Search the world's largest collection of clinical case reports
Browse case reports by:

Publish in BMJ Case Reports
Global health case reports.
These are case reports that focus on the causes of ill health, the social determinants of health and access to healthcare services, prevailing local and national issues that affect health and wellbeing, and the challenges in providing care to vulnerable populations or with limited resources.
Read the full collection now
Images in… :
31 July 2023
Unusual association of diseases/symptoms :
24 January 2024
14 July 2023
Obstetrics and gynaecology :
5 March 2024
Case report :
18 October 2023
Case Reports: Rare disease :
Case Reports by specialty
- Anaesthesia
- Dentistry and oral medicine
- Dermatology
- Emergency medicine
- Endocrinology
- General practice and family medicine
- Geriatric medicine
- Haematology
- Infectious diseases
- Obstetrics and gynaecology
- Ophthalmology
- Orthopaedics
- Paediatrics
- Respiratory medicine
- Rheumatology

Global Health Competition
Every year BMJ Case Reports selects authors of global health case reports to join our editorial team as a global health associate editor.
This is an opportunity to gain some editorial experience or join our team on research and educational projects. Students and graduates may apply.
Simply select Global Health Competition when you submit.
Latest Articles
Case Reports: Findings that shed new light on the possible pathogenesis of a disease :
26 June 2024
20 June 2024
Journal of Medical Case Reports
In the era of evidence-based practice, we need practice-based evidence. The basis of this evidence is the detailed information from the case reports of individual people which informs both our clinical research and our daily clinical care. Each case report published in this journal adds valuable new information to our medical knowledge. Prof Michael Kidd AO, Editor-in- Chief

Join the Editorial Board
We are recruiting Associate Editors to join our Editorial Board. Learn more about the role and how to apply here !

- Meet the Editors
Get to know the Editors behind Journal of Medical Case Reports !

megaflopp / Getty Images / iStock
Requirements for case reports submitted to JMCR
• Patient ethnicity must be included in the Abstract under the Case Presentation section.
• Consent for publication is a mandatory journal requirement for all case reports . Written informed consent for publication must be obtained from the patient (or their parent or legal guardian in the case of children under 18, or from the next of kin if the patient has died). For more information, please see our editorial policies .
Report of the Month
Superior mesenteric vein thrombosis due to covid-19 vaccination.
Vaccines have made a significant contribute to sowing the spread of the COVID-19 infection. However, side effects of the vaccination are beginning to appear, and one of which, thrombosis, is a particular problem as it can cuase serious complications. While cases of splanchnic venous thrombosis (SVT) after ChAdOx1 nCoV-19 vaccinations have been reported, cases of SVT mRNA-1273 vaccines are rare.
In this case report, clinicians describe a patient presenting with superior mesentric vein thrombosis following a COVID-19 vaccination, and examine the relationship between the mRNA-1273 vaccines and intestinal ischemia.
- Most accessed
Hepatitis A virus infection associated with bilateral pleural effusion, ascites, and acalculous cholecystitis in childhood: a case report
Authors: Fatima Breim, Bakri Roumi Jamal, Lama Kanaa, Saleh Bourghol, Besher Jazmati and Samia Dadah
Hemothorax caused by injury of musculophrenic artery after ultrasound-guided percutaneous liver biopsy: a case report
Authors: Jing-ru Yang, Sai Wu, Jian Li, Xiao-juan Tian, Zhuo-xi Xue and Xiao-yan Niu
Double vertical interrupted suture for optimal adaptation and stabilization of free gingival graft around dental implants: a case report
Authors: Neda Moslemi, Amirmohammad Dolatabadi, Seyedhossein Mohseni Salehimonfared and Fatemeh Goudarzimoghaddam
Mucinous cystadenoma and carcinoid tumor arising from an ovarian mature cystic teratoma in a 60 year-old patient: a case report
Authors: Amir Masoud Jafari-Nozad, Najmeh Jahani and Yoones Moniri
Bronchobiliary fistula after traumatic liver rupture: a case report
Authors: Teng Zhou, Wenming Wu, Chao Cheng, Hui Wang, Xiaochuan Hu and Zhenhui Jiang
Most recent articles RSS
View all articles
An itchy erythematous papular skin rash as a possible early sign of COVID-19: a case report
Authors: Alice Serafini, Peter Konstantin Kurotschka, Mariabeatrice Bertolani and Silvia Riccomi
Red ear syndrome precipitated by a dietary trigger: a case report
Authors: Chung Chi Chan and Susmita Ghosh
How to choose the best journal for your case report
Authors: Richard A. Rison, Jennifer Kelly Shepphird and Michael R. Kidd
The Erratum to this article has been published in Journal of Medical Case Reports 2017 11 :287
COVID-19 with repeated positive test results for SARS-CoV-2 by PCR and then negative test results twice during intensive care: a case report
Authors: Masafumi Kanamoto, Masaru Tobe, Tomonori Takazawa and Shigeru Saito
Recurrent knee arthritis diagnosed as juvenile idiopathic arthritis with a 10-year asymptomatic period after arthroscopic synovectomy: a case report
Authors: Atsushi Teramoto, Kota Watanabe, Yuichiro Kii, Miki Kudo, Hidenori Otsubo, Takuro Wada and Toshihiko Yamashita
Most accessed articles RSS
A Guide to Writing and Using Case Reports
This thematic series, published in 2016, provides a valuable resource for clinicians who are considered producing a case report. It comprises of a special editorial series of guides on writing, reviewing and using case reports.

