The Research Journal Of Biotechnology is a research journal that publishes research related to Biochemistry, Genetics and Molecular Biology; Chemical Engineering; Immunology and Microbiology . This journal is published by the Research Journal of BioTechnology. The ISSN of this journal is 22784535, 09736263 . Based on the Scopus data, the SCImago Journal Rank (SJR) of research journal of biotechnology is 0.138 .
The latest Impact Factor list (JCR) is released in June 2024.
The Impact Factor of Research Journal Of Biotechnology is 0.2.
The impact factor (IF) is a measure of the frequency with which the average article in a journal has been cited in a particular year. It is used to measure the importance or rank of a journal by calculating the times its articles are cited.
The impact factor was devised by Eugene Garfield, the founder of the Institute for Scientific Information (ISI) in Philadelphia. Impact factors began to be calculated yearly starting from 1975 for journals listed in the Journal Citation Reports (JCR). ISI was acquired by Thomson Scientific & Healthcare in 1992, and became known as Thomson ISI. In 2018, Thomson-Reuters spun off and sold ISI to Onex Corporation and Baring Private Equity Asia. They founded a new corporation, Clarivate , which is now the publisher of the JCR.
An indexed journal means that the journal has gone through and passed a review process of certain requirements done by a journal indexer.
The Web of Science Core Collection includes the Science Citation Index Expanded (SCIE), Social Sciences Citation Index (SSCI), Arts & Humanities Citation Index (AHCI), and Emerging Sources Citation Index (ESCI).
Note: ESCI journals donot come with an impact factor. However, ESCI journals are evaluated every year and those who qualified are transferred to SCIE.
The latest impact factor of research journal of biotechnology is 0.2 which is recently updated in June, 2024.
The impact factor (IF) is a measure of the frequency with which the average article in a journal has been cited in a particular year. It is used to measure the importance or rank of a journal by calculating the times it's articles are cited.
Note: Every year, The Clarivate releases the Journal Citation Report (JCR). The JCR provides information about academic journals including impact factor. The latest JCR was released in June, 2023. The JCR 2024 will be released in the June 2024.
The latest Quartile of research journal of biotechnology is Q4 .
Each subject category of journals is divided into four quartiles: Q1, Q2, Q3, Q4. Q1 is occupied by the top 25% of journals in the list; Q2 is occupied by journals in the 25 to 50% group; Q3 is occupied by journals in the 50 to 75% group and Q4 is occupied by journals in the 75 to 100% group.
The publication time may vary depending on factors such as the complexity of the research and the current workload of the editorial team. Journals typically request reviewers to submit their reviews within 3-4 weeks. However, some journals lack mechanisms to enforce this deadline, making it difficult to predict the duration of the peer review process.
The review time also depends upon the quality of the research paper.
Visit to the official website of the journal/ conference to check the details about call for papers.
If your research is related to Biochemistry, Genetics and Molecular Biology; Chemical Engineering; Immunology and Microbiology, then visit the official website of research journal of biotechnology and send your manuscript.
Final summary.
Abbreviation: Res. J. Biotechnol.
Important details.
Research Journal of Biotechnology is a journal published by Research Journal of BioTechnology . This journal covers the area[s] related to Applied Microbiology and Biotechnology, Bioengineering, Biotechnology, etc . The coverage history of this journal is as follows: 2008-2022. The rank of this journal is 22791 . This journal's impact score, h-index, and SJR are 0.30, 20, and 0.138, respectively. The ISSN of this journal is/are as follows: 22784535, 09736263 . The best quartile of Research Journal of Biotechnology is Q4 . This journal has received a total of 256 citations during the last three years (Preceding 2022).
Impact Score 2022 of Research Journal of Biotechnology is 0.30 . If a similar downward trend continues, IS may decrease in 2023 as well.
Check below the impact score trends of research journal of biotechnology. this is based on scopus data..
The h-index of Research Journal of Biotechnology is 20 . By definition of the h-index, this journal has at least 20 published articles with more than 20 citations.
The h-index (also known as the Hirsch index or Hirsh index) is a scientometric parameter used to evaluate the scientific impact of the publications and journals. It is defined as the maximum value of h such that the given Journal has published at least h papers and each has at least h citations.
The International Standard Serial Number (ISSN) of Research Journal of Biotechnology is/are as follows: 22784535, 09736263 .
The ISSN is a unique 8-digit identifier for a specific publication like Magazine or Journal. The ISSN is used in the postal system and in the publishing world to identify the articles that are published in journals, magazines, newsletters, etc. This is the number assigned to your article by the publisher, and it is the one you will use to reference your article within the library catalogues.
ISSN code (also called as "ISSN structure" or "ISSN syntax") can be expressed as follows: NNNN-NNNC Here, N is in the set {0,1,2,3...,9}, a digit character, and C is in {0,1,2,3,...,9,X}
The publisher of Research Journal of Biotechnology is Research Journal of BioTechnology . The publishing house of this journal is located in the India . Its coverage history is as follows: 2008-2022 .
Please check the official website of this journal to find out the complete details and Call For Papers (CFPs).
The International Organization for Standardization 4 (ISO 4) abbreviation of Research Journal of Biotechnology is Res. J. Biotechnol. . ISO 4 is an international standard which defines a uniform and consistent system for the abbreviation of serial publication titles, which are published regularly. The primary use of ISO 4 is to abbreviate or shorten the names of scientific journals using the technique of List of Title Word Abbreviations (LTWA).
As ISO 4 is an international standard, the abbreviation ('Res. J. Biotechnol.') can be used for citing, indexing, abstraction, and referencing purposes.
If your area of research or discipline is related to Applied Microbiology and Biotechnology, Bioengineering, Biotechnology, etc. , please check the journal's official website to understand the complete publication process.
The simplest way to find out the acceptance rate or rejection rate of a Journal/Conference is to check with the journal's/conference's editorial team through emails or through the official website.
What is the impact score of research journal of biotechnology.
The latest impact score of Research Journal of Biotechnology is 0.30. It is computed in the year 2023.
The latest h-index of Research Journal of Biotechnology is 20. It is evaluated in the year 2023.
The latest SCImago Journal Rank (SJR) of Research Journal of Biotechnology is 0.138. It is calculated in the year 2023.
The latest ranking of Research Journal of Biotechnology is 22791. This ranking is among 27955 Journals, Conferences, and Book Series. It is computed in the year 2023.
Research Journal of Biotechnology is published by Research Journal of BioTechnology. The publication country of this journal is India.
This standard abbreviation of Research Journal of Biotechnology is Res. J. Biotechnol..
Research Journal of Biotechnology is a journal published by Research Journal of BioTechnology.
For detailed scope of Research Journal of Biotechnology, check the official website of this journal.
The International Standard Serial Number (ISSN) of Research Journal of Biotechnology is/are as follows: 22784535, 09736263.
The best quartile for Research Journal of Biotechnology is Q4.
The coverage history of Research Journal of Biotechnology is as follows 2008-2022.
Top journals/conferences in bioengineering, top journals/conferences in biotechnology.
Note: The following journal information is for reference only. Please check the journal website for updated information prior to submission.
Discuss science. Find collaborators. Network.
Explore over 38,000 international journals covering a vast array of academic fields.
Researcher.Life is built on Editage's in-depth understanding of what researchers need during publication and beyond, accumulated over 20 years.
Check your submission readiness.
Find out how your manuscript stacks up against 24 technical compliance and 6 language quality checks.
Research journal of biotechnology journal specifications.
| English | Monthly | 2006 | | | Submissions Pack
One subscription packed with expert publication services and AI tools to get you published
Journal Selection Service
Benefit from our experts' recommendations of 3-5 best-suited journals, accompanied by a detailed Journal Selection report explaining the choices. Make informed decisions.
Journal Submission Service
Streamline your submission process with meticulous manuscript formatting to meet journal guidelines, crafting a tailored cover letter for the editor, and hassle-free account creation and submission
Bundle of AI tools for your research needs
Journal Finder, Paperpal, R Discovery, MindTheGraph and more
Buy now for a special Launch price!
Looking for the right journals to submit your mansucript?
Upload your manuscript and get a submission readiness score and other journal recommendations.
RESEARCH JOURNAL OF BIOTECHNOLOGY Scite analysis
795 articles received 121 citations see all
- 3 Supporting
- 115 Mentioning
- 0 Contrasting
RESEARCH JOURNAL OF BIOTECHNOLOGY Editorial notices
- 0 Retractions
- 0 Withdrawals
- 0 Corrections
- 0 Expression of Concern
FAQs on RESEARCH JOURNAL OF BIOTECHNOLOGY
How long has research journal of biotechnology been actively publishing.
RESEARCH JOURNAL OF BIOTECHNOLOGY has been in operation since 2006 till date.
What is the publishing frequency of RESEARCH JOURNAL OF BIOTECHNOLOGY?
RESEARCH JOURNAL OF BIOTECHNOLOGY published with a Monthly frequency.
What is the eISSN & pISSN for RESEARCH JOURNAL OF BIOTECHNOLOGY?
For RESEARCH JOURNAL OF BIOTECHNOLOGY, eISSN is 2278-4535 and pISSN is 2278-4535.
What is the SJR for RESEARCH JOURNAL OF BIOTECHNOLOGY?
SJR for RESEARCH JOURNAL OF BIOTECHNOLOGY is Q4.
Who is the publisher of RESEARCH JOURNAL OF BIOTECHNOLOGY?
RESEARCH JOURNAL BIOTECHNOLOGY is the publisher of RESEARCH JOURNAL OF BIOTECHNOLOGY.
Copyright 2024 Cactus Communications. All rights reserved.


Research Journal of Biotechnology Impact Factor & Key Scientometrics
Research journal of biotechnology overview, impact factor, i. basic journal info, journal issn: 09736263, 22784535, publisher: research journal biotechnology, history: 2008-ongoing, journal hompage: link, how to get published:, research categories, scope/description:, ii. science citation report (scr), research journal of biotechnology scr impact factor, research journal of biotechnology scr journal ranking, research journal of biotechnology scimago sjr rank, research journal of biotechnology scopus 2-year impact factor trend, research journal of biotechnology scopus 3-year impact factor trend, research journal of biotechnology scopus 4-year impact factor trend, research journal of biotechnology impact factor history, iii. other science influence indicators, research journal of biotechnology h-index, research journal of biotechnology h-index history.
International Journal of Biotechnology

IJBT provides an international forum and refereed authoritative source of information in the field of Biotechnology and Biotechnics, with emphasis on management and economics, as well as the political and social issues. It aims to disseminate knowledge, provide a learned reference in the field, and establish channels of communication between academic and research experts, policy makers and executives in industry, commerce and investment institutions.
Topics covered include
- Biotechnology in food
- Biotechnology in healthcare
- Biotechnology in environment
- Biotechnology in agriculture
- Biotechnology in diagnostics
- Biotechnology in therapeutics
- Biotechnology in cosmetics
- Innovation in biotechnology and bio-ethics
- Partnerships, networks and joint ventures
- Biotechnology in the developed and developing world
- Management and economics of biotechnology
- Political and social issues

Journal of Biotechnology

Subject Area and Category
- Biotechnology
- Bioengineering
- Applied Microbiology and Biotechnology
- Medicine (miscellaneous)
Elsevier B.V.
Publication type
01681656, 18734863
Information
How to publish in this journal

