions outside. That way spills can be hosed down easily.
- Painter’s Tarp & Trays If you cannot go outside a large plastic painter’s tarp is a great way to contain spills and mess. I also highly recommend doing projects on trays or cookie sheets. The raised edges help contain bubbly brews and are easy to dump out and wash.
- Dump station Have a bucket nearby to act as dump station for liquid reactions. Bring it around a table and dump at each station.
- Think about disposal Vinegar kills grass! Slime bits clogs drains! Be sure to consider where you can dump out the liquids safely.
Chemistry Projects for Kids
The following chemistry projects for kids are sorted by topic: Chemical Reactions, Acids and Bases, Carbon Reactions, Chromatography, Colloids & Solutions, Polymers, and Crystals.
Please note that many if these projects could fit in two or more categories in this post as they demonstrate various scientific and chemical processes. I only classified them once on this list.
Chemistry Projects with Chemical Reactions
What is a chemical reaction.
Chemical reactions occur when the chemical bonds in a substance are either destroyed or created. In other words the bonds in a molecule are broken during a chemical reaction and the atoms rearranged to create new molecules. Interestingly enough the number of original atoms does not change during the reaction, they are simply reconfigured.
An easy way to explain chemical reactions to kids is to use this analogy: Atoms are like letters, molecules are like words. Chemistry is like taking apart words and rearranging the letters to form a new word.
Read more about chemical reactions here.
Chemical Reactions Projects:
1. milk painting, 2. citrus battery, 3. elephant toothpaste.
<span data-mce-type=”bookmark” style=”display: inline-block; width: 0px; overflow: hidden; line-height: 0;” class=”mce_SELRES_start”></span>

4. Density Lava Lamps
To make a density lava lamp fill a plastic bottle with the following liquids: Clear corn syrup, water with a few drops of food coloring, and layer of vegetable oil. Be sure to leave a space at the top of the bottle. Wait until the liquids settle then add in a tablet of extra strength alka seltzer. Watch as the alka seltzer and water react and bubble up through the oil layer. To see this in a step by step video check out this video (Pssst this is one of our students!!!)
5. Plastic Milk and Curds & Whey Experiment
6 . color mixing.
Pour water into three clear plastic cups, then add blue, red, and yellow food coloring to each. Have an additional cup full of uncolored water available as well. Give your child an empty ice cube tray and pipettes and let them create different colors by mixing different ratios of two different primary colors in each ice cube compartment. The secondary colors are new colors created from two primary colors. This is a simple visual of how chemical reactions work.
7. Chemistry Clock
8. blow balloons with yeast and sugar, 9. shiny pennies.
- Collect dirty tarnished pennies.
- Pour different acidic liquids into shallow containers. Try vinegar, salsa, lemon & lime juice.
- Add a teaspoon of salt to each container and stir to combine.
- Place a handful of pennies in each container and soak for 5 minutes.
- Remove them from the solution and rinse in soapy water. Let dry on separate paper towels.
- Compare the results! Which ones are shiniest? Which are dull? Did any turn green?
Acids are corrosive and sour tasting. Liquids such as vinegar, lemon juice, and tomato juice are acids. Pennies are made from copper which tarnishes (turns dark) when exposed to oxygen over time. Placing the copper pennies in an acid will clean the copper oxide off them and make them shiny again.
Learn about Acids and Bases
Most liquids are either an acid or a base. Liquids with lots of hydrogen ions in them are considered acids. Liquids with many hydroxide ions are bases. Scientists use a scale called the ph scale to measure how acidic or basic a liquid is. The more hydrogen ions in a liquid the more acidic it is and ranks low on the ph scale. The more hydroxide ions in a liquid the more basic it is and ranks high on the ph scale. You can see what that looks like here.
When acids and bases are mixed chemical reactions occur and the solution becomes neutralized.
Acid and Bases Projects:
1. baking soda & vinegar volcano, 2. lemon volcano, 3. the colorful cabbage juice science experiment and acid base experiment with cabbage, 4. dancing rice, 5. green eggs & ham, 6. bubbly citric acid brew , 7. baking soda vs baking powder science experiment, 8. exploding bags, 9. rainbow rubber eggs , 10. surprise eggs , 11. rainbow wizard’s brew, chemistry projects with fire (carbon reactions).
Carbon is the most important element for life. Chemicals that contain carbon are called organic compounds. Carbon has two main forms: The first is in the hard form of diamonds and graphite, and the second is the impure form found in charcoal, coal and soot.
SAFTEY WARNING: Carbon reactions are always fascinating to watch however the presence of fire means that these experiments must be supervised by adults at all times!
Carbon Reactions Projects:
1. smoking fingers, 2. fire snake, 3. silver egg, 4. invisible ink, chromatography.
Chromatography is the process of separating mixtures. We usually think of it in terms of color hence the prefix -chroma, however in chemistry is means simply a method of separating mixtures by letting them slowly move past each other. It applies to both liquids and gasses. This is wonderful in-depth explanation of chromatography.
Chromatography Projects:
1. chromatography .
In this project you will separate the color black into other colors. Fold a coffee filter in half. Fold in half two more times until you have a triangular shape. Color the tip of the coffee filter with washable black marker. Get a good coat of ink on the filter. Add a small amount of water to a plastic cup. Place the black tip of the coffee filter in the cup Wait and observe. Come back to the filter after an hour or two and see what happens to the ink. As the coffee filter absorbs water through capillary action, the black ink moves through the filter and is separated by the water into other colors. You should see blue, green and even red as the water separates the ink.
2. Chromatography Flowers
3. chromatography art, 4. chromatography bags, colloids and solutions/solubility.
Colloids and Solutions are two types of homogenous mixtures.
- Colloids are mixtures in which a small particles of a substance are suspended throughout another substance but not chemically bonded. They are stable though and do not separate. Examples of colloids are gelatin, butter, mayonnaise, fog and smoke.
- Solutions are mixtures in which the particles of one substance are completely dissolved in another substance. The solute is the substance being dissolved and the solvent is the substance doing the dissolving. An example of a solution is saltwater.
If you want a more in-depth primer on solutions and colloids hop over here .
Colloid Projects:
1. colloid examples, 2. oobleck , 3. make butter , 4. gelatin streaking, solutions/solubility projects:, 5. ice sculptures , 6. ice cream in a bag.
A printable of the science facts at play here
7. Skittles Science
8. magical water blossoms <span data-mce-type=”bookmark” style=”display: inline-block; width: 0px; overflow: hidden; line-height: 0;” class=”mce_selres_start”></span>, 9. diffusion art, 10. paint solubility , 11. bleeding blossoms .
A polymer is a substance made up of a long chain of molecules. Polymers are typically flexible materials like plastic or gum.
The classic polymer kids LOVE to make is slime! Glue is already a polymer but when combined with sodium tetraborate (borax ) the protein molecules of the glue and the borate ions crosslink, making it harder for the molecules to move and forming the gooey, sticky, substance we know as slime.
Other polymers you are probably familiar with are plastic bags, balloons, instant snow, and even the powdery substance found in diapers that expands when wet.
Polymer Projects:
1. best basic slime .
Bonus: Get the Science Behind Slime printable here
2. Heat Sensitive Slime
3. diy bouncy balls, 4. magic plastic bag experiment, 5. instant terrariums, 6. how to make paper, 7. skewer through balloon , 8. dry erase figure and dry erase drawings, 9. recycled plastic flowers.
Crystals are a type of material that is formed by patterns of repeating molecules. There are four types of chemical bonds in crystals and therefore four categories of crystals. These are: Covalent, Molecular, Metallic, and Ionic Crystals. You can grow crystals by mixing up a super saturated solution (usually with a type of salt and water) and letting it settle over time so crystals will form. Check out the various types of easy to grow crystal below and go here to read more about the science of crystals .
Crystals Projects:
1. classic borax crystals , 2. overnight crystal garden, 3. egg geodes, 4. crystal wind catchers, 5. crystal landscapes, 6. candy geodes , 7. salt crystals, conclusion & more.
Alright you guys, do you feel like you have some good project ideas for exploring chemistry with kids? Many of these will make greats science fair projects. Be sure to start with them as a topic then start asking questions, form a hypotheses, and do some experiments.
Now I have to admit that I really fell in love with chemistry projects as an adult. Working with kids in camp, after school, and with my own kids at home I’ve had the chance to try fun chemistry projects and discovered that I love watching chemical reactions AND the reactions on the faces of kids and bystanders during demonstration or project!
If you have kids who fall in love with this branch of science please do check out the incredible book series Elements , Molecules , and Reactions by Theodore Gray (see the series in our Amazon science ideas list here ) The books are stunning, informative, easy to understand and, wait for it…funny!
Another valuable resource for kids who love chemistry is Mel Science’s Chemistry subscription box. They send you a starter kit for free with all the materials you’ll need and then each month you get a new chemistry experiment delivered to your door! This is great product because a lot of specialty chemistry ingredients are hard to find and these kits simplify getting the materials you need! Check it out here:
Are you passionate about raising creative kids?
Join over 22,179 parents and educators who want connect with kids and nurture their creative process through magical, easy projects you can do TOGETHER.
Subscribe to our email list to receive project ideas as well as offers for some our creative products.
If you want to read our privacy policy before subscribing, hop over here.
February 26, 2019 at 10:31 pm
These were some really awesome projects. I really liked the Citrus Battery projects. It seems simple and easy for a young kid. I wish schools should give more focus to such experiments instead of shoving down the theoretical knowledge down the throat of young kids.
Carol Biggs says
March 1, 2019 at 4:58 am
Is all of this info available on book form?
Ana Dziengel says
March 6, 2019 at 5:49 am
Not at this time but that’s a great idea!
Betsy Mitten says
March 5, 2019 at 10:43 am
Thank you for making this fantastic collection of experiments with clear directions and easy to understand explanations of the science behind the fun! I know I’ll refer to this list often. I especially appreciate the way the experiments are classified/organized. I teach art with science connections and we are already planning on chalkboard and magnetic slime :). I’ll be sure to tag Babble Dabble Do when I post photos of work inspired by this on target collection!
Kyra Rodriguez says
March 5, 2019 at 10:54 pm
These are all great ideas! I’m pretty sure the kids will have fun and love this activities
Leave a Reply Cancel reply
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.
Pin It on Pinterest