Aims and scope
Journal of Medical Case Reports will consider any original case report that expands the field of general medical knowledge, and original research relating to case reports.
Case reports should show one of the following:
- Unreported or unusual side effects or adverse interactions involving medications
- Unexpected or unusual presentations of a disease
- New associations or variations in disease processes
- Presentations, diagnoses and/or management of new and emerging diseases
- An unexpected association between diseases or symptoms
- An unexpected event in the course of observing or treating a patient
- Findings that shed new light on the possible pathogenesis of a disease or an adverse effect
Suitable research articles include but are not limited to: N of 1 trials, meta-analyses of published case reports, research addressing the use of case reports and the prevalence or importance of case reporting in the medical literature and retrospective studies that include case-specific information (age, sex and ethnicity) for all patients.
Article accesses
Throughout 2022, articles were accessed from the journal website more than 4.17 million times; an average of over 11 ,400 accesses per day.
Latest Tweets
Your browser needs to have JavaScript enabled to view this timeline
Peer Review Mentoring Scheme
The Editors at Journal of Medical Case Reports endorse peer review mentoring of early career researchers.
If you are a senior researcher or professor and supervise an early career researcher with the appropriate expertise, we invite you to co-write and mentor them through the peer review process. Find out how to express your interest in the scheme here .
Call for Papers
The Journal of Medical Case Reports is calling for submissions to our Collection on COVID-19 – a look at the past, present and future of the pandemic . Guest Edited by Dr. Jean Karl Soler, The Family Practice Malta, Malta

About the Editor-in-Chief
Professor Michael Kidd AO FAHMS is foundation Director of the Centre for Future Health Systems at the University of New South Wales in Sydney, Australia, and Professor of Global Primary Care and Future Health Systems with the Nuffield Department of Primary Care Health Sciences at the University of Oxford. During the COVID-19 pandemic, Prof Kidd was the Deputy Chief Medical Officer and Principal Medical Advisor with the Australian Government Department of Health and Aged Care, and Professor of Primary Care Reform at the Australian National University. He holds honorary appointments with the University of Toronto, the University of Melbourne, Flinders University, and the Murdoch Children's Research Institute, and is the Emeritus Director of the World Health Organization Collaborating Centre on Family Medicine and Primary Care. He is an elected Fellow of the Australian Academy of Health and Medical Sciences (FAHMS). In the 2023 King's Birthday Honours List he was made an Officer of the Order of Australia. Prof Kidd served as president of the World Organization of Family Doctors (WONCA) from 2013-2016, and as president of the Royal Australian College of General Practitioners from 2002-2006. He is the founder and Editor-in-Chief of the Journal of Medical Case Reports, the world's first PubMed-listed journal devoted to publishing case reports from all medical disciplines.
- Editorial Board
- Manuscript editing services
- Instructions for Editors
- Sign up for article alerts and news from this journal
Annual Journal Metrics
2022 Citation Impact 1.0 - 2-year Impact Factor 0.628 - SNIP (Source Normalized Impact per Paper) 0.284 - SJR (SCImago Journal Rank)
2023 Speed 33 days submission to first editorial decision for all manuscripts (Median) 148 days submission to accept (Median)
2023 Usage 4,048,208 downloads 2,745 Altmetric mentions
- More about our metrics

- Follow us on Twitter
ISSN: 1752-1947
- Submission enquiries: Access here and click Contact Us
- General enquiries: [email protected]