The set of journals have been ranked according to their SJR and divided into four equal groups, four quartiles. Q1 (green) comprises the quarter of the journals with the highest values, Q2 (yellow) the second highest values, Q3 (orange) the third highest values and Q4 (red) the lowest values.
| Category | Year | Quartile |
|---|
| Applied Microbiology and Biotechnology | 1999 | Q1 |
| Applied Microbiology and Biotechnology | 2000 | Q2 |
| Applied Microbiology and Biotechnology | 2001 | Q1 |
| Applied Microbiology and Biotechnology | 2002 | Q1 |
| Applied Microbiology and Biotechnology | 2003 | Q1 |
| Applied Microbiology and Biotechnology | 2004 | Q1 |
| Applied Microbiology and Biotechnology | 2005 | Q1 |
| Applied Microbiology and Biotechnology | 2006 | Q1 |
| Applied Microbiology and Biotechnology | 2007 | Q1 |
| Applied Microbiology and Biotechnology | 2008 | Q1 |
| Applied Microbiology and Biotechnology | 2009 | Q1 |
| Applied Microbiology and Biotechnology | 2010 | Q1 |
| Applied Microbiology and Biotechnology | 2011 | Q1 |
| Applied Microbiology and Biotechnology | 2012 | Q1 |
| Applied Microbiology and Biotechnology | 2013 | Q1 |
| Applied Microbiology and Biotechnology | 2014 | Q1 |
| Applied Microbiology and Biotechnology | 2015 | Q1 |
| Applied Microbiology and Biotechnology | 2016 | Q1 |
| Applied Microbiology and Biotechnology | 2017 | Q1 |
| Applied Microbiology and Biotechnology | 2018 | Q1 |
| Applied Microbiology and Biotechnology | 2019 | Q1 |
| Applied Microbiology and Biotechnology | 2020 | Q2 |
| Applied Microbiology and Biotechnology | 2021 | Q2 |
| Applied Microbiology and Biotechnology | 2022 | Q2 |
| Applied Microbiology and Biotechnology | 2023 | Q2 |
| Bioengineering | 2018 | Q2 |
| Bioengineering | 2019 | Q2 |
| Bioengineering | 2020 | Q2 |
| Bioengineering | 2021 | Q2 |
| Bioengineering | 2022 | Q2 |
| Bioengineering | 2023 | Q2 |
| Biotechnology | 1999 | Q2 |
| Biotechnology | 2000 | Q2 |
| Biotechnology | 2001 | Q2 |
| Biotechnology | 2002 | Q2 |
| Biotechnology | 2003 | Q1 |
| Biotechnology | 2004 | Q1 |
| Biotechnology | 2005 | Q1 |
| Biotechnology | 2006 | Q1 |
| Biotechnology | 2007 | Q2 |
| Biotechnology | 2008 | Q1 |
| Biotechnology | 2009 | Q1 |
| Biotechnology | 2010 | Q1 |
| Biotechnology | 2011 | Q1 |
| Biotechnology | 2012 | Q1 |
| Biotechnology | 2013 | Q1 |
| Biotechnology | 2014 | Q1 |
| Biotechnology | 2015 | Q1 |
| Biotechnology | 2016 | Q1 |
| Biotechnology | 2017 | Q2 |
| Biotechnology | 2018 | Q1 |
| Biotechnology | 2019 | Q1 |
| Biotechnology | 2020 | Q2 |
| Biotechnology | 2021 | Q2 |
| Biotechnology | 2022 | Q2 |
| Biotechnology | 2023 | Q2 |
| Medicine (miscellaneous) | 1999 | Q1 |
| Medicine (miscellaneous) | 2000 | Q1 |
| Medicine (miscellaneous) | 2001 | Q1 |
| Medicine (miscellaneous) | 2002 | Q1 |
| Medicine (miscellaneous) | 2003 | Q1 |
| Medicine (miscellaneous) | 2004 | Q1 |
| Medicine (miscellaneous) | 2005 | Q1 |
| Medicine (miscellaneous) | 2006 | Q1 |
| Medicine (miscellaneous) | 2007 | Q1 |
| Medicine (miscellaneous) | 2008 | Q1 |
| Medicine (miscellaneous) | 2009 | Q1 |
| Medicine (miscellaneous) | 2010 | Q1 |
| Medicine (miscellaneous) | 2011 | Q1 |
| Medicine (miscellaneous) | 2012 | Q1 |
| Medicine (miscellaneous) | 2013 | Q1 |
| Medicine (miscellaneous) | 2014 | Q1 |
| Medicine (miscellaneous) | 2015 | Q1 |
| Medicine (miscellaneous) | 2016 | Q1 |
| Medicine (miscellaneous) | 2017 | Q1 |
| Medicine (miscellaneous) | 2018 | Q1 |
| Medicine (miscellaneous) | 2019 | Q1 |
| Medicine (miscellaneous) | 2020 | Q2 |
| Medicine (miscellaneous) | 2021 | Q2 |
| Medicine (miscellaneous) | 2022 | Q2 |
| Medicine (miscellaneous) | 2023 | Q2 |
The SJR is a size-independent prestige indicator that ranks journals by their 'average prestige per article'. It is based on the idea that 'all citations are not created equal'. SJR is a measure of scientific influence of journals that accounts for both the number of citations received by a journal and the importance or prestige of the journals where such citations come from It measures the scientific influence of the average article in a journal, it expresses how central to the global scientific discussion an average article of the journal is.
| Year | SJR |
|---|
| 1999 | 0.841 |
| 2000 | 0.686 |
| 2001 | 0.754 |
| 2002 | 0.786 |
| 2003 | 0.943 |
| 2004 | 1.028 |
| 2005 | 1.193 |
| 2006 | 1.109 |
| 2007 | 1.133 |
| 2008 | 1.147 |
| 2009 | 1.224 |
| 2010 | 1.135 |
| 2011 | 1.165 |
| 2012 | 1.238 |
| 2013 | 1.183 |
| 2014 | 1.116 |
| 2015 | 1.068 |
| 2016 | 1.004 |
| 2017 | 0.929 |
| 2018 | 1.003 |
| 2019 | 0.992 |
| 2020 | 0.901 |
| 2021 | 0.637 |
| 2022 | 0.657 |
| 2023 | 0.741 |
Evolution of the number of published documents. All types of documents are considered, including citable and non citable documents.
| Year | Documents |
|---|
| 1999 | 201 |
| 2000 | 217 |
| 2001 | 180 |
| 2002 | 184 |
| 2003 | 187 |
| 2004 | 223 |
| 2005 | 245 |
| 2006 | 352 |
| 2007 | 400 |
| 2008 | 208 |
| 2009 | 239 |
| 2010 | 257 |
| 2011 | 249 |
| 2012 | 329 |
| 2013 | 317 |
| 2014 | 389 |
| 2015 | 384 |
| 2016 | 402 |
| 2017 | 334 |
| 2018 | 246 |
| 2019 | 222 |
| 2020 | 226 |
| 2021 | 232 |
| 2022 | 176 |
| 2023 | 115 |
This indicator counts the number of citations received by documents from a journal and divides them by the total number of documents published in that journal. The chart shows the evolution of the average number of times documents published in a journal in the past two, three and four years have been cited in the current year. The two years line is equivalent to journal impact factor ™ (Thomson Reuters) metric.
| Cites per document | Year | Value |
|---|
| Cites / Doc. (4 years) | 1999 | 1.689 |
| Cites / Doc. (4 years) | 2000 | 1.545 |
| Cites / Doc. (4 years) | 2001 | 1.845 |
| Cites / Doc. (4 years) | 2002 | 2.037 |
| Cites / Doc. (4 years) | 2003 | 2.472 |
| Cites / Doc. (4 years) | 2004 | 2.630 |
| Cites / Doc. (4 years) | 2005 | 3.273 |
| Cites / Doc. (4 years) | 2006 | 3.248 |
| Cites / Doc. (4 years) | 2007 | 3.331 |
| Cites / Doc. (4 years) | 2008 | 3.116 |
| Cites / Doc. (4 years) | 2009 | 3.350 |
| Cites / Doc. (4 years) | 2010 | 3.550 |
| Cites / Doc. (4 years) | 2011 | 3.486 |
| Cites / Doc. (4 years) | 2012 | 3.733 |
| Cites / Doc. (4 years) | 2013 | 3.589 |
| Cites / Doc. (4 years) | 2014 | 3.521 |
| Cites / Doc. (4 years) | 2015 | 3.267 |
| Cites / Doc. (4 years) | 2016 | 3.255 |
| Cites / Doc. (4 years) | 2017 | 3.047 |
| Cites / Doc. (4 years) | 2018 | 3.248 |
| Cites / Doc. (4 years) | 2019 | 3.600 |
| Cites / Doc. (4 years) | 2020 | 3.816 |
| Cites / Doc. (4 years) | 2021 | 4.293 |
| Cites / Doc. (4 years) | 2022 | 4.221 |
| Cites / Doc. (4 years) | 2023 | 4.228 |
| Cites / Doc. (3 years) | 1999 | 1.689 |
| Cites / Doc. (3 years) | 2000 | 1.621 |
| Cites / Doc. (3 years) | 2001 | 1.741 |
| Cites / Doc. (3 years) | 2002 | 2.104 |
| Cites / Doc. (3 years) | 2003 | 2.456 |
| Cites / Doc. (3 years) | 2004 | 2.666 |
| Cites / Doc. (3 years) | 2005 | 3.182 |
| Cites / Doc. (3 years) | 2006 | 3.200 |
| Cites / Doc. (3 years) | 2007 | 3.157 |
| Cites / Doc. (3 years) | 2008 | 3.032 |
| Cites / Doc. (3 years) | 2009 | 3.345 |
| Cites / Doc. (3 years) | 2010 | 3.289 |
| Cites / Doc. (3 years) | 2011 | 3.276 |
| Cites / Doc. (3 years) | 2012 | 3.860 |
| Cites / Doc. (3 years) | 2013 | 3.501 |
| Cites / Doc. (3 years) | 2014 | 3.383 |
| Cites / Doc. (3 years) | 2015 | 3.214 |
| Cites / Doc. (3 years) | 2016 | 3.089 |
| Cites / Doc. (3 years) | 2017 | 2.848 |
| Cites / Doc. (3 years) | 2018 | 3.285 |
| Cites / Doc. (3 years) | 2019 | 3.710 |
| Cites / Doc. (3 years) | 2020 | 3.789 |
| Cites / Doc. (3 years) | 2021 | 3.928 |
| Cites / Doc. (3 years) | 2022 | 4.206 |
| Cites / Doc. (3 years) | 2023 | 4.599 |
| Cites / Doc. (2 years) | 1999 | 1.513 |
| Cites / Doc. (2 years) | 2000 | 1.377 |
| Cites / Doc. (2 years) | 2001 | 1.720 |
| Cites / Doc. (2 years) | 2002 | 2.118 |
| Cites / Doc. (2 years) | 2003 | 2.459 |
| Cites / Doc. (2 years) | 2004 | 2.526 |
| Cites / Doc. (2 years) | 2005 | 3.002 |
| Cites / Doc. (2 years) | 2006 | 2.893 |
| Cites / Doc. (2 years) | 2007 | 2.896 |
| Cites / Doc. (2 years) | 2008 | 3.009 |
| Cites / Doc. (2 years) | 2009 | 3.035 |
| Cites / Doc. (2 years) | 2010 | 3.036 |
| Cites / Doc. (2 years) | 2011 | 3.274 |
| Cites / Doc. (2 years) | 2012 | 3.458 |
| Cites / Doc. (2 years) | 2013 | 3.256 |
| Cites / Doc. (2 years) | 2014 | 3.263 |
| Cites / Doc. (2 years) | 2015 | 2.952 |
| Cites / Doc. (2 years) | 2016 | 2.820 |
| Cites / Doc. (2 years) | 2017 | 2.767 |
| Cites / Doc. (2 years) | 2018 | 3.258 |
| Cites / Doc. (2 years) | 2019 | 3.771 |
| Cites / Doc. (2 years) | 2020 | 3.147 |
| Cites / Doc. (2 years) | 2021 | 3.650 |
| Cites / Doc. (2 years) | 2022 | 4.463 |
| Cites / Doc. (2 years) | 2023 | 4.490 |
Evolution of the total number of citations and journal's self-citations received by a journal's published documents during the three previous years. Journal Self-citation is defined as the number of citation from a journal citing article to articles published by the same journal.
| Cites | Year | Value |
|---|
| Self Cites | 1999 | 46 |
| Self Cites | 2000 | 53 |
| Self Cites | 2001 | 43 |
| Self Cites | 2002 | 45 |
| Self Cites | 2003 | 68 |
| Self Cites | 2004 | 60 |
| Self Cites | 2005 | 62 |
| Self Cites | 2006 | 129 |
| Self Cites | 2007 | 145 |
| Self Cites | 2008 | 77 |
| Self Cites | 2009 | 127 |
| Self Cites | 2010 | 80 |
| Self Cites | 2011 | 105 |
| Self Cites | 2012 | 89 |
| Self Cites | 2013 | 125 |
| Self Cites | 2014 | 203 |
| Self Cites | 2015 | 113 |
| Self Cites | 2016 | 152 |
| Self Cites | 2017 | 130 |
| Self Cites | 2018 | 85 |
| Self Cites | 2019 | 75 |
| Self Cites | 2020 | 66 |
| Self Cites | 2021 | 56 |
| Self Cites | 2022 | 80 |
| Self Cites | 2023 | 22 |
| Total Cites | 1999 | 934 |
| Total Cites | 2000 | 822 |
| Total Cites | 2001 | 1015 |
| Total Cites | 2002 | 1258 |
| Total Cites | 2003 | 1427 |
| Total Cites | 2004 | 1469 |
| Total Cites | 2005 | 1890 |
| Total Cites | 2006 | 2096 |
| Total Cites | 2007 | 2589 |
| Total Cites | 2008 | 3023 |
| Total Cites | 2009 | 3211 |
| Total Cites | 2010 | 2786 |
| Total Cites | 2011 | 2306 |
| Total Cites | 2012 | 2876 |
| Total Cites | 2013 | 2923 |
| Total Cites | 2014 | 3028 |
| Total Cites | 2015 | 3326 |
| Total Cites | 2016 | 3367 |
| Total Cites | 2017 | 3346 |
| Total Cites | 2018 | 3679 |
| Total Cites | 2019 | 3643 |
| Total Cites | 2020 | 3039 |
| Total Cites | 2021 | 2726 |
| Total Cites | 2022 | 2860 |
| Total Cites | 2023 | 2916 |
Evolution of the number of total citation per document and external citation per document (i.e. journal self-citations removed) received by a journal's published documents during the three previous years. External citations are calculated by subtracting the number of self-citations from the total number of citations received by the journal’s documents.
| Cites | Year | Value |
|---|
| External Cites per document | 1999 | 1.606 |
| External Cites per document | 2000 | 1.517 |
| External Cites per document | 2001 | 1.667 |
| External Cites per document | 2002 | 2.028 |
| External Cites per document | 2003 | 2.339 |
| External Cites per document | 2004 | 2.557 |
| External Cites per document | 2005 | 3.077 |
| External Cites per document | 2006 | 3.003 |
| External Cites per document | 2007 | 2.980 |
| External Cites per document | 2008 | 2.955 |
| External Cites per document | 2009 | 3.213 |
| External Cites per document | 2010 | 3.195 |
| External Cites per document | 2011 | 3.126 |
| External Cites per document | 2012 | 3.741 |
| External Cites per document | 2013 | 3.351 |
| External Cites per document | 2014 | 3.156 |
| External Cites per document | 2015 | 3.104 |
| External Cites per document | 2016 | 2.950 |
| External Cites per document | 2017 | 2.737 |
| External Cites per document | 2018 | 3.209 |
| External Cites per document | 2019 | 3.633 |
| External Cites per document | 2020 | 3.707 |
| External Cites per document | 2021 | 3.847 |
| External Cites per document | 2022 | 4.088 |
| External Cites per document | 2023 | 4.565 |
| Cites per document | 1999 | 1.689 |
| Cites per document | 2000 | 1.621 |
| Cites per document | 2001 | 1.741 |
| Cites per document | 2002 | 2.104 |
| Cites per document | 2003 | 2.456 |
| Cites per document | 2004 | 2.666 |
| Cites per document | 2005 | 3.182 |
| Cites per document | 2006 | 3.200 |
| Cites per document | 2007 | 3.157 |
| Cites per document | 2008 | 3.032 |
| Cites per document | 2009 | 3.345 |
| Cites per document | 2010 | 3.289 |
| Cites per document | 2011 | 3.276 |
| Cites per document | 2012 | 3.860 |
| Cites per document | 2013 | 3.501 |
| Cites per document | 2014 | 3.383 |
| Cites per document | 2015 | 3.214 |
| Cites per document | 2016 | 3.089 |
| Cites per document | 2017 | 2.848 |
| Cites per document | 2018 | 3.285 |
| Cites per document | 2019 | 3.710 |
| Cites per document | 2020 | 3.789 |
| Cites per document | 2021 | 3.928 |
| Cites per document | 2022 | 4.206 |
| Cites per document | 2023 | 4.599 |
International Collaboration accounts for the articles that have been produced by researchers from several countries. The chart shows the ratio of a journal's documents signed by researchers from more than one country; that is including more than one country address.
| Year | International Collaboration |
|---|
| 1999 | 14.43 |
| 2000 | 20.74 |
| 2001 | 23.89 |
| 2002 | 20.65 |
| 2003 | 19.25 |
| 2004 | 22.87 |
| 2005 | 20.41 |
| 2006 | 21.59 |
| 2007 | 15.50 |
| 2008 | 21.63 |
| 2009 | 22.18 |
| 2010 | 22.18 |
| 2011 | 22.89 |
| 2012 | 24.01 |
| 2013 | 22.71 |
| 2014 | 23.91 |
| 2015 | 24.22 |
| 2016 | 21.89 |
| 2017 | 25.45 |
| 2018 | 28.05 |
| 2019 | 22.52 |
| 2020 | 30.97 |
| 2021 | 28.88 |
| 2022 | 27.27 |
| 2023 | 24.35 |
Not every article in a journal is considered primary research and therefore "citable", this chart shows the ratio of a journal's articles including substantial research (research articles, conference papers and reviews) in three year windows vs. those documents other than research articles, reviews and conference papers.
| Documents | Year | Value |
|---|
| Non-citable documents | 1999 | 24 |
| Non-citable documents | 2000 | 6 |
| Non-citable documents | 2001 | 12 |
| Non-citable documents | 2002 | 12 |
| Non-citable documents | 2003 | 12 |
| Non-citable documents | 2004 | 9 |
| Non-citable documents | 2005 | 6 |
| Non-citable documents | 2006 | 4 |
| Non-citable documents | 2007 | 4 |
| Non-citable documents | 2008 | 8 |
| Non-citable documents | 2009 | 12 |
| Non-citable documents | 2010 | 13 |
| Non-citable documents | 2011 | 11 |
| Non-citable documents | 2012 | 12 |
| Non-citable documents | 2013 | 16 |
| Non-citable documents | 2014 | 20 |
| Non-citable documents | 2015 | 23 |
| Non-citable documents | 2016 | 20 |
| Non-citable documents | 2017 | 16 |
| Non-citable documents | 2018 | 13 |
| Non-citable documents | 2019 | 10 |
| Non-citable documents | 2020 | 9 |
| Non-citable documents | 2021 | 4 |
| Non-citable documents | 2022 | 2 |
| Non-citable documents | 2023 | 1 |
| Citable documents | 1999 | 529 |
| Citable documents | 2000 | 501 |
| Citable documents | 2001 | 571 |
| Citable documents | 2002 | 586 |
| Citable documents | 2003 | 569 |
| Citable documents | 2004 | 542 |
| Citable documents | 2005 | 588 |
| Citable documents | 2006 | 651 |
| Citable documents | 2007 | 816 |
| Citable documents | 2008 | 989 |
| Citable documents | 2009 | 948 |
| Citable documents | 2010 | 834 |
| Citable documents | 2011 | 693 |
| Citable documents | 2012 | 733 |
| Citable documents | 2013 | 819 |
| Citable documents | 2014 | 875 |
| Citable documents | 2015 | 1012 |
| Citable documents | 2016 | 1070 |
| Citable documents | 2017 | 1159 |
| Citable documents | 2018 | 1107 |
| Citable documents | 2019 | 972 |
| Citable documents | 2020 | 793 |
| Citable documents | 2021 | 690 |
| Citable documents | 2022 | 678 |
| Citable documents | 2023 | 633 |
Ratio of a journal's items, grouped in three years windows, that have been cited at least once vs. those not cited during the following year.
| Documents | Year | Value |
|---|
| Uncited documents | 1999 | 193 |
| Uncited documents | 2000 | 190 |
| Uncited documents | 2001 | 209 |
| Uncited documents | 2002 | 188 |
| Uncited documents | 2003 | 149 |
| Uncited documents | 2004 | 128 |
| Uncited documents | 2005 | 119 |
| Uncited documents | 2006 | 106 |
| Uncited documents | 2007 | 156 |
| Uncited documents | 2008 | 176 |
| Uncited documents | 2009 | 165 |
| Uncited documents | 2010 | 162 |
| Uncited documents | 2011 | 124 |
| Uncited documents | 2012 | 95 |
| Uncited documents | 2013 | 153 |
| Uncited documents | 2014 | 169 |
| Uncited documents | 2015 | 194 |
| Uncited documents | 2016 | 198 |
| Uncited documents | 2017 | 223 |
| Uncited documents | 2018 | 187 |
| Uncited documents | 2019 | 147 |
| Uncited documents | 2020 | 130 |
| Uncited documents | 2021 | 105 |
| Uncited documents | 2022 | 90 |
| Uncited documents | 2023 | 77 |
| Cited documents | 1999 | 360 |
| Cited documents | 2000 | 317 |
| Cited documents | 2001 | 374 |
| Cited documents | 2002 | 410 |
| Cited documents | 2003 | 432 |
| Cited documents | 2004 | 423 |
| Cited documents | 2005 | 475 |
| Cited documents | 2006 | 549 |
| Cited documents | 2007 | 664 |
| Cited documents | 2008 | 821 |
| Cited documents | 2009 | 795 |
| Cited documents | 2010 | 685 |
| Cited documents | 2011 | 580 |
| Cited documents | 2012 | 650 |
| Cited documents | 2013 | 682 |
| Cited documents | 2014 | 726 |
| Cited documents | 2015 | 841 |
| Cited documents | 2016 | 892 |
| Cited documents | 2017 | 952 |
| Cited documents | 2018 | 933 |
| Cited documents | 2019 | 835 |
| Cited documents | 2020 | 672 |
| Cited documents | 2021 | 589 |
| Cited documents | 2022 | 590 |
| Cited documents | 2023 | 557 |
Evolution of the percentage of female authors.
| Year | Female Percent |
|---|
| 1999 | 27.85 |
| 2000 | 31.46 |
| 2001 | 29.90 |
| 2002 | 30.21 |
| 2003 | 31.76 |
| 2004 | 35.05 |
| 2005 | 35.17 |
| 2006 | 32.88 |
| 2007 | 35.39 |
| 2008 | 32.27 |
| 2009 | 35.75 |
| 2010 | 32.09 |
| 2011 | 34.65 |
| 2012 | 35.02 |
| 2013 | 33.40 |
| 2014 | 37.10 |
| 2015 | 37.94 |
| 2016 | 40.73 |
| 2017 | 37.34 |
| 2018 | 39.06 |
| 2019 | 43.18 |
| 2020 | 38.37 |
| 2021 | 40.46 |
| 2022 | 40.59 |
| 2023 | 41.12 |
Evolution of the number of documents cited by public policy documents according to Overton database.
| Documents | Year | Value |
|---|
| Overton | 1999 | 0 |
| Overton | 2000 | 0 |
| Overton | 2001 | 0 |
| Overton | 2002 | 0 |
| Overton | 2003 | 2 |
| Overton | 2004 | 11 |
| Overton | 2005 | 5 |
| Overton | 2006 | 11 |
| Overton | 2007 | 17 |
| Overton | 2008 | 8 |
| Overton | 2009 | 13 |
| Overton | 2010 | 8 |
| Overton | 2011 | 14 |
| Overton | 2012 | 5 |
| Overton | 2013 | 9 |
| Overton | 2014 | 8 |
| Overton | 2015 | 5 |
| Overton | 2016 | 9 |
| Overton | 2017 | 8 |
| Overton | 2018 | 1 |
| Overton | 2019 | 3 |
| Overton | 2020 | 1 |
| Overton | 2021 | 1 |
| Overton | 2022 | 1 |
| Overton | 2023 | 0 |
Evoution of the number of documents related to Sustainable Development Goals defined by United Nations. Available from 2018 onwards.
| Documents | Year | Value |
|---|
| SDG | 2018 | 54 |
| SDG | 2019 | 63 |
| SDG | 2020 | 53 |
| SDG | 2021 | 61 |
| SDG | 2022 | 71 |
| SDG | 2023 | 33 |
Leave a comment
Name * Required
Email (will not be published) * Required
* Required Cancel
The users of Scimago Journal & Country Rank have the possibility to dialogue through comments linked to a specific journal. The purpose is to have a forum in which general doubts about the processes of publication in the journal, experiences and other issues derived from the publication of papers are resolved. For topics on particular articles, maintain the dialogue through the usual channels with your editor.