Homemade Chemistry – Household Chemical Reaction Experiments for Kids
Krystal DeVille
Updated on: July 30, 2023

You don’t always need a laboratory to carry out science experiments.
In fact, how about educating your kids with some household chemical reaction experiments? Here’s a list of some interesting hands-on activities that talk about different chemical reactions.
Table of Contents
9 Simple Chemistry Experiments for Your Kids to Try at Home
1. bubble power.

What You’ll Need:
- A plastic bottle
- 150 ml of hot water
The Process:
Take the plastic bottle and add three tsp of dry yeast and two tsp of sugar into it.
Now, start pouring hot water into the bottle slowly.
Cover the bottle with a balloon and wait for around half an hour.
What did you see? The balloon expands, and the liquid starts to foam. As the yeast mixes with sugar and releases carbon dioxide gas, you’ll see many bubbles rising to the surface, causing foamy liquid and balloon inflation. This is called the fermentation process.
2. Soot Experiment
Light the candle and hold the knife in the center of the flame for a few seconds.
Did you see the blade turning black? Keeping the blade in the flame leads to incomplete combustion of paraffin that is present in the candle. This produces carbon particles that eventually cover the blade.
3. Dry Ice Smoke Experiment
- Dry ice (solid carbon dioxide)
- A large container or bowl
- Preparation : First, make sure to handle dry ice with care. It’s extremely cold (-78.5 °C or -109.3 °F), so use gloves or tongs to handle it. Adult supervision is a must here.
- Add Warm Water : Fill a large container or bowl with warm water.
- Add Dry Ice : Using gloves or tongs, place a piece of dry ice into the warm water.
- Observe the Fog : You’ll see a thick fog start to form. This is actually a cloud of tiny water droplets condensing from the water vapor, as the dry ice sublimates directly from a solid to a gas (carbon dioxide).
- Optional Enhancement : You can add a few drops of dish soap to create bubbles filled with fog, or add food coloring for a visual effect.
Safety Considerations:
- Always handle dry ice with care, using gloves or tongs.
- Adult supervision is needed, especially when handling the dry ice.
- Dry ice should never be enclosed in a sealed container, as it can build up pressure and explode.
- Make sure the area is well-ventilated, as the carbon dioxide gas could displace oxygen in a confined space.
4. Vinegar Power

Take a glass and place an eggshell into it. Fill half of the glass with vinegar.
Wait for 12 hours and observe the change.
As vinegar is acidic, it breaks down various substances (like calcium carbonate) present in the eggshell. This causes the eggshell to dissolve in the vinegar.
I go more in-depth about this and have a dedicated article step-by-step here, How to Make An Eggshell Disappear Science Experiment .
5. Color Experiment