Clinical Cases
Litfl clinical cases database.
The LITFL Clinical Case Collection includes over 250 Q&A style clinical cases to assist ‘ Just-in-Time Learning ‘ and ‘ Life-Long Learning ‘. Cases are categorized by specialty and can be interrogated by keyword from the Clinical Case searchable database.
Search by keywords; disease process; condition; eponym or clinical features…
| Topic | Title | Keywords | ||||
|---|---|---|---|---|---|---|
| ECG | ||||||
| ECG | WCT, ECG, Broad complex, fascicular, RVOT | |||||
| Toxicology | valproate, valproic acid, hyperammonemia | |||||
| Toxicology | valproate, valproic acid, hyperammonemia | |||||
| Toxicology | ||||||
| Metabolic | priapsim, intracavernosal, cavernosal gas, Ischaemic priapism, stuttering priapism, urology | |||||
| Metabolic | RTA, strong ion difference, hypocalcaemia | |||||
| Bone and Joint | DRUJ, dislcoation | |||||
| ICE | wellens, ECG, cardiac, delay | |||||
| ICE | SJS, stevens-johnson syndrome, erythema multiforme, rash | |||||
| ICE | pneumothorax | |||||
| ICE | ||||||
| ICE | tibia, fracture, toddler, toddler's fracture | |||||
| ICE | ECG, EKG, hyperkalaemia, hyperkalemia | |||||
| ICE | dengue, returned traveler, traveller | |||||
| ICE | Lisfranc | |||||
| ICE | mountain, mount everest, alkalaemia, alkalemia | |||||
| ICE | pancreatitis, alcohol | |||||
| ICE | segond fracture | |||||
| ICE | Brugada | |||||
| ICE | STEMI, hyperacture, myocardial ischemia, anterior | |||||
| ICE | eryhthema nodosum, panniculitis | |||||
| ICE | BOS fracture, battle sign, mastoid ecchymosis, bruising | |||||
| ICE | Galleazi, fracture dislocation | |||||
| Toxicology | methylene blue, Methaemoglobinemia, methemoglobin | |||||
| Toxicology | clozapine | |||||
| Toxicology | Methamphetamine, body stuffing, body packer, body stuffer | |||||
| Toxicology | TCA, tricyclic, overdose, sodium channel blockade | |||||
| Toxicology | alprazolam, BZD, benzo, benzodiazepine, benzodiazepines, flumazenil | |||||
| Toxicology | lithium, neurotoxicity, acute toxicity | |||||
| Toxicology | baclofen, GABA, Gamma-Hydroxybutyrate, GHB | |||||
| Toxicology | Carbamazepine, toxidrome, carbamazepine cardiotoxicity, Tegretol, multiple-dose activated charcoal, MDAC | |||||
| Toxicology | Hepatotoxicity, Acetaminophen, Schiodt score, hepatic encephalopathy, N-acetylcysteine, NAC | |||||
| Toxicology | beta-blocker, B Blocker, | |||||
| Toxicology | Cannabinoid Hyperemesis Syndrome, cyclical vomiting, THC, delta-nine-tetrahydrocannabinol | |||||
| Toxicology | Colchicine | |||||
| Toxicology | Clonidine | |||||
| Toxicology | Bath salts | |||||
| Toxicology | Mephedrone | |||||
| Toxicology | Bromo-DragonFLY, M-ket, Kmax, Mexxy, Meow-Meow, Mephedrone, Methoxetamine, Naphyrone, NRG-1, Salvia, K2, Spice | |||||
| Toxicology | ixodes holocyclus, tick, paralysis, | |||||
| Toxicology | cyanide, carbon monoxide | |||||
| Toxicology | hypoglycemia | |||||
| Toxicology | Ciguatera, Scombroid, fugu, puffer fish | |||||
| Toxicology | ethylene glycol, HAGMA, high anion gap metabolic acidosis, osmolar gap, Fomepizole, alcohol, ethanol | |||||
| Toxicology | iron toxicity, Desferrioxamine chelation therapy | |||||
| Toxicology | chloroquine | |||||
| Toxicology | corrosive agent | |||||
| Toxicology | Antidote | |||||
| Toxicology | Oculogyric crisis, OGC, acute dystonia, Acute Dystonic Reaction, butyrophenone, Metoclopramide, haloperidol, prochlorperazine, Benztropine | |||||
| Toxicology | Tricyclic, Theophylline, Sulfonylureas, Propanolol, Opioids, Dextropropoxyphene, Chloroquine, Calcium channel blockers, Amphetamines, ectasy | |||||