Follow us on @ScimagoJR Scimago Lab , Copyright 2007-2024. Data Source: Scopus®

Cookie settings
Cookie Policy
Legal Notice
Privacy Policy

- STM Journals
- Special Issues
- Conferences
- Editorial Board Members
- Reviewers Board Members
- Advisory Panel
- Indexing Bodies
- For Authors
- For Reviewers
- For Editors
- For Advisory Board
- Special Issue Guidelines
- Peer-Review Policy
- Manuscript Submission and Processing
- Publication Ethics and Virtue
- Article Processing Charge
- Editorial Policy
- Advertising Policy
- STM Website and Link Policy
- Distribution and dessemination of Research
- Informed consent Policy
- DOI Payment
"Connect with colleagues and showcase your academic achievements."
"Unleashing the potential of your words"
"Explore a vast collection of books and broaden your horizons."
"Empower yourself with the knowledge and skills needed to succeed."
"Collaborate with like-minded professionals and share your knowledge."
"Learn from experts and engage with a community of learners."


Research & Reviews : A Journal of Biotechnology
ISSN: 2231-3826
Journal Menu
Editor overview.
rrjobt maintains an Editorial Board of practicing researchers from around the world, to ensure manuscripts are handled by editors who are experts in the field of study.

Prof. Manchikatla Venkat Rajam
STM Journals, An imprint of Consortium e-Learning Network Pvt. Ltd.
E-mail: [email protected] Tel: (+91) 0120- 4781 200, (+91) 120 478 1221 Mob: (+91) 981-007-8958, (+91)-966-7725-932
Foliar Application of Zinc Nanoparticles Improves Photosynthetic Performance in Wheat Plants
Import Substitute for Production of Ultrapure Water by Membrane Integrated Process for Medical and Biotechnological Applications
Macro and Micronutrient Interactions with Malnutrition after Nutrition Education Intervention, Specifically with Regard to Nutritional Therapy and Diet Modification
About the Journal
Research & Reviews : A Journal of Biotechnology: 2231-3826(e) is a peer-reviewed hybrid open-access journal launched in 2015 focused on the publication of current research work carried out in the area of Biotechnology. View Full Focus and Scope…
Journal Particulars
[email protected]
+91 120 478 1235
Latest Article
Vol-14 Issue-02 2024
Recycling of Expired Chemical Drugs and Controlling of Organic Chemical Compounds
Nagham Mahmood Aljamali, Shireen Ridha Rasool, Ali Jassim Al-Zuhairi, Noor Saad Jaafer, Marwah Tahseen Neamah Keywords: Expired drugs, Chemical drugs, recycling, Thermal effect on compounds, organic compounds.
Association of Vitamin D binding protein BP rs2282679 Gene polymorphism and Serum Levels of Vitamin D in Patients with Vitiligo
Ali S. Shakir, Alaa T. Hamza Keywords: Vitiligo, Polymorphism, Vitamin D, Genotype, Cholecalciferol
Investigations Into the Potential for Anthracene Decomposition of Novel Soil Bacterium, Pseudomonas Putida P7
Umesh H.R, Kushalatha M Keywords: Anthracene decomposition, strain, pseudomonas, 16S rRNA, phylogenetic analysis.
Exploring the Bioactive Potential of Himalayan Lichens: Phytochemical and Antimicrobial Profiling of Xanthoparmelia tinctina and Pyxine subcinerea Extracts
Priyansh Srivastava Keywords: lichens, secondary metabolites, antibacterial, antioxidant, metabolite profile.
Lifesciences Journal Keywords: Vitiligo, Polymorphism, Vitamin D, Genotype, Cholecalciferol.