- Copper coin (a penny will work fine even though they are mostly zinc)
- Ammonia solution
The Process :
For this chemical reaction experiment, take a dark-coated copper coin. Pour ammonia solution over it. You’ll see the solution turning blue. When copper comes into contact with ammonia under the influence of oxygen, it forms a complex compound that results in a color change.
6. Fire Experiment
- Potassium permanganate crystals
Use potassium permanganate crystals to make a small hill-like structure.
Create a hollow in it and pour glycerin (add only a little quantity).
In case there’s no fire, add one or a maximum of two drops of water.
You’ll see the mixture catching fire as there’s a chemical reaction between potassium permanganate and glycerin.
7. Skittles Experiment

- Skittles (or any other such coated sweets)
- A plate or container (preferably white)
Take a white container or plate and arrange your skillets (or any other coated sweets) over it. Try to arrange them with alternate colors.
Now, pour the warm water into the container slowly. Push the skittles back into their place if they happen to move due to water.
Observe what happens next.
Skittles are coated with sugar and food coloring. So, as soon as you pour water over them, the sugar and color will start to dissolve and spread along through the water, giving you a colorful effect.
8. Invisible Ink Experiment
- Cotton swab
Take a glass, squeeze some lemon juice and mix it with a few drops of water.
Now, dip a cotton swab into the mixture and write something on the paper with it.
Allow the paper to dry. Once dry, hold it over the burning candle. Did you see the text? Is it magic?
The answer is – as lemon juice contains acid, it darkens at high temperatures. So, when you bring the paper closer to the burning candle, you see the text appearing.
9. Mint Candy Fountain

- Mint flavored candies
- A 2-liter bottle of soft drink
- A tube or a piece of paper
Caution: Carry out this household chemical reaction in an outdoor area, preferably your backyard.
Open the soft drink bottle and place it on the flat surface, so it does not tip over. It’s recommended to place the bottle in an area that is not surrounded by anything.
Now, take the paper, roll it up in the cylinder-shape. Make sure that the width is enough for the candies to slide through.
Seal the bottom of the roll with your hand and add the candies to the paper tube.
Hold the paper tube above the soft drink bottle and slide your finger in a way that the candies drop straight in. Drop all the candies in the bottle at the same time and move away from the bottle as fast as possible.
The carbon dioxide gas in the bottle does not form bubbles until you open the bottle. But the gas will escape with force while spilling some soft drink out if you open the bottle immediately after shaking it. So, adding anything to the soft drink will promote bubble formation while forcing it to escape.
Now, as you drop the mint candies in the drink, it offers a lot of surface area for the bubbles to form in huge numbers. So, as it begins to dissolve, it pushes the entire content, resulting in an incredible soft drink blast.
My kids really enjoyed trying other things to see if they can get a bigger reaction, sand surprising works great.
I put together a big list of Messy STEM Science Experiments for Kids.

Final Thoughts
Aren’t these household chemical reaction experiments worth a try? They’ll not only keep your kids engaged but will also help them learn some amazing chemistry lessons in the comfort of your home. If you’re looking for some interesting chemistry experiment kit for your kid, you may want to check out – Colorful Chemistry .
For many of these, all you need is a variety of household cleaning products you probably already have stocked at home. When doing any experiment, make sure the proper precautions are also taken to avoid any possible toxic fumes or combustion reactions.
Have you tried any of these experiments before? What was the result? Did your kids enjoy it? Do share your experience with us in the comments.
Leave a Comment Cancel reply
Save my name, email, and website in this browser for the next time I comment.
most recent

Activities and Games , Toy Gift Guides
Best stem subscription boxes for kids: hands-on reviews.

Science , Engineering , Math , Technology
Kiwico vs mel science: a complete breakdown.

Best Chemistry Kits for Kids (for Every Age!)

Teach Kids to Code , Activities and Games , Technology , Toy Gift Guides
11 best coding robots to teach kids to code (for all ages).

Activities and Games , Math
20 stem projects that are great for middle school.

Engineering
6 projects for learning about simple machines.

Amplify Your Outdoor Living Space with Garden Décor
STEM Education Guide
[email protected] STEM Education Guide 9125 SVL BOX Victorville, CA 92395
Your Compass for STEM Discovery
© 2024 STEM Education Guide

- Science Notes Posts
- Contact Science Notes
- Todd Helmenstine Biography
- Anne Helmenstine Biography
- Free Printable Periodic Tables (PDF and PNG)
- Periodic Table Wallpapers
- Interactive Periodic Table
- Periodic Table Posters
- Science Experiments for Kids
- How to Grow Crystals
- Chemistry Projects
- Fire and Flames Projects
- Holiday Science
- Chemistry Problems With Answers
- Physics Problems
- Unit Conversion Example Problems
- Chemistry Worksheets
- Biology Worksheets
- Periodic Table Worksheets
- Physical Science Worksheets
- Science Lab Worksheets
- My Amazon Books
Color Change Chemistry Projects – Chemical Reactions and More

Color change chemistry projects are among the most interesting and entertaining science experiments . Color change chemical reactions visually indicate chemical changes in matter. They illustrate pH changes, acid-base reactions, oxidation-reduction, and other key chemistry concepts. Other projects change color due to conformation changes, light emission, and simply mixing colors. There’s a color change chemistry project perfect for any occasion.
Here is a collection of color change projects.
Is Color Change a Chemical Change?
Color change often indicates a chemical change or chemical reaction, but sometimes colors change without altering the composition of a substance. For example, mixing blue and yellow food coloring makes a green liquid, but no chemical change occurs.
Color Change Chemical Reactions
These reactions are chemical changes. They include acid-base reactions, redox reactions, and precipitate formation.
- Briggs-Rauscher Oscillating Clock : A solution oscillates between colorless, amber, and blue in this classic chemistry demonstration.
- Blue Bottle Chemical Reaction : In this project, a blue liquid turns colorless before turning blue again.
- Chemical Traffic Light : This variation of the blue bottle reaction uses indigo carmine. The solution shifts colors between green, yellow, and red.
- Water to Wine to Milk to Beer : Pour liquid from one glass to another and watch it change into what looks like water, wine, milk, and finally beer.
- Two Color Chemiluminescence : Watch a solution produce a red glow and then a blue glow.
- Gatorade and Drain Cleaner : This variation of the blue bottle demonstration uses Gatorade and drain cleaner instead of chemicals from a lab. The colors you get depend on the products you choose.
- Red Cabbage Juice pH Indicator : Cabbage juice changes color depending on a liquid’s pH. The color change covers the entire rainbow.
- Fake Blood Chemical Reaction : Reacting an iron(III) solution and potassium thiocyanate solution makes the iron thiocyanate complex, which resembles fake blood.
- Color Change Violets : Change the color of violets or other flowers using household ammonia.
- Color Changing Chemical Volcano : This chemical volcano erupts lava that changes colors from purple to orange.
- Bubbles That Change Colors : These soap bubbles change colors using pH indicators.
- Disappearing Ink : Acid-base chemistry makes colored “ink” disappear as it dries and reappear using household ammonia.
- Color Change Liquid Thermometer : Tell the temperature by whether the liquid is pink or blue.
- Color-Changing Chameleon Eggs : This edible color change chemistry project changes the color of gelatin spheres, depending on the pH of the liquid surrounding them.
- Easy Color Changing Drinks : Turn a soft drink or cocktail into a color change chemistry project without making it taste bad.
- Color Changing Crystals : These crystals spontaneously change color due to a slow chemical reaction within them.
Holiday Color Change Chemistry Projects
These chemical reactions involve colors associated with holidays and other events.
- Natural Easter Egg Dyes : Use safe cooking ingredients as egg dyes and learn about natural pH indicators.
- Vanishing Valentine : A pink solution turns colorless as the oxidation state of its chemicals change.
- Hot and Cold Valentine : A pH indicator changes liquid from pink to colorless. A temperature change shifts the equilibrium between a normal and ionized form of a molecule, changing the solution pH and color.
- Water Into Wine or Water Into Blood : A clear liquid turns red. Use this color change chemistry project for either Easter or Halloween.
- Old Nassau or Halloween Reaction : A clear liquid turns orange and then black.
- Bleeding Paper : Goldenrod paper starts out golden yellow and turns blood red when you write your message.
- Christmas Chemistry Demonstration : This clock reaction is perfect for the winter holidays, as the liquid changes from green to red.
- Poinsettia pH Indicator : Extract pH indicator from a poinsettia plant and see test liquids to see if they turn the indicator red or green.
- Olympic Rings Demonstration : Pour clear liquid into five glasses and watch it turn the colors of the Olympic rings.
Colored Fire
Combustion is a chemical reaction and thus an example of a chemical change. However, the reason flames change color in these projects is because of light emission. Heating colorants excites their electrons. Photons released as electrons return to more stable energy states cause the color change.
- Colored Fire : Make a rainbow of colored flames using common chemicals.
- Colored Fire Pinecones : Use colorants on ordinary pinecones to spice up your campfire.
- Rainbow Flames Jack o’ Lantern : Instead of illuminating your pumpkin with a boring candle, make a rainbow of colored flames.
More Color Change Chemistry Projects
These colorful chemistry projects don’t necessarily involve chemical reactions. Other reasons for color changes include alterations in molecule shape, mixing colors, and scattering light.
- Rainbow Roses : Turn a white rose into a rainbow use. With a little know-how, change black roses into rainbows, too.
- Copper, Silver, and Gold Pennies : Change the chemical composition of coins and watch their colors change, too.
- Change Egg Yolk Color : Color egg whites using food coloring. Coloring egg yolks is trickier because the yolks are oil-based rather than water-based.
- Change Urine Color : It’s easy to change urine color. You may have done it accidentally once or twice!
- Make Rainbow Paper : Coat paper with a thin film that shows the colors of the rainbow.
- Thermal Nail Polish : Thermal nail polish changes color according to temperature. See how it works and make it yourself.
Related Posts
Science Fun