Special Issue

Subscribe Email Notofocation
WEBSITE DISCLAIMER
Last updated: 2022-06-15
The information provided by STM Journals (“Company”, “we”, “our”, “us”) on https://journals.stmjournals.com / (the “Site”) is for general informational purposes only. All information on the Site is provided in good faith, however, we make no representation or warranty of any kind, express or implied, regarding the accuracy, adequacy, validity, reliability, availability, or completeness of any information on the Site.
UNDER NO CIRCUMSTANCE SHALL WE HAVE ANY LIABILITY TO YOU FOR ANY LOSS OR DAMAGE OF ANY KIND INCURRED AS A RESULT OF THE USE OF THE SITE OR RELIANCE ON ANY INFORMATION PROVIDED ON THE SITE. YOUR USE OF THE SITE AND YOUR RELIANCE ON ANY INFORMATION ON THE SITE IS SOLELY AT YOUR OWN RISK.
EXTERNAL LINKS DISCLAIMER
The Site may contain (or you may be sent through the Site) links to other websites or content belonging to or originating from third parties or links to websites and features. Such external links are not investigated, monitored, or checked for accuracy, adequacy, validity, reliability, availability, or completeness by us.
WE DO NOT WARRANT, ENDORSE, GUARANTEE, OR ASSUME RESPONSIBILITY FOR THE ACCURACY OR RELIABILITY OF ANY INFORMATION OFFERED BY THIRD-PARTY WEBSITES LINKED THROUGH THE SITE OR ANY WEBSITE OR FEATURE LINKED IN ANY BANNER OR OTHER ADVERTISING. WE WILL NOT BE A PARTY TO OR IN ANY WAY BE RESPONSIBLE FOR MONITORING ANY TRANSACTION BETWEEN YOU AND THIRD-PARTY PROVIDERS OF PRODUCTS OR SERVICES.
PROFESSIONAL DISCLAIMER
The Site can not and does not contain medical advice. The information is provided for general informational and educational purposes only and is not a substitute for professional medical advice. Accordingly, before taking any actions based on such information, we encourage you to consult with the appropriate professionals. We do not provide any kind of medical advice.
Content published on https://journals.stmjournals.com / is intended to be used and must be used for informational purposes only. It is very important to do your analysis before making any decision based on your circumstances. You should take independent medical advice from a professional or independently research and verify any information that you find on our Website and wish to rely upon.
THE USE OR RELIANCE OF ANY INFORMATION CONTAINED ON THIS SITE IS SOLELY AT YOUR OWN RISK.
AFFILIATES DISCLAIMER
The Site may contain links to affiliate websites, and we may receive an affiliate commission for any purchases or actions made by you on the affiliate websites using such links.
TESTIMONIALS DISCLAIMER
The Site may contain testimonials by users of our products and/or services. These testimonials reflect the real-life experiences and opinions of such users. However, the experiences are personal to those particular users, and may not necessarily be representative of all users of our products and/or services. We do not claim, and you should not assume that all users will have the same experiences.
YOUR RESULTS MAY VARY.
The testimonials on the Site are submitted in various forms such as text, audio, and/or video, and are reviewed by us before being posted. They appear on the Site verbatim as given by the users, except for the correction of grammar or typing errors. Some testimonials may have been shortened for the sake of brevity, where the full testimonial contained extraneous information not relevant to the general public.
The views and opinions contained in the testimonials belong solely to the individual user and do not reflect our views and opinions.
ERRORS AND OMISSIONS DISCLAIMER
While we have made every attempt to ensure that the information contained in this site has been obtained from reliable sources, STM Journals is not responsible for any errors or omissions or the results obtained from the use of this information. All information on this site is provided “as is”, with no guarantee of completeness, accuracy, timeliness, or of the results obtained from the use of this information, and without warranty of any kind, express or implied, including, but not limited to warranties of performance, merchantability, and fitness for a particular purpose.
In no event will STM Journals, its related partnerships or corporations, or the partners, agents, or employees thereof be liable to you or anyone else for any decision made or action taken in reliance on the information in this Site or for any consequential, special or similar damages, even if advised of the possibility of such damages.
GUEST CONTRIBUTORS DISCLAIMER
This Site may include content from guest contributors and any views or opinions expressed in such posts are personal and do not represent those of STM Journals or any of its staff or affiliates unless explicitly stated.
LOGOS AND TRADEMARKS DISCLAIMER
All logos and trademarks of third parties referenced on https://journals.stmjournals.com / are the trademarks and logos of their respective owners. Any inclusion of such trademarks or logos does not imply or constitute any approval, endorsement, or sponsorship of STM Journals by such owners.
Should you have any feedback, comments, requests for technical support, or other inquiries, please contact us by email: [email protected] .
Identifiers
Linking ISSN (ISSN-L): 0973-6263
Google https://www.google.com/search?q=ISSN+%220973-6263%22
Bing https://www.bing.com/search?q=ISSN+%220973-6263%22
Yahoo https://search.yahoo.com/search?p=ISSN%20%220973-6263%22
National Library of India http://nationallibraryopac.nvli.in/cgi-bin/koha/opac-search.pl?advsearch=1&idx=ns&q=0973-6263&weight_search=1&do=Search&sort_by=relevance
Resource information
Title proper: Research Journal of Biotechnology.
Country: India
Medium: Print
Record information
Last modification date: 27/05/2024
Type of record: Confirmed
ISSN Center responsible of the record: ISSN National Centre for India For all potential issues concerning the description of the publication identified by this bibliographic record (missing or wrong data etc.), please contact the ISSN National Centre mentioned above by clicking on the link.
downloads requested
Discover all the features of the complete ISSN records
Display mode x.
Labelled view
MARC21 view
UNIMARC view
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Review Article
- Open access
- Published: 17 September 2024
Evidence for widespread human exposure to food contact chemicals
- Birgit Geueke ORCID: orcid.org/0000-0002-0749-3982 1 ,
- Lindsey V. Parkinson ORCID: orcid.org/0000-0002-6219-0546 1 ,
- Ksenia J. Groh ORCID: orcid.org/0000-0002-3778-4721 2 ,
- Christopher D. Kassotis ORCID: orcid.org/0000-0002-0990-2428 3 ,
- Maricel V. Maffini ORCID: orcid.org/0000-0002-3853-9461 4 ,
- Olwenn V. Martin ORCID: orcid.org/0000-0003-2724-7882 5 ,
- Lisa Zimmermann ORCID: orcid.org/0000-0001-6801-6859 1 ,
- Martin Scheringer ORCID: orcid.org/0000-0002-0809-7826 6 , 7 &
- Jane Muncke ORCID: orcid.org/0000-0002-6942-0594 1
Journal of Exposure Science & Environmental Epidemiology ( 2024 ) Cite this article
7605 Accesses
1409 Altmetric
Metrics details
Over 1800 food contact chemicals (FCCs) are known to migrate from food contact articles used to store, process, package, and serve foodstuffs. Many of these FCCs have hazard properties of concern, and still others have never been tested for toxicity. Humans are known to be exposed to FCCs via foods, but the full extent of human exposure to all FCCs is unknown.
To close this important knowledge gap, we conducted a systematic overview of FCCs that have been monitored and detected in human biomonitoring studies according to a previously published protocol.
We first compared the more than 14,000 known FCCs to five biomonitoring programs and three metabolome/exposome databases. In a second step, we prioritized FCCs that have been frequently detected in food contact materials and systematically mapped the available evidence for their presence in humans.
For 25% of the known FCCs (3601), we found evidence for their presence in humans. This includes 194 FCCs from human biomonitoring programs, with 80 of these having hazard properties of high concern. Of the 3528 FCCs included in metabolome/exposome databases, most are from the Blood Exposome Database. We found evidence for the presence in humans for 63 of the 175 prioritized FCCs included in the systematic evidence map, and 59 of the prioritized FCCs lack hazard data.
Significance
Notwithstanding that there are also other sources of exposure for many FCCs, these data will help to prioritize FCCs of concern by linking information on migration and biomonitoring. Our results on FCCs monitored in humans are available as an interactive dashboard (FCChumon) to enable policymakers, public health researchers, and food industry decision-makers to make food contact materials and articles safer, reduce human exposure to hazardous FCCs and improve public health.
Impact statement
We present systematically compiled evidence on human exposure to 3601 food contact chemicals (FCCs) and highlight FCCs that are of concern because of their known hazard properties. Further, we identify relevant data gaps for FCCs found in food contact materials and foods. This article improves the understanding of food contact materials’ contribution to chemical exposure for the human population and highlights opportunities for improving public health.

Similar content being viewed by others
Aspartame exposures in the US population: Demonstration of a novel approach for exposure estimates to food additives using NHANES data
Phthalate and novel plasticizer concentrations in food items from U.S. fast food chains: a preliminary analysis
A prospective whole-mixture approach to assess risk of the food and chemical exposome
Introduction.
Humans are exposed to synthetic chemicals from food, drugs, household and personal care products, and environmental pollutants. Some of these chemicals have been associated with the increasing prevalence of non-communicable diseases [ 1 , 2 , 3 ]. Food packaging and other food contact articles (FCAs), such as tableware and food processing equipment, contribute to the human chemical burden via oral exposure, because food contact chemicals (FCCs) migrate from different food contact materials (FCMs) into foodstuffs and are then ingested [ 4 , 5 , 6 , 7 , 8 ].
For individual FCCs, such as bisphenol A (BPA) and several phthalates, the contribution of chemical migration from FCMs to human exposure has been studied in detail, taking into account that other exposure sources exist [ 9 , 10 , 11 , 12 ]. BPA is banned in some food contact applications, such as baby bottles, in many parts of the world, but is still regularly measured in FCMs (e.g [ 13 , 14 , 15 ].). Currently, a complete ban on BPA in FCMs is proposed by the European Commission [ 16 ]. However, hundreds of FCCs have been shown to migrate from FCMs into foods, and thousands of FCCs have been extracted from FCMs [ 5 ]. In total, over 12,000 FCCs could be intentionally used during the manufacturing of different types of FCMs [ 17 ] and even more chemicals could be present in FCMs as non-intentionally added substances (NIAS) that are introduced or formed during manufacture or use [ 5 , 18 , 19 ].
Many FCCs are of concern for human health because they have hazard properties such as carcinogenicity, mutagenicity, and reprotoxicity (CMR), endocrine disrupting properties, bioaccumulation potential, and/or persistence [ 17 , 20 , 21 ]. In addition, toxicity data are often incomplete or missing, which means that safe use cannot be assessed [ 17 , 22 , 23 ]. Therefore, reducing exposure to known hazardous FCCs and assessing untested FCCs can contribute to the prevention of non-communicable diseases that are associated with chemical exposures [ 24 , 25 ].
The challenges in regulating FCMs and managing the health risks associated with FCCs are diverse and legislation often does not keep up with the latest scientific understanding [ 26 , 27 ]. Publicly available evidence on intentionally used FCCs and their known hazards is available in our earlier work where we compiled the Food Contact Chemicals Database (FCCdb) [ 17 ]. The FCCdb gives an overview of all chemicals that are known to be used in the manufacture of FCMs. Further, we systematically mapped data on migrating and extractable FCCs, and our Database on Migrating and Extractable Food Contact Chemicals (FCCmigex) provides evidence for FCCs that have been detected in extracts of FCMs and/or their migrates into food and food simulants, indicating the potential for human exposure [ 5 ]. Only 30% of the chemicals present in FCMs are listed in the FCCdb, based on information from the most recent update of the FCCmigex database [ 28 ]. This indicates that the non-listed FCCs are either NIAS or have been intentionally used although they are not recorded in any of the FCCdb’s sources. Even though it is well-established that chemicals migrating from FCMs contribute to human exposure, the presence of FCCs in human samples has not yet been systematically assessed.
Here, we provide a systematic overview of FCCs that have been monitored and detected in humans by including information from biomonitoring programs, metabolome and exposome databases, and the primary scientific literature. We detailed our approach in a previously published protocol [ 29 ]. The resulting Database on Food Contact Chemicals Monitored in Humans (FCChumon) is a publicly available tool integrating empirical data on FCCs in human samples, and it complements the FCCdb and FCCmigex databases. Our goal is to provide scientific evidence that supports advancing global FCM regulations and the safety assessments of FCCs.
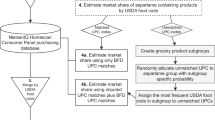
Overview of the two-step approach
The evidence for FCCs that have been monitored and detected in human samples was compiled according to a protocol initially registered on Zenodo in September 2022 and updated in April 2023 [ 29 ]. We followed the structure of a Population-Outcome (PO) question:
Question: Which known FCCs have been monitored in the human body?
Population (P): Human samples, such as blood, urine, hair, and breast milk, from people of any age, gender, or ethnicity
Outcome (O): Any result describing the monitoring/detection of a known FCC or its metabolite
As detailed in the protocol and further specified below, we applied a stepwise approach and referred to biomonitoring programs, databases on the human exposome and metabolome, and the primary scientific literature to map the evidence for FCCs’ presence in humans. Briefly, in step 1, FCCs included in the FCCdb and the FCCmigex databases were matched to the chemicals listed in biomonitoring programs and metabolome and exposome databases (Fig. 1 ). During protocol development, we found that thousands of FCCs were neither included in the selected metabolome/exposome databases nor in biomonitoring programs, while the primary scientific literature reported the monitoring of some of these FCCs in human samples. In step 2, we therefore applied the methodology of a systematic evidence map to obtain relevant information from the scientific literature. FCCs not found in any of the sources consulted in step 1 were prioritized based on their presence in FCMs, according to evidence from FCCmigex. These prioritized FCCs were included in the systematic evidence mapping performed in step 2 to understand their presence in human samples.