Crazy Chalk Chemical Reaction Science Experiment
In this fun and easy science experiment, we’re going to explore and investigate endothermic chemical reactions by mixing chalk and vinegar.
- Colored chalk
- Zip close baggie
- Clear plastic cup
Instructions:
- Put at least two different colors of chalk in the zip close baggie.
- Use the hammer to carefully smash the chalk into dust.
- Fill the clear plastic cup about ¾ of the way full with vinegar.
- Put about a tablespoon of the colored chalk dust into the vinegar and observe what happens.
EXPLORE AWESOME SCIENCE EXPERIMENT VIDEOS!
How it Works:
You may not have realized that regular old chalk fizzes and bubbles when mixed with vinegar. Chalk is made of calcium carbonate and when mixed with vinegar an endothermic chemical reaction occurs that releases carbon dioxide gas. This gas is what causes the fizzing and bubbles.
Make This A Science Project:
Find a location that can get messy. Put the chalk dust in a new baggie. Add vinegar, seal the baggie securely, and observe what happens. Test different types and brands of chalk to see if there are any observable differences.
EXPLORE TONS OF FUN AND EASY SCIENCE EXPERIMENTS!
SUBSCRIBE AND NEVER MISS A NEW SCIENCE FUN VIDEO!
previous experiment
Next experiment.
Easy Chemistry Experiments to Do at Home
These 12 projects use materials you probably already have
- Activities for Kids
- Chemical Laws
- Periodic Table
- Projects & Experiments
- Scientific Method
- Biochemistry
- Physical Chemistry
- Medical Chemistry
- Chemistry In Everyday Life
- Famous Chemists
- Abbreviations & Acronyms
- Weather & Climate
- Ph.D., Biomedical Sciences, University of Tennessee at Knoxville
- B.A., Physics and Mathematics, Hastings College
Looking for fun, educational activities to do at home? This list of easy chemistry experiments and science activities will allow you to perform experiments with materials you likely already have in your kitchen cupboards .
You don't need esoteric chemicals and a lab to have a good time with chemistry. Your average fourth-grader can make slime , and it doesn't get any less fun when you're older, so this is a good at-home experiment for kids and adults alike.
Borax Snowflake
Anne Helmenstine
Making a sparkly borax snowflake is a crystal-growing project that's safe and easy enough for kids. You can make shapes other than snowflakes, and you can color the crystals. If you use these as Christmas decorations and store them, the borax is a natural insecticide and will help keep your long-term storage area pest-free. If they develop a white precipitant, lightly rinse them but don't dissolve too much crystal.
Mentos and Diet Soda Fountain
This is a backyard activity best accompanied by a garden hose . The Mentos fountain is more spectacular than a baking soda volcano . If you make the volcano and find the eruption to be disappointing, substitute these ingredients.
Penny Chemistry
You can clean pennies, coat them with verdigris, and plate them with copper. This project demonstrates several chemical processes , yet the materials are easy to find and the science is safe enough for kids.
Invisible Ink
Invisible inks either react with another chemical to become visible or else weaken the structure of the paper so the message appears if you hold it over a heat source. But we're not talking about fire here; the heat of a normal light bulb is all that's required to darken the lettering. This baking soda recipe is nice because if you don't want to use a light bulb to reveal the message, you can just swab the paper with grape juice instead.
Colored Fire
Fire is fun. Colored fire is even better. These additives are safe. They won't, in general, produce smoke that is any better or worse for you than normal wood smoke. Depending on what you add, the ashes will have a different elemental composition from a normal wood fire, but if you're burning trash or printed material, you have a similar result. This is suitable for a home fire or campfire, plus most chemicals are found around the house (even of non-chemists).
Seven-Layer Density Column
Make a density column with many liquid layers . Heavier liquids sink to the bottom, while lighter (less dense) liquids float on top. This is an easy, fun, colorful science project that illustrates the concepts of density and miscibility.
Homemade Ice Cream in a Plastic Bag
Science experiments can taste good! Whether you're learning about freezing point depression or not, the ice cream is a delicious result either way. This cooking chemistry project potentially uses no dishes, so cleanup can be very easy.
Hot Ice (Sodium Acetate)
Got vinegar and baking soda ? If so, you can make " hot ice ," or sodium acetate , and then cause it to instantly crystallize from a liquid into "ice." The reaction generates heat, so the ice is hot. It happens so quickly that you can form crystal towers as you pour the liquid into a dish.
Burning Money
The " burning money trick " is a magic trick using chemistry . You can set a bill on fire, yet it won't burn. Are you brave enough to try it? All you need is a real bill.
Coffee Filter Chromatography
Exploring separation chemistry with coffee filter chromatography is a snap. A coffee filter works well, though if you don't drink coffee you can substitute a paper towel. You can also devise a project comparing the separation you get using different brands of paper towels. Leaves from outdoors can provide pigments. Frozen spinach is another good choice.
Baking Soda and Vinegar Foam Fight
The foam fight is a natural extension of the baking soda volcano . This easy chemistry experiment is a lot of fun and a little messy, but quick enough to clean up as long as you don't add food coloring to the foam.
- Easy Science Projects
- How to Make Flaming Gel
- Kitchen Science Experiments for Kids
- How to Make Elephant Toothpaste
- Safe Science Experiments
- Homemade Silly Putty Recipes
- Science Projects Photo Gallery
- Fried Green Egg Food Science Project
- Baggie Chemistry Experiments
- Top Chemistry Projects for Bored Kids
- Mad Scientist Party Theme
- Second Grade Science Fair Projects
- Chemistry Experiments With Pennies
- Science Experiments and Activities for Preschoolers
- Baking Soda Science Projects
- How to Make Glow in the Dark Slime
Your browser is not supported
Sorry but it looks as if your browser is out of date. To get the best experience using our site we recommend that you upgrade or switch browsers.
Find a solution
- Skip to main content
- Skip to navigation

- Back to parent navigation item
- Primary teacher
- Secondary/FE teacher
- Early career or student teacher
- Higher education
- Curriculum support
- Literacy in science teaching
- Periodic table
- Interactive periodic table
- Climate change and sustainability
- Resources shop
- Collections
- Remote teaching support
- Starters for ten
- Screen experiments
- Assessment for learning
- Microscale chemistry
- Faces of chemistry
- Classic chemistry experiments
- Nuffield practical collection
- Anecdotes for chemistry teachers
- On this day in chemistry
- Global experiments
- PhET interactive simulations
- Chemistry vignettes
- Context and problem based learning
- Journal of the month
- Chemistry and art
- Art analysis
- Pigments and colours
- Ancient art: today's technology
- Psychology and art theory
- Art and archaeology
- Artists as chemists
- The physics of restoration and conservation
- Ancient Egyptian art
- Ancient Greek art
- Ancient Roman art
- Classic chemistry demonstrations
- In search of solutions
- In search of more solutions
- Creative problem-solving in chemistry
- Solar spark
- Chemistry for non-specialists
- Health and safety in higher education
- Analytical chemistry introductions
- Exhibition chemistry
- Introductory maths for higher education
- Commercial skills for chemists
- Kitchen chemistry
- Journals how to guides
- Chemistry in health
- Chemistry in sport
- Chemistry in your cupboard
- Chocolate chemistry
- Adnoddau addysgu cemeg Cymraeg
- The chemistry of fireworks
- Festive chemistry
- Education in Chemistry
- Teach Chemistry
- On-demand online
- Live online
- Selected PD articles
- PD for primary teachers
- PD for secondary teachers
- What we offer
- Chartered Science Teacher (CSciTeach)
- Teacher mentoring
- UK Chemistry Olympiad
- Who can enter?
- How does it work?
- Resources and past papers
- Top of the Bench
- Schools' Analyst
- Regional support
- Education coordinators
- RSC Yusuf Hamied Inspirational Science Programme
- RSC Education News
- Supporting teacher training
- Interest groups

- More navigation items
Experiments
Try these with your students

Acid–base back titration | 16–18 years
Write balanced equations and calculate reacting masses and moles to find the limiting reagent

‘Gold’ coins on a microscale | 14–16 years
Practical experiment where learners produce ‘gold’ coins by electroplating a copper coin with zinc, includes follow-up worksheet

Toothpaste investigation: neutralisation reactions
Test the basicity of toothpastes and give context to neutralisation reactions

Qualitative tests for organic functional groups | practical videos | 16–18 students
Video and supporting resources to support a practical investigation to identify organic functional groups using a range of qualitative tests

Preparation of an organic liquid | practical videos | 16–18 students
Video and supporting resources to support practical work based on synthesis of an organic liquid, the experiment includes the stages of preparation, separation and purification

Electrochemical cells | practical videos | 16–18 students
Video and supporting resources to support electrochemistry practical work, including two microscale experiments, animation and cell diagrams

Rates of reaction | practical videos | 16–18 students
Video and supporting resources, includes an initial rate (iodine clock reaction) and continuous monitoring method (volume of gas)

Practical potions microscale | 11–14 years
Observe chemical changes in this microscale experiment with a spooky twist.

Microscale neutralisation and precipitation reactions | 11–14 years
Hone your learners’ observation skills with two microscale reactions: neutralising citric acid and creating a lead iodide precipitate

Antibacterial properties of the halogens | 14–18 years
Use this practical to investigate how solutions of the halogens inhibit the growth of bacteria and which is most effective

Equilibrium and Le Chatelier’s principle
Investigate the effects of concentration, pressure and temperature on equilibrium and explore Le Chatelier’s principle in this series of demonstrations.

Microscale technicians in trouble! investigation
Some solutions have been mixed up – help the technicians work out which is which

Rates of hydrolysis – practical videos | 16–18 students
Video resources and associated questions on the topic of hydrolysis of halogenoalkanes.
Redox – practical videos | 16–18 students
Video resources to support the teaching of popular classroom-based redox titrations.

Practical videos | 16–18 years
Videos of key practical techniques and apparatus for revision, flipped learning or remote teaching

Electrolysis of brine
In association with Nuffield Foundation
Use this colourful practical to introduce students to the electrolysis of brine, or sodium chloride solution. Includes kit list and safety instructions.

The equilibrium between two coloured cobalt species
In this demonstration the equilibrium between two different coloured cobalt species is disturbed. Le Chatelier’s principle is used to predict a colour change.

Precipitation reactions of lead nitrate
Compare the colours of various lead compounds to identify which would be good pigments in this microscale practical. Includes kit list and safety instructions.

Some reactions of sulfur dioxide
Observe the reactions of sulfur dioxide with potassium manganate (IV), iodide/iodate mixture and indicator solution. Includes kit list and safety instructions.

The determination of copper in brass
Try this microscale class practical to investigate how much copper there is in brass using nitric acid. Includes kit list and safety instructions.

Microscale reactions of hydrogen sulfide
Observe the reactions of hydrogen sulfide with lead nitrate, silver nitrate and potassium manganate(VII) in this microscale practical. Includes kit list and safety instructions.

Microscale reactions of ammonia
Try this practical to explore the reactions of ammonia with indicator solution, copper(II) sulfate solution and Nessler’s reagent. Includes kit list and safety instructions.

Measuring density
By measuring the relative mass of seawater and tap water, students will be able to discover the density of these liquids. Includes kit list and safety instructions.

The chemistry of thiosulfate ions
Sodium thiosulfate has several interesting reactions with a variety of chemicals. This experiment will let students explore and record these reactions. Includes kit list and safety instructions.
Some reactions of nitrogen dioxide
Using a range of chemicals and solutions, students can create an experiment that will explore some of the reactions of nitrogen dioxide. Includes kit list and safety instructions.
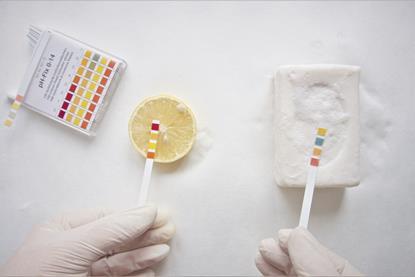
Testing acids and bases on a microscale
Test various substances with indicator solution and look for colour changes in this microscale class practical. Includes kit list and safety instructions.

Mass changes in chemical reactions
Perform two chemical reactions to see whether any mass changes occur in this microscale class practical. Includes kit list and safety instructions.

The oxidation of cyclohexanol by nitric acid
Perform a ring opening oxidation using nitric acid to produce the dicarboxylic acid, 1,6-hexanedioic acid (adipic acid) – and then use the solid crystals that form to determine a melting point. Includes kit list and safety instructions.

Exploring the chemistry of chromium, molybdenum and tungsten
Discover how transition elements differ in aspects of colour, precipitate formation, changes in oxidation state and equilibria. Includes kit list and safety instructions.

Brady’s test for aldehydes and ketones
Identify aldehydes and ketones using Brady’s reagent (2,4-dinitrophenylhydrazine) in this microscale experiment. Includes kit list and safety instructions.
The chemical properties of phenol
Observe and interpret some of the chemical reactions of hydroxybenzene (phenol), by adding five different substances to a Petri dish, and noting down findings. Includes kit list and safety instructions.

Preparing ethyne on a microscale
Generate ethyne gas with calcium carbide and test its properties in this microscale class practical. Includes kit list and safety instructions.

Observing chemical changes
Try this microscale practical to explore the chemical changes in displacement, redox and precipitation reactions. Includes kit list and safety instructions.

Diffusion of gases on a microscale
Try this class practical to explore the diffusion of gases and how relative molecular mass affects rate of diffusion. Includes kit list and safety instructions.

Redox chemistry with dichromate ions
Observe the colour changes that occur with the reduction of dichromate ions by hydrogen peroxide. Includes kit list and safety instructions.

Oxidation states of iron
Compare the two main oxidation states of iron and consider explanations for differences in this microscale practical. Includes kit list and safety instructions.

Microscale reactions of metals with acids
Try this class practical to explore reactivity series with various metals as they react with acids on a microscale. Includes kit list and safety instructions.

Unsaturation test with potassium manganate(VII)
Use a solution of potassium manganate to test for unsaturation in organic compounds in this microscale practical. Includes kit list and safety instructions.

Properties of group 2 elements
Microscale experiment where various anion solutions are added to drops of group 2 element cations. Includes kit list and safety instructions.

Testing for unsaturation with bromine on a microscale
Try this class experiment to prepare elemental bromine and use it to test for unsaturation in organic compounds. Includes kit list and safety instructions.

Oxygen and methylene blue
Reacting hydrogen peroxide, and potassium manganate together will produce detectable oxygen so by using methylene blue solution, and a gas generating apparatus students can test for the presence of oxygen in this practical. Includes kit list and safety instruction.
Synthesis of aspirin on a microscale
Use this class practical to produce aspirin in a microscale esterification reaction using phosphoric acid as a catalyst. Includes kit list and safety instructions.

Energy changes in neutralisation
Study energy changes in two chemical reactions using thermometer strips to measure temperature in this experiment. Includes kit list and safety instructions.

Formation of TCP (2,4,6-trichlorohydroxybenzene)
Delve into preparing TCP by reacting hydroxybenzene (phenol) with chlorine gas, and create this distinctive smelling compound.
Investigating redox reactions on a microscale
Carry out two redox reactions and observe and interpret the results in this microscale class practical. Includes kit list and safety instructions.

The microscale synthesis of indigo dye
Carry out a microscale organic synthesis, the result of which will leave students with indigo dye. Includes kit list and safety instructions.