We compared known FCCs to biomonitoring programs and metabolome/exposome databases (step 1) and systematically mapped the evidence for presence of additional, priority FCCs in humans (step 2). The results of steps 1 and step 2 (red boxes) comprise the Database of Food Contact Chemicals Monitored in Humans (FCChumon).
Information sources for chemical comparisons
Together, the FCCdb and the FCCmigex databases presently consist of 14,402 known FCCs with assigned CAS Registry Numbers (Fig. 1 ). The FCCdb is an inventory for FCCs that are potentially used in the manufacture of FCMs and FCAs [ 17 ]. It currently contains 12,285 distinct FCCs of which 11,593 have a CAS Registry Number. The FCCmigex database systematically maps scientific evidence of FCCs that have been measured in FCMs and FCAs [ 5 , 28 ]. The most recent version of the FCCmigex database contains 4262 chemicals with a CAS Registry Number, of which 3995 FCCs have been detected at least once in an FCM migrate or extract. Each FCCmigex database entry is linked to the reference from which it was generated and provides information about the FCC, what type of FCA and which FCM(s) were tested, details about the experimental set-up, and whether the FCC was detected or not. Chemicals that have been targeted but never detected in FCMs, and that are not in the FCCdb, are not included in this study.
In the first step, we consulted five biomonitoring programs that encompass different ranges of chemicals and provide wide geographic coverage, namely the National Health and Nutrition Examination Survey (NHANES) of the US [ 30 ], the Canadian Health Measures Survey (CHMS) [ 31 ], the Human Biomonitoring for Europe project (HBM4EU) [ 32 , 33 ], the Korean National Environmental Health Survey (KoNEHS) [ 34 ], and Biomonitoring California [ 35 ]. Further, three metabolome/exposome databases were used to identify FCCs that have been monitored in humans: the Human Metabolome Database (HMDB) [ 36 , 37 ]; the Blood Exposome Database [ 38 , 39 ], and the Exposome Explorer [ 40 , 41 ]. In addition to these sources, in the second step we systematically searched the primary scientific literature for human biomonitoring data on specific FCCs, using bibliographic databases (PubMed, Web of Science Core Collection (WoS), ScienceDirect, and CAS SciFinder n ).
Data processing and comparisons (step 1)
All known FCCs with CAS Registry Numbers were included in the comparisons of step 1, regardless of whether the CAS Registry Number indicates a specific structure or a chemical mixture. If available, additional chemical identifiers, such as INChI Keys and SMILES, were retrieved from the collections of FCCs associated with lists S77 and S112 from the NORMAN Suspect List Exchange [ 42 , 43 , 44 ].
In step 1A, information on chemicals that are part of any of the biomonitoring programs was downloaded from the respective sources. We also collected information on whether a chemical has been ‘monitored but never detected’ or ‘monitored and detected’. If it was stated in the biomonitoring programs that the analyte was a metabolite of a specific parent compound, we paired the metabolite and the parent compound for comparison with the known FCCs. For example, the analyte mono-ethyl phthalate (CAS 863029-89-4) is listed as a metabolite of di-ethyl phthalate (CAS 84-66-2) in NHANES, and we used both CAS Registry Numbers in the comparisons to the known FCCs. In this way, we ensured that FCCs were identified in the biomonitoring programs regardless of whether detection in human samples was reported for parent compounds or their metabolites. We manually added CAS Registry Numbers to chemicals missing these identifiers in the biomonitoring lists to enable their comparisons to the FCCs.
In step 1B, the data set ‘biomarkers’ was downloaded from the Exposome Explorer, and the full content of the Blood Exposome Database was retrieved. From the HMDB, all chemicals were included that were labeled by metabolite status as ‘detected and quantified’, ‘detected but not quantified’, and ‘expected but not quantified’. The metabolome/exposome databases do not systematically report links between parent compounds and metabolites. We used these chemical lists from the metabolome/exposome databases without any further editing.
Based on their CAS Registry Numbers, InChI Keys, or SMILES identifiers, FCCs were then compared to the chemical lists retrieved from the biomonitoring programs and metabolome/exposome databases. These comparisons were performed by means of Python (v3.10.8) pandas package (v1.5.3).
Systematic evidence mapping (step 2)
Prioritization and grouping of fccs.
In step 2, we focused on the FCCs that were not found in any of the sources of step 1, i.e., all FCCs, or their metabolites, that have never been included in a biomonitoring program (regardless of whether they have been detected or not) and all FCCs that did not generate any match in the metabolome/exposome databases. These FCCs not monitored in any of the sources of step 1 were candidates for the systematic evidence mapping in step 2. For this step, we prioritized FCCs that have at least five database entries in the FCCmigex, reporting their detection in migrates and/or extracts of FCMs. To verify the absence of any prioritized chemicals in step 1, we also searched the HMDB for the chemical names that are used in the FCCmigex database and in Norman SLE.
For further data analysis and interpretation, prioritized FCCs were assigned to chemical groups based on functional categories and/or chemical structures. During grouping, we referred to the primary literature included in this systematic evidence map and in the FCCmigex database to understand the function and/or chemical features of an FCC. Additionally, we used the tool Classyfire [ 45 ], the Plastics Additives Handbook [ 46 ], and expert knowledge to group FCCs based on their applications in FCMs and/or chemical features, such as functional groups and structural properties.
Literature searches and screening
For each of the prioritized FCCs, individual literature searches were performed. For PubMed, WoS, and ScienceDirect, search strategies included the chemical name as used in the FCCdb or the FCCmigex, and generic search terms related to human biomonitoring (e.g., human, blood, urine, biomonitoring) that were connected by the Boolean operator OR. Searches in CAS SciFinder n used CAS Registry Numbers instead of chemical names. Search strings and settings were slightly adapted depending on the requirements of each database. The searches were not restricted by publication date or language and included all literature published by February 2023. Full details on search strings, applied filters, and settings have been published previously [ 29 ].
Individual literature searches were stored in separate Endnote files, from which duplicates were removed. All individual libraries were uploaded into the online evidence synthesis tool Cadima [ 47 ], where further duplicates were deleted. The references were then screened in a two-level process, beginning with title-and-abstract screening and followed by full-text screening. During the screening, the eligibility criteria specified in the protocol were applied to all prioritized FCCs that were analyzed in the respective reference [ 29 ]. In brief, studies were considered eligible and included in the systematic evidence map if the analyzed sample originated from a human specimen (e.g., urine, blood, and breast milk) and at least one prioritized FCC was analyzed. Ten percent of the references were independently screened by two reviewers in parallel at title-and-abstract and full-text levels, and disagreements were resolved bilaterally. Reasons for exclusion were recorded during full-text screening.
Data extraction
Eligible studies were used to collect information on whether FCCs have been monitored in human samples and if they have been detected. Details on the sample type and analytical approaches were part of the data extraction process (see Supplementary Information). The process was based on the data extraction software tool SciExtract [ 5 ] which allowed us to use precoded options to systematically compile the data. SciExtract was also used to organize and manage the workflow and to store the extracted data.
Hazard mapping
For FCCs included in the biomonitoring programs (step 1A) and those prioritized in step 2, we compiled the hazard properties according to human-health-related criteria described in the EU’s Chemicals Strategy for Sustainability (CSS) [ 48 ]. The CSS seeks to ban the most harmful chemicals from consumer products, including FCMs, and defines chemicals as ‘most harmful’ to human health if they are carcinogenic, mutagenic or toxic to reproduction (CMR) or exhibit specific target organ toxicity (STOT). Hazards associated with endocrine-disrupting properties, persistence, bioaccumulation, and mobility of a chemical are also mentioned in the CSS but were not included in this analysis. We consulted the European Chemicals Agency’s (ECHA) Classification and Labelling Inventory aligned with the Globally Harmonized System (GHS) for chemical classification and labeling [ 49 ] and referred to GHS-aligned classifications by the Japanese Government [ 50 ] for identifying human health-related hazards. Following the GHS criteria for classification and labeling, we identified chemicals as ‘high concern’ if they exhibit CMR properties belonging to categories 1A and 1B (known and presumed CMR, respectively) and/or have been classified as STOT category 1 after repeated exposure (RE) (Fig. S1 ). Chemicals of ‘medium concern’ were those suspected to have CMR and/or STOT RE properties, as indicated by their classifications in category 2. Chemicals that have been classified based on other concerns, such as aquatic toxicity or skin sensitization, were marked as ‘other concern’. FCCs with data in at least one hazard category and without any classification were labeled as ‘not classified’. FCCs that were not included in the hazard inventories, or for which no data were available in any hazard category, were labelled with ‘no hazard data’.
Overall evidence for the presence of FCCs in humans
For a total of 3601 (or 25%) of the 14,402 known FCCs, we found evidence for their presence in human samples (Fig. 2 ). Of these, 194 FCCs have been detected in biomonitoring programs, and 3528 FCCs are listed in metabolome/exposome databases, with an overlap of 184 FCCs found in both types of sources. The total of 3601 FCCs also includes 63 out of 175 prioritized FCCs that have been detected in humans according to the results of the systematic evidence map (step 2).

Schematic representation of the FCCs monitored and detected in biomonitoring programs and/or listed in metabolome/exposome databases (step 1) and additional FCCs detected in humans, based on evidence from the scientific literature for a set of prioritized FCCs (step 2).
Based on the results of this stepwise approach, we set up the FCChumon database, which is provided as an interactive tool that is freely available, searchable, and linking to the relevant sources ( https://www.foodpackagingforum.org/fcchumon ).
Analysis of biomonitoring programs and metabolome/exposome databases
In step 1, we identified 3538 FCCs that have been detected in humans, which can be divided into 1883, 863, and 792 FCCs that are included only in the FCCdb, only in the FCCmigex, and in both databases, respectively (Fig. 3 , lower panel). These numbers indicate that 23% of the FCCs in the FCCdb and 41% of the FCCs in the FCCmigex are listed in at least one of the sources in Step 1. Sixty-seven percent of FCCs that are listed in both FCC databases have evidence of presence in humans.

The upper panel illustrates the FCCs from the FCCdb (green outline), the FCCmigex (yellow outline), and their overlap. The left part of the middle panel shows the number of known FCCs that have been detected in biomonitoring programs and, in brackets, the total number of monitored FCCs. The right part of the middle panel displays the FCCs that are listed in metabolome/exposome databases. FCCs that have been detected in humans are indicated by the orange filling of the respective areas; white areas represent FCCs without any evidence of the presence in humans and the FCCs that have been monitored but not detected. The figure in the lower panel is the result of the overall comparison of the known FCCs with all sources of step 1.
Of the 265 FCCs monitored in at least one of the five biomonitoring programs, 194 FCCs (or their metabolites) have been detected in human samples, and 71 FCCs (or their metabolites) have been monitored but not detected in any of the biomonitoring programs (Fig. 3 , middle panel; Table S1 ). The most extensive national program, NHANES, has monitored over 400 different chemicals in human samples since 1999, and 154 of these are FCCs (Figure S2). We also found 84, 66, 66, and 25 FCCs with evidence for the presence in humans in the biomonitoring programs CHMS, HBM4EU, Biomonitoring California, and KoNEHS, respectively. One hundred and twenty-four FCCs have only been monitored in a single biomonitoring program, and 55 of these have not been detected, whereas 13 FCCs have been included across all five programs, of which 8 have been detected in all programs (Figure S3; Table S1 ).
The overlap of known FCCs with metabolome/exposome databases is much larger than the overlap with biomonitoring programs: of the three metabolome/exposome databases, the Blood Exposome Database includes the highest number of FCCs (2918 FCCs), followed by the HMDB (2211 FCCs) and the Exposome Explorer (253 FCCs) (Fig. 3 , middle panel; Figure S4). The HMDB lists 367, 1072, and 772 FCCs that are labelled as “detected and quantified”, “detected but not quantified”, and “expected but not quantified”, respectively, according to the classification system of the database (Figure S5) [ 36 ].
Sixty-one out of the 71 FCCs that have been monitored but not detected in biomonitoring programs are listed in at least one of the metabolome/exposome databases. This means that only 10 FCCs fall under the category “monitored but not detected” in step 1 (Fig. 2 ).
Systematic evidence mapping of prioritized FCCs
In step 1 we show that 75% of the known FCCs are not listed in any of the biomonitoring programs or metabolome/exposome databases. However, for some of these FCCs, scoping searches resulted in additional evidence from the primary literature. Therefore, we decided to systematically map the evidence for 175 FCCs which we prioritized based on the number of FCCmigex database entries that report their detection in FCMs.
In this systematic approach, we found 3152 scientific studies for 147 out of the 175 prioritized FCCs (Figure S6) and considered 251 and 159 studies eligible after title-and abstract and full-text screening, respectively. These studies refer to 68 FCCs – for the other 107 FCCs, no studies fulfilled the eligibility criteria.
Of the 68 FCCs for which scientific studies were found, 63 have been detected in human samples and five have been monitored, but not detected, i.e., Irganox 1330 (CAS 1709-70-2), 2,6-(1,1-dimethylethyl)phenol (CAS 128-39-2), phenyl-bis-(2,4,6-trimethylbenzoyl) phosphinoxid (CAS 162881-26-7), 2,5-bis(5-tert-butyl-2-benzoxazolyl) thiophene (CAS 7128-64-5), and Tinuvin 622 (CAS 65447-77-0) (Fig. 4A ). The detected chemicals have been detected in urine (28 FCCs), serum (20), blood (13), and plasma (12) (Fig. 4B ). FCCs have also been found in breast milk (13) and samples taken from umbilical cords (18) and placentas (6). One hundred and thirteen studies have used targeted analyses, whereas 47 studies have used non-targeted approaches (Fig. 4C ), and only one study has applied both methods [ 51 ]. The vast majority of FCCs have been detected directly, i.e. as parent compounds, in human samples (Fig. 4D ), while antioxidant 1098 (CAS 23128-74-7) and Irganox 1035 (CAS 4148-35-9) have been putatively identified based on an unspecific common metabolite in one study [ 52 ].

A Numbers of FCCs with and without evidence from the primary scientific literature indicating their presence in humans. B Types of human samples in which the 63 FCCs have been detected (multiple sample types possible). C Types of applied analytical methods per study and per detected FCC. D Numbers of FCCs that have been analyzed directly (as parent compound) or as specific or unspecific metabolite.
FCCs monitored in humans
Fccs detected in biomonitoring programs.
Among the 235 FCCs present in FCMs that have been included in human biomonitoring programs, there are 51 volatile organic compounds (VOCs), 29 per- and polyfluoroalkyl substances (PFAS), 25 pesticides, 23 metals, 23 dioxin-like compounds, 20 flame retardants, and 19 phthalates and their alternatives (Fig. 5A , right panel; Table S1 ). Phthalates and alternative plasticizers, and metals are frequently detected FCCs in FCMs and have also been often found in humans (Fig. 5A , bar charts). Furthermore, PFAS, VOCs, and phenolic compounds, including bisphenols, parabens, and benzophenones, have been frequently monitored and detected in FCMs and in humans. In contrast, for dioxin-like compounds, pesticides, flame retardants, polyaromatic hydrocarbons (PAHs), amines, and perchlorate there is less evidence for their presence in FCMs. Interestingly, 71 of the 95 FCCs belonging to these six groups would not be expected to be present in FCMs, since they are not included in the FCCdb (Table S1 ). The evidence for presence of FCCs in FCMs varies widely between but also within chemical groups. For example, the VOC styrene (CAS 100-42-5) has been listed 99 times as “detected in FCMs” in the FCCmigex database, while 16 other VOCs found in humans have been listed less than ten times each (Table S1 ). The presence of styrene, or its metabolites, in humans has been shown by NHANES, CHMS, and KoNEHS, but there is no evidence for 18 of the 51 VOCs from any of the five biomonitoring programs.

A 235 FCCs detected in FCMs and included in biomonitoring programs (step 1A). B 175 FCCs prioritized based on their detection in FCMs and their absence in step 1 (step 2). The yellow bar charts illustrate the evidence for the presence of FCC groups in FCMs, based on the sum of database entries from the FCCmigex that report the detection of FCCs in FCMs. The orange bar charts show the evidence of the presence of FCC groups in humans. In step 1A, this is based on the number of biomonitoring programs that have monitored individual FCCs in humans and the addition of these counts by group. In step 2, the orange bars represent the number of studies that have monitored at least one FCC of the respective group. The pie charts show how many FCCs per group have been monitored and detected at least once and how many FCCs have been monitored but not detected in any sample. For step 2, the pie charts also include the chemicals for which there is no evidence in the scientific literature.
FCCs included in the systematic evidence map
Among the 175 FCCs included in the systematic evidence map, there are 38 oligomers (mainly siloxane, polyamide, and polyethylene terephthalate (PET) derivatives), 15 antioxidants and degradation products, 14 photoinitiators, and 14 plasticizers (Fig. 5B , right panel; Table S2 ).
For oligomers and antioxidants and their degradation products, 424 and 499 FCCmigex database entries, respectively, imply that FCMs play a role in human exposure to these chemical groups (Fig. 5B ). However, there is limited evidence for the presence of antioxidants and oligomers in humans, as indicated by 6 and 12 studies, respectively, reporting the detection of the chemicals of these groups. For only five out of 38 prioritized oligomers, we found evidence for their detection in humans: a PET cyclic trimer (CAS 7441-32-9), three cyclic siloxanes (D7, CAS 107-50-6; D8, CAS 556-68-3 and D9, CAS 556-71-8), and 1,6-dioxacyclododecane-7,12-dione (CAS 777-95-7) (Table S2 ). With 209 FCCmigex database entries and 9 studies reporting detection in humans, photoinitiators are regularly found in FCMs, but less frequently monitored in humans. For the five BADGE derivatives BADGE·H 2 O, BADGE·2H 2 O, BADGE·HCl, BADGE·2HCl, and BADGE·H 2 O·HCl, 23 studies confirm the detection of at least one of these FCCs in humans. In addition, they have 65 database entries in the FCCmigex, confirming their regular detection in migrates and/or extracts from coated metal FCMs.
FCCs of concern
Of the 235 FCCs included in biomonitoring studies and with evidence for their presence in FCMs, 100 FCCs have hazard properties of high concern for human health, and 44 FCCs have hazard properties of medium concern, i.e., they are assigned to categories 1 and 2, respectively (Fig. 6A , Table S1 ). Among the FCCs detected in humans are several category 1 A and 1B carcinogens, of which, e.g., styrene, benzophenone (CAS 119-61-9), formaldehyde (CAS 50-00-0), and cadmium (CAS 7440-43-9) have also been frequently found in FCMs. Dozens of FCCs are classified as toxic to reproduction, for example, nine phthalates, which are all classified as 1B reprotoxicants. Over 30 FCCs are mutagens (e.g., benzene (CAS 71-43-2), lead, cadmium, and cobalt), and many more exhibit specific target organ toxicity after repeated exposure (e.g., 4,4’-methylenedianiline (CAS 101-77-9) and perfluorooctanoic acid (CAS 335-67-1)). Seventy-seven FCCs have other concerns or have not been classified as hazardous based on the available data, and 14 do not have hazard data or are not listed.