The treatment of oil spills
Tackle the real-life environment problem of oil spills in your classroom, by creating and then treating a micro version of an oil event. Includes kit list and safety instructions.

The microscale synthesis of azo dyes
Synthesise an azo dye, and use it to change the colour of cotton, with this class experiment. Includes kit list and safety instructions.

Sulfate and carbonate solubility of Groups 1 and 2
Try this microscale practical to explore the properties of elements in Groups 1 and 2 as they form various precipitates. Includes kit list and safety instructions.

Exploring the properties of the carvones
Test the smell of each enantiomer of carvone and detect the differences

Measuring the amount of vitamin C in fruit drinks
Explore ascorbic acid in fruit drinks through titration in this experiment, with specimen results and calculations, stock solutions, and detailed notes included.

Displacement reactions of metals on a microscale
Examine the reactions between various metals and metal salt solutions in this microscale class practical. Includes kit list and safety instructions.

Electrolysis using a microscale Hoffman apparatus
Investigate the electrolysis of sodium sulfate solution using a microscale Hoffman apparatus in this class practical. Includes kit list and safety instructions.

The chemistry of silver
Discover the properties of silver compounds with redox reactions, complex formation and colour/state changes. Includes kit list and safety instructions.

Transition elements and complex compounds microscale experiment | 16–18 years
Try this microscale practical investigating the transition elements, complex formation and change in oxidation state. Includes kit list and safety instructions
Analysis of aspirin tablets on a microscale
Try this microscale class practical to analyse aspirin tablets and find out how much salicylic acid is present. Includes kit list and safety instructions.
The temperature changes induced by evaporation
Explore the rate of evaporation for a trio of liquids, using just a temperature strip, and our worksheet. Includes kit list and safety instructions.

Properties of stereoisomers
By soaking cotton wool in two limonene enantiomers, and adding a stereoisomer, students can explore the differences between each chemical and discuss how they each might react in different conditions. Includes kit list and safety instructions.

Turning copper coins into ‘silver’ and ‘gold’
Perform what looks like alchemy with ordinary copper coins in this teacher demonstration. Includes kit list and safety instructions.

The effect of temperature on solubility
Hot or cold, which water is better for soluble substances? Explore your finding from this practical into the effect of temperature on solubility. Includes kit list and safety instructions.

Particles in motion?
Explore the movement of gas particles in this practical but reacting calcium carbonate with hydrochloric acid. Includes kit list and safety instructions.

The reactivity of the group 2 metals
Compare group 1 and group 2 metals with this practical that shows their reactivity rates, where students can take control of their own observations and come to their own conclusions

Producing a foam
Explore foams and their properties in this experiment, so students learn how foam is produced and produce their own. Includes kit list and safety instructions.

Electricity from chemicals
Use various metals, in pairs, and n electrolyte to form a cell. Then observe the formation of ions around the reactive metal, and compare the speed with which they form around the less reactive metal. Includes kit list and safety instructions.

The electrolysis of solutions
Electricity is passed through various solutions and the products are identified. Includes kit list and safety instructions

The volume of 1 mole of hydrogen gas
Understand the volume of one mole of hydrogen gas through a magnesium and acid reaction, taking note of the temperature and pressure. Includes kit list and safety instructions.

The effect of temperature on reaction rate
Discover more about collision theory in this practical, where a sodium thiosulfate and hydrochloric acid mixture produce an interesting reaction. Includes kit list and safety instructions.

The effect of concentration and temperature on reaction rate
Reaction rate can be altered by many things, in this practical students explore how temperature and concentration effect reaction in an closer look at kinetics. Includes kit list and safety instructions.

Reacting elements with oxygen
Different members of the periodic table will exhibit different reactions when exposed to oxygen, often through heating. This practical supports students to understand the diversity of chemicals and their principles.

Creating an effervescent universal indicator ‘rainbow’
This quick practical uses existing chemicals in your learning space, for students to observe the effervescent reaction that causes universal indicator to create a ‘rainbow’ of colour. Kit list and safety instructions included.

The reaction of magnesium with steam
Plunge a burning magnesium ribbon into the steam above boiling water and allow the hydrogen that is formed to burn – or collect it over water and test it with a lighted spill.

Making a reaction tube
Guide students through this practical to create a reaction tube. Includes kit list and safety instructions.

Properties of the transition metals and their compounds
Student discover the diversity of transition metals in this practical that puts their knowledge of these common elements to the test. Includes kit list and safety instructions.

Disappearing ink
Explore the reaction between acids and bases as students create disappearing ink, in this favourite classroom practical.

Testing salts for anions and cations
A full range of chemicals will guide students into discovering how to identify the composition of unknown substances. Includes kit list and safry instructions.

Rubber band experiment
A rubber band, a hairdryer, and a curious mind will see students discover the principles of heat based reactions. Includes kit list and safety instruction.

A Cartesian diver
An old favourite experiment, the Cartesian diver is easy for students to complete. Explore important ideas that build a foundation of knowledge.

Chemistry and electricity
Create coloured writing from acids, alkali, and salt solution, all activated through electrolysis.

Compare the viscosity of thick and thin liquids in this experiment, which gets young learners exploring how viscosity alters the speed of an air bubble through the substances. Includes kit list and safety instructions.

The oxidation states of vanadium
Introduce your students to the idea that different oxidation states of transition metal ions often have different colours, and that electrode potentials can be used to predict the course of the redox reactions. Includes kit list and safety instructions.

Burning milk powder
Gather a Bunsen burner, and some common powdered milk to help students grasp the ideas of surface area and reaction rates. Includes kit list and safety instructions.

A visible activated complex
A simple demonstration of catalysis also introducing the idea of an activated complex and to allow discussion of the mechanism of catalysis. Includes kit list and safety instructions.

Hydrogen peroxide decomposition using different catalysts
Collect a range of catalysts to explore the decomposition of hydrogen peroxide, paying close attention to the varied reaction rates. Includes kit list and safety instructions.

Competition for oxygen | reacting metals with oxides
Explore the reactions of metals when exposed to the oxide of another metal. When reactions like these occur, the two metals compete for the oxygen. Includes kit list and safety instructions.

Electrolysis of potassium iodide solution
Find out how the electrolysis of a potassium iodide solution works with this practical. Includes kit list, and safety instructions.

An alternative to using compressed gas cylinders
Getting gas under pressure allows exciting demonstrations such as igniting balloons filled with hydrogen gas. Includes kit list and safety instructions.