A 235 FCCs detected in FCMs and included in biomonitoring programs. B 175 FCCs prioritized based on their detection in FCMs and their absence in step 1. On the left side of both Sankey diagrams, the number of FCCs monitored and detected in humans (red), monitored but not detected in humans (light gray), and without any evidence for the presence in humans (dark gray) are shown. On the right sides, the diagrams visualize the number of chemicals of high (red) and medium concern (yellow), chemicals of other concerns or not classified chemicals (light gray), and chemicals with no hazard data (dark gray). The thickness of connecting lines represents the numbers of chemicals that belong to a hazard category and their evidence for presence in humans. *Many hazard classifications lack information for specific hazard categories. This means that chemicals may be newly categorized or reassigned to other hazard categories when more information becomes available in the future.
Among the 175 FCCs included in the systematic evidence map, 5 and 13 FCCs are classified in categories 1 and 2, respectively, resulting in high and medium concern for CMR and/or STOT RE properties (Fig. 6B , Table S2 ). Di-n-octylisophthalate (CAS 137-89-3), 2-benzyl-2-(dimethylamino)-4-morpholino-butyrophenone (CAS 119313-12-1), ethyl-4-dimethylaminobenzoate (CAS 10287-53-3), and medium-chain chlorinated paraffins (CAS 85535-85-9) are reproductive toxicants of high concern (category 1B) and have been detected in FCMs and in humans. For the category 1B carcinogen 2,4’-methylenedianiline (CAS 1208-52-2), however, we found no evidence concerning its presence in humans. Ninety-eight FCCs are allocated to other hazard categories or have not been classified, and 59 FCCs are not listed in the hazard inventories, indicating a lack of data for these chemicals. Based on this evidence map, 49 FCCs without hazard data have also never been targeted in human samples, but they are known to migrate so the implications of the probable human exposure from these FCCs are unknown. Among these are 29 oligomers that have been mainly detected in PA, PET, and siloxane FCMs.
Relevance of this study
There is evidence of human exposure for at least 3601 (or 25%) of the known FCCs (Fig. 1 ). While other exposure sources (than FCMs) exist for FCCs, it is likely that humans are exposed to more FCCs than reported here, as we only searched the scientific literature for a small subset of chemicals. The novel database on FCCs monitored in humans (FCChumon) lends itself to integration with our previously published database of chemicals present in/migrating from specific FCMs (FCCmigex) [ 5 ], thereby enabling hypothesis-driven research for closing pertinent knowledge gaps on human exposure to chemicals originating from FCMs. Together, these databases can also be used as information sources for elucidating FCCs’ health impacts and highlighting other priority research needs.
Parent compounds vs. metabolites
For the exposure assessment of chemicals with well-known metabolic fate in humans, such as phthalates and certain VOCs, metabolites instead of their parent compounds are monitored [ 53 , 54 ]. We considered this aspect when comparing FCCs to chemicals from the biomonitoring programs and when analyzing the primary literature. Various tools could support identifying FCC metabolites by predicting chemical biotransformation [ 55 , 56 ], but they are associated with large scientific uncertainty, as shown, e.g., for the metabolism of agrochemicals in rats [ 57 ] or for 15 structurally different groups of flame retardants [ 58 ]. Given the high number of FCCs included in this study, we did not attempt to systematically predict potential metabolites and only considered information on specific metabolites if it was readily available in the biomonitoring programs. Only one unspecific metabolite was identified in the systematic evidence map, indicating potential exposure to two antioxidants [ 52 ].
Focus on chemical groups
FCMs are a well-known and relevant exposure source for phthalates and their alternatives, metals, VOCs, and phenolic compounds. These chemicals are regularly monitored and detected in human biomonitoring programs and frequently found in FCMs (Fig. 5A ), and there is ample evidence for their migration, e.g. [ 17 , 59 , 60 , 61 ]. There is also evidence for the presence of PFAS in humans and in FCMs. Although most PFAS have never been authorized for food contact use [ 62 ], the contribution of food packaging to human exposure has been mapped [ 63 ]. Dioxin-like compounds, many pesticides, and flame retardants are not intentionally added FCCs, but they may be present in FCMs because they are introduced or formed during FCM use, manufacture, and recycling, as their detection in FCMs shows [ 64 , 65 , 66 ]. FCMs may therefore contribute to human exposure to FCCs intentionally used in the manufacture of FCMs, various types of NIAS, and illicitly added chemicals. Yet, for most FCCs, comprehensive assessments of the relative contribution of FCMs to human body burden are missing.
Antioxidants are of special interest because many are high-production volume chemicals that are widely used in plastic food packaging [ 67 ] and robust evidence for their presence in FCMs exists (Fig. 5B , Table S2 ). Important groups of antioxidants are sterically hindered phenols and phosphite antioxidants that are very common in FCMs, e.g., Irgafos 168 (CAS 31570-04-4), Irganox 1076 (CAS 2082-79-3), and Irganox 1010 (CAS 6683-19-8). However, neither of these substances is included in the biomonitoring programs and exposome/metabolome databases (step 1), and we found only limited evidence for their presence in humans in step 2 [ 52 , 68 , 69 ]. Major degradation products of these antioxidants, such as 2,4-di-tert-butylphenol (CAS 96-76-4), 2,6-di-tert-butylbenzoquinone (CAS 719-22-2), and tris(2,4-di-tert-butylphenyl)phosphate (CAS 95906-11-9), have been detected in humans in a few studies, but at high levels and with frequent detection in sampled populations [ 70 , 71 , 72 ]. These results show that the contribution of FCMs to human exposure to antioxidants and their degradation products has not yet received much attention. Such gaps need to be filled by better understanding the overall exposure to antioxidants and their metabolism in humans.
Oligomers are another group of FCCs requiring more attention. PET, PA, and siloxane oligomers are known side-products of polymerization, and they have been detected in extracts and migrates of FCMs. There is however only very limited evidence for their presence in humans, e.g. for PET oligomers [ 73 ]. This is likely due to the challenging chemical analysis of oligomers, especially in complex media, such as human samples, and the fact that chemical standards required for the identification and quantification of oligomers are rarely available [ 74 , 75 ]. BADGE and its derivatives are commonly observed side-products formed during the polymerization of epoxy resins [ 76 ]. Toxic effects, such as endocrine disruption, genotoxicity, and allergic reactions, have been linked to BADGE derivatives and epoxy resins, but information on their toxicity is still limited [ 77 ]. Seventeen BADGE derivatives have been detected in extracts or migrates of FCMs, and five of them have been found in humans. This illustrates that targeted analysis of structurally related chemicals is possible and should be prioritized, to close this important knowledge gap on human exposure to expected side-products of polymerization reactions [ 76 ].
Photoinitiators form a group of structurally diverse FCCs that are used in various FCMs, such as coatings, printing inks, and adhesives [ 78 ]. While there is substantial evidence for their presence in FCMs, their presence in human samples has not been extensively investigated. Liu and Mabury showed that 18 photoinitiators and their sulfoxidation products are present in human sera [ 79 ], and human exposure, environmental occurrence, and toxicity of 25 photoinitiators have recently been reviewed [ 78 ]. According to the FCCmigex and FCChumon databases, several of these photoinitiators have been detected in FCMs and there is evidence for human exposure. Among these, benzophenone (CAS 119-61-9) is the most frequently detected photoinitiator in FCMs. Since benzophenone is a presumed carcinogen (class 1B, Table S1 ) as well as a suspected endocrine disruptor [ 80 ], exposure via FCMs should be prevented.
Limitations affecting data interpretation
The sources used for the compilation of the FCChumon data vary with respect to the chemical space, curation level, and details provided. In general, we consider data collected in biomonitoring programs (Step 1A) as having a high level of confidence because they are usually derived from a representative population by following strict analytical standards and guidelines [ 81 ]. However, only a limited number of several hundred chemicals is monitored in these programs. We also rate the results of step 2 with a high level of confidence because they were generated by the robust approach of a systematic evidence map (including data extraction by a trained team of scientists but excluding the quality rating of each included study [ 29 ]). Conversely, the metabolome/exposome databases contain many thousands of different chemicals that have been assembled by different means, also including automated approaches [ 36 , 38 ]. The matches between the known FCCs and these databases may therefore require further review before being used in future assessments (e.g., by checking the “metabolite status” integrated in the HMDB).
Some of the FCCs listed in the FCCdb and FCCmigex consist of chemical mixtures of, e.g., polymeric molecules, stereoisomers, or structural isomers. Converting the CAS Registry Number of such mixtures into other identifiers was not always possible and could therefore result in some FCCs not being found in some sources of step 1. For example, short-chain chlorinated paraffins (SCCPs, CAS 85535-84-8) and medium-chain chlorinated paraffins (MCCPs, CAS 85535-85-9) do not have any identifiers other than CAS and were not matched in step 1, but we found ample evidence for the presence of these mixtures in humans in step 2, because they have been monitored regularly and the chemical names are reported in a standardized manner in the primary literature e.g. [ 82 , 83 , 84 ]. Nonylphenol (CAS 25154-52-3) is another example of a mixture of undefined stereoisomers and structural isomers that was not found in step 1 but prioritized in step 2. However, due to the listing of more defined nonylphenol isomers in the FCC databases as well as the metabolome/exposome databases, we decided to exclude this technical mixture from the systematic evidence map. These examples show that searches for (alternative) names and/or identifiers were helpful during the systematic evidence map and may be recommended for users of the FCChumon database.
Implications for assessing and managing FCCs
The data presented here lend support to the possible contribution of FCMs towards human exposure to FCCs. Since there are various FCCs with hazard properties of concern among the chemicals detected in humans and FCMs, their use in FCMs should be restricted to minimize human exposure. This is now recognized and currently under discussion for a few of these chemicals, including PFAS [ 85 , 86 ], BPA [ 10 , 16 ] and phthalates [ 87 ]. However, it does not mean that the remaining FCCs can be considered safe, as shown, e.g., by the absence of biomonitoring and hazard data for 107 (61%) and 59 (34%), respectively, of the 175 FCCs included in step 2. Importantly, even for chemicals where hazard data have been submitted to authorities there are significant data gaps for one or more hazard categories, as has been demonstrated for certain PFAS [ 62 , 88 ]. For FCCs migrating into foods, such related hazard data gaps need to be filled with high priority to characterize risk on human health [ 89 ]. This is especially urgent for intentionally added FCCs found at high levels in humans, such as antioxidants and photoinitiators, and expected NIAS, such as oligomers and BADGE derivatives.
In summary, this study systematically maps 3601 chemicals from different FCAs (food packaging, tableware, etc.) for which there is evidence for human exposure, and for 10,786 FCCs, no evidence could be provided at all. Only 15 FCCs have been monitored but have never been detected in humans. Based on two subsets totalling 410 FCCs, this study further identifies 105 FCCs of high concern due to their hazard properties and highlights the many data gaps related to hazards and human health risks. We make these data accessible in the user-friendly, freely accessible FCChumon dashboard, which complements our previously published FCCmigex dashboard on extractable and migrating FCCs. In combination, FCChumon and FCCmigex enable the prioritization of FCCs requiring more detailed investigations, either because they are frequently found in FCMs, despite having only little or no information on their presence in humans, or because they are measured in humans but lack hazard information. Furthermore, this evidence base supports policy and decision-making and highlights the urgent need to ban the most hazardous chemicals shown to migrate from food packaging and other types of FCAs into foods, to protect human health.
Data availability
The data are publicly and freely available as interactive dashboard that is based on Microsoft PowerBI under the following link ( https://www.foodpackagingforum.org/fcchumon ). The references that were included in the systematic evidence map (step 2) are also provided under this link.
WHO. Human biomonitoring: Facts and figures. Copenhagen: WHO Regional Office for Europe. 2015.
Choi J, Mørck TA, Joas A, Knudsen LE. Major national human biomonitoring programs in chemical exposure assessment. AIMS Environ Sci. 2015;2:782–802.
Article Google Scholar
Landrigan PJ, Fuller R, Acosta NJR, Adeyi O, Arnold R, Basu N, et al. The Lancet Commission on pollution and health. Lancet. 2018;391:462–512.
Article PubMed Google Scholar
Grob K, Biedermann M, Scherbaum E, Roth M, Rieger K. Food contamination with organic materials in perspective: packaging materials as the largest and least controlled source? A view focusing on the European situation. Crit Rev Food Sci Nutr. 2006;46:529–35.
Article CAS PubMed Google Scholar
Geueke B, Groh KJ, Maffini MV, Martin OV, Boucher JM, Chiang Y-T, et al. Systematic evidence on migrating and extractable food contact chemicals: Most chemicals detected in food contact materials are not listed for use. Crit Rev Food Sci Nutr. 2022;63:9425–35.
Jeddi MZ, Boon PE, Cubadda F, Hoogenboom R, Mol H, Verhagen H, et al. A vision on the ‘foodture’ role of dietary exposure sciences in the interplay between food safety and nutrition. Trends Food Sci Tech. 2022;120:288–300.
Article CAS Google Scholar
Barnes KA, Sinclair CR, Watson DH, (eds.). Chemical migration and food contact materials. Woodhead Publishing, 2007.
Hahladakis JN, Velis CA, Weber R, Iacovidou E, Purnell P. An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. J Hazard Mater. 2018;344:179–99.
Wang Y, Zhu H, Kannan K. A review of biomonitoring of phthalate exposures. Toxics. 2019;7:21.
Article PubMed PubMed Central Google Scholar
EFSA CEP Panel. Re-evaluation of the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs. EFSA J. 2023;21:e06857.
Google Scholar
EFSA CEP Panel. Update of the risk assessment of di-butylphthalate (DBP), butyl-benzyl-phthalate (BBP), bis(2-ethylhexyl)phthalate (DEHP), di-isononylphthalate (DINP) and di-isodecylphthalate (DIDP) for use in food contact materials. EFSA J. 2019;17:e05838.
HBM4EU. Substance report - Bisphenols. 2022. https://www.hbm4eu.eu/wp-content/uploads/2022/07/Bisphenols_Substance-report.pdf . Accessed 7 February 2024.
Banaderakhshan R, Kemp P, Breul L, Steinbichl P, Hartmann C, Fürhacker M. Bisphenol A and its alternatives in Austrian thermal paper receipts, and the migration from reusable plastic drinking bottles into water and artificial saliva using UHPLC-MS/MS. Chemosphere. 2022;286:131842.
Siddique S, Zhang G, Coleman K, Kubwabo C. Investigation of the migration of bisphenols from baby bottles and sippy cups. Curr Res Food Sci. 2021;4:619–26.
Article CAS PubMed PubMed Central Google Scholar
Marchiandi J, Alghamdi W, Dagnino S, Green MP, Clarke BO. Exposure to endocrine disrupting chemicals from beverage packaging materials and risk assessment for consumers. J Hazard Mater. 2024;465:133314.
EC. Draft regulation on the use of BPA and other bisphenols in FCMs. 2024. https://ec.europa.eu/transparency/comitology-register/core/api/integration/ers/400274/097818/1/attachment . Accessed 16 July 2024.
Groh KJ, Geueke B, Martin O, Maffini M, Muncke J. Overview of intentionally used food contact chemicals and their hazards. Environ Int. 2021;150:106225.
Nerín C, Bourdoux S, Faust B, Gude T, Lesueur C, Simat T, et al. Guidance in selecting analytical techniques for identification and quantification of non-intentionally added substances (NIAS) in food contact materials (FCMS). Food Addit Contam A. 2022;39:620–43.
Geueke B. Dossier - Non-intentionally added substances. 2018. https://www.foodpackagingforum.org/fpf-2016/wp-content/uploads/2018/06/FPF_Dossier03_NIAS_2nd-edition.pdf . Accessed 7 February 2024.