Flame tests (the wooden splint method)
Find a new method to perform flame tests using wooden splints soaked in chlorides. Includes kit list and safety instructions.

Making nylon: the ‘nylon rope trick’
The ‘nylon rope trick’ is a classic of chemistry classrooms, by mixing decanedioyl dichloride and in cyclohexane you can create a solution that will form nylon strings when floated on an aqueous solution of 1,6-diaminohexane. Kit list and safety instructions included.
Neutralisation circles
Support students to explore neutralisation circles in this experiment that can be performed with common chemistry classroom equipment. Kit list and safety instructions included.

The methane rocket
Ignite methane with oxygen in a bottle, and amaze students with this methane rocket. Contains kit list and safety instructions.
- Contributors
- Email alerts
Site powered by Webvision Cloud

- Sign in / Register
- Administration
- Edit profile

The PhET website does not support your browser. We recommend using the latest version of Chrome, Firefox, Safari, or Edge.

IMAGES
VIDEO
COMMENTS
Explore the wonders of chemical reactions with these fun and interactive activities. From making hot ice and exploding baggies to extracting DNA and creating crystals, you can discover the properties and principles of chemistry in action.
These 10 chemistry experiments will make anyone enjoy science. Chemistry is king when it comes to making science cool. These 10 chemistry experiments will make anyone enjoy science. ... There's even a famous chemical reaction that will emit blue light and a characteristic barking or woofing sound. 01. of 10. Copper and Nitric Acid .
Learn about chemical changes with eight fun and easy activities that show gas formation, heat production, solid creation, color change, and more. Follow the instructions and ask questions to observe and understand the reactions.
Learn about chemistry concepts and have fun with these easy and exciting activities. From colorful milk to giant bubbles, from candy crystals to naked eggs, discover the magic of science with simple supplies.
7. Elephant Toothpaste. This elephant toothpaste experiment is another simple chemical reaction that you can do at home using common household items. All you'll need are an empty bottle (plastic or glass), 6% hydrogen peroxide, dry yeast, dish soap, food coloring, and warm water. Similar to the foaming volcano, this elephant toothpaste ...
Learn about different types of chemical reactions and how to observe and control them with these free resources for K-12 students. Find video lessons, lesson plans, activities, and science projects to explore chemical reactions in action.
Learn about chemical reactions with fun and easy experiments you can do at home. Find out how to make a secret message, a green penny, a rocket boat, a rubber egg, and more.
Make a Hygrometer to Measure Humidity - STEM activity. Hydroponics in a 2-Liter Soda Bottle - STEM activity. Uncover the inner workings of reactions, mixtures, and chemical phenomena through exciting experiments. Explore classic and cutting-edge high school science experiments in this collection of top-quality science investigations.
Mixing baking soda and vinegar is a popular way to see what happens when chemicals react. But, plenty of other experiments demonstrate cool chemical reactions, several of which are easy to produce in a school science lab. The 10 experiments below result in some of the most amazing chemical reactions. The thermite reaction is an example of what ...
These cool chemical reactions show why chemistry experiments and demonstrations are among the most exciting in science. Elephant Toothpaste. Elephant toothpaste is a classic cool chemical reaction between hydrogen peroxide and potassium iodide. The reaction generates a column of steaming foam that looks like giant toothpaste an elephant might use.
Learn how to perform spectacular and educational chemistry demonstrations with this web page. Find out how to make elephant toothpaste, colored fire, thermite, and more with easy instructions and references.
Chemical Reactions. A chemical reaction is a process where two or more substances react together to form a new chemical substance. This might look like a gas formed, cooking or baking, milk souring, etc. Sometimes a physical change occurs, like our popcorn experiment or melting crayons, rather than a chemical change.However, these experiments below are all great examples of chemical change ...
Color change reactions make fantastic chemistry demonstrations. Blend Images - Hill Street Studios/Harmik Nazarian/Getty Images. Of all the chemical reactions, the color change reactions may be the most memorable. The oscillating clock reactions get their name because the colors transition between two or more hues as the conditions change.
TOP 10 chemical reactions that you can repeat at home. Safe and spectacular experiments. 1. The power of bubbles. Materials: teaspoon. Experiment procedure. Pour three teaspoons of dry yeast and two teaspoons of sugar into a bottle. Slowly pour hot water into the bottle.
Find chemistry experiments for kids from preschool to high school that are fun and easy to do at home. Learn about chemical reactions, properties, and applications with hands-on activities, printables, and videos.
An easy way to explain chemical reactions to kids is to use this analogy: Atoms are like letters, molecules are like words. Chemistry is like taking apart words and rearranging the letters to form a new word. Read more about chemical reactions here. Chemical Reactions Projects: 1. Milk Painting 2. Citrus Battery 3. Elephant Toothpaste
For this chemical reaction experiment, take a dark-coated copper coin. Pour ammonia solution over it. You'll see the solution turning blue. When copper comes into contact with ammonia under the influence of oxygen, it forms a complex compound that results in a color change. 6. Fire Experiment.
Color change chemical reactions visually indicate chemical changes in matter. They illustrate pH changes, acid-base reactions, oxidation-reduction, and other key chemistry concepts. Other projects change color due to conformation changes, light emission, and simply mixing colors. There's a color change chemistry project perfect for any occasion.
In this fun and easy science experiment, we're going to explore and investigate endothermic chemical reactions by mixing chalk and vinegar. Materials: Colored chalk Vinegar Hammer Zip close baggie Clear plastic cup Instructions: Put at least two different colors of chalk in the zip close baggie. Use the hammer to carefully smash the chalk into dust. Fill the clear plastic cup about ¾ of the ...
Easy Chemistry Experiments to Do at Home. These 12 projects use materials you probably already have. By. Anne Marie Helmenstine, Ph.D. Ph.D., Biomedical Sciences, University of Tennessee at Knoxville. Dr. Helmenstine holds a Ph.D. in biomedical sciences and is a science writer, educator, and consultant. She has taught science courses at the ...
Support students to explore neutralisation circles in this experiment that can be performed with common chemistry classroom equipment. Kit list and safety instructions included. Ignite methane with oxygen in a bottle, and amaze students with this methane rocket. Contains kit list and safety instructions.
Explore what makes a reaction happen by colliding atoms and molecules. Design experiments with different reactions, concentrations, and temperatures. When are reactions reversible? What affects the rate of a reaction?