Zimmermann L, Scheringer M, Geueke B, Boucher JM, Parkinson LV, Groh KJ, et al. Implementing the EU Chemicals Strategy for Sustainability: The case of food contact chemicals of concern. J Hazard Mater. 2022;437:129167.
vom Saal FS, Vandenberg LN. Update on the health effects of bisphenol A: Overwhelming evidence of harm. Endocrinology. 2020;162.
Neltner TG, Alger HM, Leonard JE, Maffini MV. Data gaps in toxicity testing of chemicals allowed in food in the United States. Reprod Toxicol. 2013;42:85–94.
Muncke J, Andersson A-M, Backhaus T, Belcher SM, Boucher JM, Carney Almroth B, et al. A vision for safer food contact materials: Public health concerns as drivers for improved testing. Environ Int. 2023;180:108161.
Balbus JM, Barouki R, Birnbaum LS, Etzel RA, Gluckman PD, Grandjean P, et al. Early-life prevention of non-communicable diseases. Lancet. 2013;381:3–4.
Muncke J, Myers JP, Scheringer M, Porta M. Food packaging and migration of food contact materials: will epidemiologists rise to the neotoxic challenge? J Epidemiol Commun H. 2014;68:592.
Simoneau C, Raffael B, Garbin S, Hoekstra E, Mieth A, Lopes J, et al. Non-harmonised food contact materials in the EU: Regulatory and market situation. Baseline study, final report. Publications Office of the European Union. 2016; JRC104198.
Muncke J, Backhaus T, Geueke B, Maffini MV, Martin OV, Myers JP, et al. Scientific challenges in the risk assessment of food contact materials. Environ Health Perspect. 2017;125:095001.
Food Packaging Forum. FCCmigex Database. 2023. https://www.foodpackagingforum.org/resources/fccmigex . Accessed 16 July 2024.
Geueke B, Parkinson LV, Dolenc J, Groh KJ, Kassotis CD, Maffini MV et al. Protocol for assessing the evidence of food contact chemicals monitored in humans. 2023; https://doi.org/10.5281/zenodo.7857837 .
National Center for Environmental Health, National Center for Health Statistics, National Health and Nutrition Examination Survey. Fourth national report on human exposure to environmental chemicals. Updated tables, March 2021, volume three: Analysis of pooled serum samples for select chemicals, NHANES 2005-16. 2021.
Health Canada. Sixth report on human biomonitoring of environmental chemicals in Canada. Minister of Health, Ottawa, ON. 2021. https://www.canada.ca/en/health-canada/services/environmental-workplace-health/reports-publications/environmental-contaminants/sixth-report-human-biomonitoring.html . Accessed 7 February 2023.
HBM4EU. EU HBM Dashboard. 2022. https://www.hbm4eu.eu/what-we-do/european-hbm-platform/eu-hbm-dashboard/ . Accessed 7 February 2023.
IPCHEM. Human Biomonitorin Data Module: HBM4EU-aggregated - HBM4EU aggregated workbook by VITO. 2022. https://ipchem.jrc.ec.europa.eu/#showmetadata/HBM4EUAGGREGATED . Accessed 7 February 2023.
Jung SK, Choi W, Kim SY, Hong S, Jeon HL, Joo Y, et al. Profile of environmental chemicals in the Korean population - Results of the Korean National Environmental Health Survey (KoNEHS) cycle 3, 2015-2017. Int J Environ Res Pub He. 2022;19:626.
Biomonitoring California. Chemicals biomonitored in California. 2022. https://biomonitoring.ca.gov/chemicals/chemicals-biomonitored-california . Accessed 7 February 2023.
Wishart DS, Guo A, Oler E, Wang F, Anjum A, Peters H, et al. HMDB 5.0: the Human Metabolome Database for 2022. Nucleic Acids Res. 2022;50:D622–31.
Wishart Research Group. The Human Metabolome Database. https://hmdb.ca/ . Accessed 7 February 2023.
Barupal DK, Fiehn O. Generating the Blood Exposome Database using a comprehensive text mining and database fusion approach. Environ Health Perspect. 2019;127:97008.
Barupal D, Fiehn O. Blood Exposome Database. https://bloodexposome.org/ . Accessed 9 February 2023.
Neveu V, Nicolas G, Salek RM, Wishart DS, Scalbert A. Exposome-Explorer 2.0: an update incorporating candidate dietary biomarkers and dietary associations with cancer risk. Nucleic Acids Res. 2019;48:D908–12.
PubMed Central Google Scholar
IARC. Exposome Explorer 3.0. 2021. http://exposome-explorer.iarc.fr/ . Accessed 9 February 2023.
Geueke B, Parkinson LV. S112 | FCCMIGEX | List of Migrating & Extractable Food Contact Chemicals (FCCmigex) by FPF. Zenodo. 2024. https://doi.org/10.5281/zenodo.10551195 .
Groh KJ, Geueke B, Chirsir P, Schymanski EL, Muncke J. S77 | FCCDB | Food Contact Chemicals Database v5.0. Zenodo. 2022. https://doi.org/10.5281/zenodo.7304977 .
Mohammed Taha H, Aalizadeh R, Alygizakis N, Antignac J-P, Arp HPH, Bade R, et al. The NORMAN Suspect List Exchange (NORMAN-SLE): facilitating European and worldwide collaboration on suspect screening in high resolution mass spectrometry. Environ Sci Eur. 2022;34:104.
Djoumbou Feunang Y, Eisner R, Knox C, Chepelev L, Hastings J, Owen G, et al. ClassyFire: automated chemical classification with a comprehensive, computable taxonomy. J Cheminform. 2016;8:61.
Zweifel H, Maier RD, Schiller M Plastics Additives Handbook. Carl Hanser Verlag: Munich, Germany, 2009.
Kohl C, McIntosh EJ, Unger S, Haddaway NR, Kecke S, Schiemann J, et al. Online tools supporting the conduct and reporting of systematic reviews and systematic maps: a case study on CADIMA and review of existing tools. Environ Evid. 2018;7:8.
EC. Chemicals Strategy for Sustainability, towards a toxic-free environment. COM(2020) 667 final. 2020.
ECHA. C&L Inventory. 2023. https://echa.europa.eu/information-on-chemicals/cl-inventory-database . Accessed 16 November 2023.
NITE. Chemical Management - GHS General Information. 2023. https://www.nite.go.jp/chem/english/ghs/ghs_index.html . Accessed 16 November 2023.
Tkalec Ž, Codling G, Tratnik JS, Mazej D, Klánová J, Horvat M, et al. Suspect and non-targeted screening-based human biomonitoring identified 74 biomarkers of exposure in urine of Slovenian children. Environ Pollut. 2022;313:120091.
Liu R, Mabury SA. Rat metabolism study suggests 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid as a potential urinary biomarker of human exposure to representative 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate antioxidants. Environ Sci Technol. 2021;55:14051–8.
Frederiksen H, Upners EN, Ljubicic ML, Fischer MB, Busch AS, Hagen CP, et al. Exposure to 15 phthalates and two substitutes (DEHTP and DINCH) assessed in trios of infants and their parents as well as longitudinally in infants exclusively breastfed and after the introduction of a mixed diet. Environ Int. 2022;161:107107.
Haines DA, Saravanabhavan G, Werry K, Khoury C. An overview of human biomonitoring of environmental chemicals in the Canadian Health Measures Survey: 2007-2019. Int J Hyg Environ Health. 2017;220:13–28.
de Bruyn Kops C, Šícho M, Mazzolari A, Kirchmair J. GLORYx: Prediction of the metabolites resulting from phase 1 and phase 2 biotransformations of xenobiotics. Chem Res Toxicol. 2021;34:286–99.
Mazzolari A, Scaccabarozzi A, Vistoli G, Pedretti A. MetaClass, a comprehensive classification system for predicting the occurrence of metabolic reactions based on the MetaQSAR database. Molecules. 2021;26:17.
Scholz VA, Stork C, Frericks M, Kirchmair J. Computational prediction of the metabolites of agrochemicals formed in rats. Sci Total Environ. 2023;895:11.
Kincaid B, Piechota P, Golden E, Maertens M, Hartung T, Maertens A. Using in silico tools to predict flame retardant metabolites for more informative exposomics-based approaches. Front Toxicol. 2023;5:16.
Rudel RA, Gray JM, Engel CL, Rawsthorne TW, Dodson RE, Ackerman JM, et al. Food packaging and bisphenol A and bis(2-ethyhexyl) phthalate exposure: findings from a dietary intervention. Environ Health Perspect. 2011;119:914–20.
Tang W, Hemm I, Eisenbrand G. Estimation of human exposure to styrene and ethylbenzene. Toxicology. 2000;144:39–50.
Stahl T, Falk S, Rohrbeck A, Georgii S, Herzog C, Wiegand A, et al. Migration of aluminum from food contact materials to food-a health risk for consumers? Part I of III: exposure to aluminum, release of aluminum, tolerable weekly intake (TWI), toxicological effects of aluminum, study design, and methods. Environ Sci Eur. 2017;29:19.
Phelps DW, Parkinson LV, Boucher JM, Muncke J, Geueke B. Per- and polyfluoroalkyl substances in food packaging: migration, toxicity, and management strategies. Environ Sci Technol. 2024;58:5670–84.
Holder C, DeLuca N, Luh J, Alexander P, Minucci JM, Vallero DA, et al. Systematic evidence mapping of potential exposure pathways for per- and polyfluoroalkyl substances based on measured occurrence in multiple media. Environ Sci Technol. 2023;57:5107–16.
Paseiro-Cerrato R, Ackerman L, de Jager L, Begley T. Brominated flame retardants (BFRs) in contaminated food contact articles: identification using DART-HRMS and GC-MS. Food Addit Contam A. 2021;38:350–9.
BEUC. Towards safe and sustainable food packaging - European consumer organisations call for action on single-use tableware made of alterantives to plastic. BEUC-X-2021-050. 2021.
Cramer G, Bolger M, Henry S, Lorentzen R. Usfda assessment of exposure to 2,3,7,8-TCDD and 2,3,7,8-TCDF from foods contacting bleached paper products. Chemosphere. 1991;23:1537–50.
Huang Y-Q, Zeng Y, Mai J-L, Huang Z-S, Guan Y-F, Chen S-J. Disposable plastic waste and associated antioxidants and plasticizers generated by online food delivery services in china: national mass inventories and environmental release. Environ Sci Technol. 2024; https://doi.org/10.1021/acs.est.3c06345 .
Cortéjade A, Buleté A, Prouteau L, Chatti S, Cren C, Vulliet E. Development and optimisation of home-made stir bar sorptive extraction for analysis of plastic additives: application in human urine. Anal Methods. 2017;9:3549–60.
Pouech C, Kiss A, Lafay F, Léonard D, Wiest L, Cren-Olivé C, et al. Human exposure assessment to a large set of polymer additives through the analysis of urine by solid phase extraction followed by ultra high performance liquid chromatography coupled to tandem mass spectrometry. J Chromatogr A. 2015;1423:111–23.
Liu R, Mabury SA. Unexpectedly high concentrations of 2,4-di-tert-butylphenol in human urine. Environ Pollut. 2019;252:1423–8.
Raman M, Ahmed I, Gillevet PM, Probert CS, Ratcliffe NM, Smith S, et al. Fecal microbiome and volatile organic compound metabolome in obese humans with nonalcoholic fatty liver disease. Clin Gastroenterol H. 2013;11:868–75.
Ibrahim B, Basanta M, Cadden P, Singh D, Douce D, Woodcock A, et al. Non-invasive phenotyping using exhaled volatile organic compounds in asthma. Thorax. 2011;66:804–9.
Diamantidou D, Mastrogianni O, Tsochatzis E, Theodoridis G, Raikos N, Gika H, et al. Liquid chromatography-mass spectrometry method for the determination of polyethylene terephthalate and polybutylene terephthalate cyclic oligomers in blood samples. Anal Bioanal Chem. 2022;414:1503–12.
Alberto Lopes J, Tsochatzis ED. Poly(ethylene terephthalate), poly(butylene terephthalate), and polystyrene oligomers: occurrence and analysis in food contact materials and food. J Agr Food Chem. 2023;71:2244–58.
Schreier VN, Appenzeller-Herzog C, Bruschweiler BJ, Geueke B, Wilks MF, Simat TJ, et al. Evaluating the food safety and risk assessment evidence-base of polyethylene terephthalate oligomers: Protocol for a systematic evidence map. Environ Int. 2022;167:107387.
Wang D, Zhao H, Fei X, Synder SA, Fang M, Liu M. A comprehensive review on the analytical method, occurrence, transformation and toxicity of a reactive pollutant: BADGE. Environ Int. 2021;155:106701.
Xue J, Liu Y, Yang D, Zhao Y, Cai Y, Zhang T, et al. A review of properties, production, human exposure, biomonitoring, toxicity, and regulation of bisphenol A diglycidyl ethers and novolac glycidyl ethers. J. Environ Chem Ecotoxicol. 2022;4:216–30.
Ji X, Liang J, Liu J, Shen J, Li Y, Wang Y, et al. Occurrence, fate, human exposure, and toxicity of commercial photoinitiators. Environ Sci Technol. 2023;57:11704–17.
Liu R, Mabury SA. First detection of photoinitiators and metabolites in human sera from United States donors. Environ Sci Technol. 2018;52:10089–96.
Lee J, Kim S, Park YJ, Moon H-B, Choi K. Thyroid hormone-disrupting potentials of major benzophenones in two cell lines (GH3 and FRTL-5) and embryo-larval zebrafish. Environ Sci Technol. 2018;52:8858–65.
Govarts E, Gilles L, Rodriguez Martin L, Santonen T, Apel P, Alvito P, et al. Harmonized human biomonitoring in European children, teenagers and adults: EU-wide exposure data of 11 chemical substance groups from the HBM4EU Aligned Studies (2014-2021). Int J Hyg Environ Health. 2023;249:114119.
Liu Y, Aamir M, Li M, Liu K, Hu Y, Liu N, et al. Prenatal and postnatal exposure risk assessment of chlorinated paraffins in mothers and neonates: Occurrence, congener profile, and transfer behavior. J Hazard Mater. 2020;395:122660.
Martínez C, Martínez Arroyo A, Barrientos Alemán D, Gavilán García A, Caba M, Calderón Garcidueñas AL, et al. Persistent organic compounds in human milk and evaluation of the effectiveness of the Stockholm convention in Mexico. Environ Adv. 2022;8:100190.
Xia D, Gao L-R, Zheng M-H, Li J-G, Zhang L, Wu Y-N, et al. Health risks posed to infants in rural China by exposure to short- and medium-chain chlorinated paraffins in breast milk. Environ Int. 2017;103:1–7.
ECHA. Annex XV. Restriction Report, Proposal for a restriction, Per and polyfluoroalkyl substances (PFASs). 2023. https://echa.europa.eu/documents/10162/1c480180-ece9-1bdd-1eb8-0f3f8e7c0c49 . Accessed 6 February 2024.
US FDA. FDA Announces PFAS Used in Grease-Proofing Agents for Food Packaging No Longer Being Sold in the U.S. 2024. https://www.fda.gov/food/cfsan-constituent-updates/fda-announces-pfas-used-grease-proofing-agents-food-packaging-no-longer-being-sold-us . Accessed 28 February 2024.
EFSA CEP Panel. Identification and prioritisation for risk assessment of phthalates, structurally similar substances and replacement substances potentially used as plasticisers in materials and articles intended to come into contact with food. EFSA Journal. 2022;20:e07231.
Rudin E, Glüge J, Scheringer M. Per- and polyfluoroalkyl substances (PFASs) registered under REACH-What can we learn from the submitted data and how important will mobility be in PFASs hazard assessment? Sci Total Environ. 2023;877:162618.
Pellizzari ED, Woodruff TJ, Boyles RR, Kannan K, Beamer PI, Buckley JP, et al. Identifying and prioritizing chemicals with uncertain burden of exposure: Opportunities for biomonitoring and health-related research. Environ Health Persp. 2019;127:126001.
Download references
Acknowledgements
We thank all members of the FCCH project’s scientific advisory group for their contributions, especially John Peterson Myers, Katie Pelch, Rob Sargis, Emma Schymanski, and Martin Wagner. We thank Jozica Dolenc for help with the development of the literature search strategy and Frank Gwodsz and Christian Kohl for technical support during the compilation of the systematic evidence map.
This work was carried out as part of the FCCH project, which is funded by project-related funds from Adessium Foundation, MAVA Foundation, Stiftung Minerva, Sympany Stiftung, and the Food Packaging Forum’s own resources from unrestricted donations. All FPF funding sources are listed here: https://www.foodpackagingforum.org/about-us/funding .
Author information
Authors and affiliations.
Food Packaging Forum Foundation, Zurich, Switzerland
Birgit Geueke, Lindsey V. Parkinson, Lisa Zimmermann & Jane Muncke
Department of Environmental Toxicology, Eawag, Swiss Federal Institute of Aquatic Science and Technology, Duebendorf, Switzerland
Ksenia J. Groh
Institute of Environmental Health Sciences and Department of Pharmacology, Wayne State University, Detroit, MI, USA
Christopher D. Kassotis
Independent Consultant, Frederick, MD, USA
Maricel V. Maffini
Department of Arts & Science, Plastic Waste Innovation Hub, University College London, London, UK
Olwenn V. Martin
RECETOX, Masaryk University, Brno, Czech Republic
Martin Scheringer
Department of Environmental Systems Science, ETH Zurich, Zurich, Switzerland
You can also search for this author in PubMed Google Scholar
Contributions
BG: Conceptualization, Literature screening, Data extraction, Visualization, Writing (original draft), Project administration; LVP: Data processing, Dashboard development, Writing (review & editing); LZ: Literature screening, Data extraction, Writing (review & editing); KJG: Conceptualization, Writing (review & editing); CK: Conceptualization, Writing (review & editing); MVM: Conceptualization, Writing (review & editing); OVM: Conceptualization, Writing (review & editing); MS: Data interpretation, Writing (review & editing); JM : Conceptualization, Data interpretation, Writing (review & editing), Funding acquisition. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Correspondence to Birgit Geueke .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Protocol for assessing the evidence of food contact chemicals monitored in humans, supplementary tables, rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ .
Reprints and permissions
About this article
Cite this article.
Geueke, B., Parkinson, L.V., Groh, K.J. et al. Evidence for widespread human exposure to food contact chemicals. J Expo Sci Environ Epidemiol (2024). https://doi.org/10.1038/s41370-024-00718-2
Download citation
Received : 08 May 2024
Revised : 23 August 2024
Accepted : 28 August 2024
Published : 17 September 2024
DOI : https://doi.org/10.1038/s41370-024-00718-2
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies






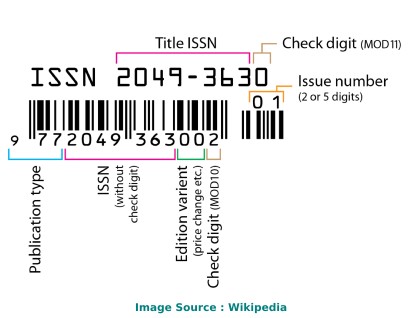





















IMAGES
VIDEO
COMMENTS
Publisher. Research Journal of BioTechnology. H-Index. 22. Publication type. Journals. ISSN. 09736263, 22784535. Coverage. 2008-2023. Information. Homepage. ... The chart shows the evolution of the average number of times documents published in a journal in the past two, three and four years have been cited in the current year. The two years ...
Research Journal of BioTechnology (ISSN: 0973-6263) Share Your Research, Maximize Your Social Impacts ... BioTechnology Journal is published quarterly-four issues in a year. We request you to contribute your research papers and review papers at your earliest. All papers received by us are sent to two experts for their comments and if both the ...
Research Journal of Biotechnology is a journal published by Research Journal of BioTechnology. Check Research Journal of Biotechnology Impact Factor, Overall Ranking, Rating, h-index, Call For Papers, Publisher, ISSN, Scientific Journal Ranking (SJR), Abbreviation, Acceptance Rate, Review Speed, Scope, Publication Fees, Submission Guidelines, other Important Details at Resurchify
The Research Journal Of Biotechnology is a research journal that publishes research related to Biochemistry, Genetics and Molecular Biology; Chemical Engineering; Immunology and Microbiology. This journal is published by the Research Journal of BioTechnology. The ISSN of this journal is 22784535, 09736263.
Know all about RESEARCH JOURNAL OF BIOTECHNOLOGY - Impact factor, Acceptance rate, Scite Analysis, H-index, SNIP Score, ISSN, Citescore, SCImago Journal Ranking (SJR), Aims & Scope, Publisher, and Other Important Metrics. Click to know more about RESEARCH JOURNAL OF BIOTECHNOLOGY Review Speed, Scope, Publication Fees, Submission Guidelines.
Research Journal of Biotechnology is a journal published by Research Journal of BioTechnology. This journal covers the area[s] related to Applied Microbiology and Biotechnology, Bioengineering, Biotechnology, etc. The coverage history of this journal is as follows: 2008-2022. The rank of this journal is 22791. This journal's impact score, h ...
Journal Rankings on Biotechnology. All subject areas. Biotechnology. Agricultural and Biological Sciences (miscellaneous) Biochemistry, Genetics and Molecular Biology (miscellaneous) Business, Management and Accounting (miscellaneous) Economics, Econometrics and Finance (miscellaneous) Organizational Behavior and Human Resource Management.
The Journal of Biotechnology (J. Biotech.) offers publication for both full-length articles and short communications on a wide range of biotechnology-related topics.Also, review articles can be submitted if they are pre-approved by one of the editors. The journal will only accept submissions with novel scientific research results that are directly relevant to biotechnological systems and/or ...
Last modification date: 13/08/2024. Type of record: Confirmed. ISSN Center responsible of the record: ISSN National Centre for India. For all potential issues concerning the description of the publication identified by this bibliographic record (missing or wrong data etc.), please contact the ISSN National Centre mentioned above by clicking on ...
Publisher. Research Journal of BioTechnology Publication Frequency. Quarterly Year Publication Started. 2006 Annual Article Volume. 242 Open Access. NO Contact. RESEARCH JOURNAL BIOTECHNOLOGY, SECTOR A-80, SCHEME NO 54, VIJAY NAGAR, A B ROAD, INDORE, INDIA, 452 010 MP ...
Scope. Journal of Biotech Research (JBR) is an open access, international scientific electronic journal which publishes original peer-reviewed articles, short communications, and critical review papers from all areas related to Biotechnology. The journal aims at a broad interdisciplinary readership, which includes both academia and industries ...
Get access to RESEARCH JOURNAL OF BIOTECHNOLOGY details, impact factor, Journal Ranking, H-Index, ISSN, Citescore, Scimago Journal Rank (SJR). Check top authors, submission guidelines, Acceptance Rate, Review Speed, Scope, Publication Fees, Submission Guidelines at one place. Improve your chances of getting published in RESEARCH JOURNAL OF BIOTECHNOLOGY with Researcher.Life.
Publisher: » In order to submit a manuscript to this journal, please read the guidelines for authors in the journal's homepage. » For a more in-depth analysis of the journal, you should subscribe and check it out on Journal Citation Reports (JCR). » If you need a journal template (Word or Latex), you can read this entry.
Research Journal of Biotechnology Scopus 4-Year Impact Factor Trend. Note: impact factor data for reference only ... is a scientometric factor based on the yearly average number of citations on articles published by a particular journal in the last two years. A journal impact factor is frequently used as a proxy for the relative importance of a ...
The Journal of Biotechnology (J. Biotech.) offers publication for both full-length articles and short communications on a wide range of biotechnology-related topics.Also, review articles can be submitted if they are pre-approved by one of the editors. The journal will only accept submissions with novel scientific research results that are directly relevant to biotechnological systems and/or ...
Current Research in Biotechnology (CRBIOT) is a new primary research, gold open access journal from Elsevier. CRBIOT publishes original papers, reviews, and short communications (including viewpoints and perspectives) resulting from research in biotechnology and biotech-associated disciplines. Curre…. View full aims & scope.
A monthly journal covering the science and business of biotechnology. It publishes new concepts and enabling technology of relevance to the biological, biomedical, agricultural and environmental ...
RESEARCH JOURNAL OF BIOTECHNOLOGY. ISSN 0973-6263; Diffusion; Title: RESEARCH JOURNAL OF BIOTECHNOLOGY related ISSN: 2278-4535 Country: India. Subject: BIOLOGY. Academic field: BIOTECHNOLOGY AND APPLIED MICROBIOLOGY ...
Inderscience is a global company, a dynamic leading independent journal publisher disseminates the latest research across the broad fields of science, engineering and technology; management, ... The main objective of IJBT is to promote policy-oriented research in the field of biotechnology (covering medicine, agriculture, industry and allied ...
The Journal provides a medium for the rapid publication of both full-length articles and short communications on novel and innovative aspects of biotechnology. The Journal will accept papers ranging from genetic or molecular biological positions to those covering biochemical, chemical or bioprocess engineering aspects as well as computer ...
Journal of Bioscience and Bioengineering stood out as the most productive journal, having contributed to 4.0% of all publications, more than double of the contribution by the 2nd journal (Biotechnology and Bioengineering; contributing 1.8%).This is different from other recently analyzed sets of literature in which no single journal clearly outperformed the others, e.g., in the research fields ...
Regular Issue Subscription Original Research Published on :- August 14, 2024. Umesh H.R, Kushalatha M. Keywords: Anthracene decomposition, strain, pseudomonas, 16S rRNA, phylogenetic analysis. View. PDF (30 USD) 28-43. is a peer-reviewed hybrid open-access journal launched in 2015 focused on the publication of current research work carried out ...
Last modification date: 27/05/2024. Type of record: Confirmed. ISSN Center responsible of the record: ISSN National Centre for India. For all potential issues concerning the description of the publication identified by this bibliographic record (missing or wrong data etc.), please contact the ISSN National Centre mentioned above by clicking on ...
Over 1800 food contact chemicals (FCCs) are known to migrate from food contact articles used to store, process, package, and serve foodstuffs. Many of these FCCs have hazard properties of concern ...
Furthermore, guidelines were published to improve the comparability and reproducibility of microplastic research (79, 80). These developments mark steps toward addressing the complexities of microplastic pollution, emphasizing the need for comprehensive and realistic testing methods to better understand and mitigate the environmental impacts of ...
Biases favoring the wealthy are ubiquitous, and they support and bolster vast resource inequalities across individuals and groups; yet, when these biases are acquired remains unknown. In Experiments 1 through 5 (Total N = 232), using multiple methods, we found that 14- to 18-month-old infants track individuals' wealth (Experiments 1-5), prefer and selectively help rich (vs. poor ...