| Toxicology | verapamil, calcium channel blocker, cardiotoxic, HIET, high-dose insulin euglycemic therapy, | |||||
| Toxicology | aroma, smell | |||||
| Toxinology | snake-bite, snake bite, Brown snake, Black, Death adder, Taipan, sea snake, tiger | |||||
| Toxicology | Anticholinergic syndrome, Malignant hyperthermia, Neuroleptic malignant syndrome, Serotonin toxicity | |||||
| Toxicology | Serotonin toxicity, Serotonin syndrome, toxidrome | |||||
| Toxicology | proconvulsive, venlafaxine, tramadol, amphetamines, Bupropion, Otis Campbell | |||||
| Toxicology | TCA, tricyclic, overdose, sodium channel blockade, Amitriptyline | |||||
| Toxicology | anticoagulation, warfarin | |||||
| Toxicology | Mickey Finn, pear, | |||||
| Toxicology | thyrotoxic storm, Thyroxine, T4 | |||||
| Toxinology | white-tailed spider, Lampona, L. cylindrata, L. murina | |||||
| Toxicology | Citalopram, SSRI, | |||||
| Toxicology | warfarin | |||||
| Toxicology | warfarin, accidental ingestion, toddler | |||||
| Toxicology | ||||||
| Toxinology | Marine, envenoming | |||||
| Toxinology | Marine, envenoming, penetrating, barb, steve irwin, | |||||
| Toxinology | Marine, envenoming, Blue-Ringed Octopus, BRO, Hapalochlaena | |||||
| Toxinology | Jellyfish, marine, Chironex fleckeri, Box Jellyfish | |||||
| Toxinology | Jellyfish, marine, Jack Barnes, Carukia barnesi, Irukandji Syndrome, Darwin | |||||
| Toxinology | Jellyfish, marine, Jack Barnes, Carukia barnesi, Irukandji Syndrome | |||||
| Toxicology | Strychnine, opisthotonus, risus sardonicus | |||||
| Toxicology | naloxone, Buprenorphine | |||||
| Toxinology | snake-bite, snake bite, SVDK | |||||
| Toxinology | Red back spider, redback, envenoming, RBS | |||||
| Toxinology | Red back spider, redback, envenoming, RBS | |||||
| Toxicology | ||||||
| Toxicology | Acetaminophen, N-acetylcysteine, NAC | |||||
| Pediatric |
| Henoch-Schonlein Purpura, HSP, Henoch-Schönlein | ||||
| Pediatric |
| adrenal insufficiency, glucocorticoid deficiency, NAGMA, endocrine emergency | ||||
| Pediatric |
| Penile Zipper Entrapment, foreskin, release, Zip | ||||
| Pediatric |
| diarrohea, vomiting, hypokalemia, hypokalaemia, dehydration | ||||
| Pediatric |
| infantile colic, TIM CRIES, crying baby | ||||
| Pediatric |
| Pyloric stenosis, projectile vomit, hypertrophic pyloric stenosis, HPS, Rankin | ||||
| Pediatric |
| respiratory distress, wheeze, foreign body, RMB, CXR, right main bronchus | ||||
| Pediatric |
| airway obstruction, stridor, severe croup, harsh cough, heliox, intubation, sevoflurane | ||||
| Pediatric |
| boot-shaped, TOF, coeur en sabot, Tetralogy of Fallot | ||||
| Pediatric |
| Spherocytes, Shistocytes, Polychromasia, reticulocytosis, anemia, anaemia, hemolytic uremic syndrome, HUS | ||||
| Pediatric |
| Reye syndrome, ammonia, metabolic encephalopathy, aspirin | ||||
| Pediatric |
| Ketamine, procedural sedation, pediatric sedation | ||||
| Pediatric |
| Foreign Body, ketamine, laryngospasm, Larson's point, laryngospasm notch | ||||
| EYE |
| ophthalmology, eye trauma, Eyelid laceration, lacrimal punctum | ||||
| EYE |
| ophthalmology, Retrobulbar hemorrhage, haemorrhage, RAPD, lateral canthotomy, DIP-A CONE-G, cantholysis | ||||
| EYE |
| ophthalmology, corneal abrasion, eye trauma, eyelid eversion | ||||
| EYE |
| ophthalmology, commotio retinae, eye trauma, traumatic eye injury | ||||
| EYE |
| ophthalmology, Traumatic iritis, hyphaema, hyphema, | ||||
| EYE |
| ophthalmology, lens dislocation, Anterior dislocation of an intraocular lens | ||||
| EYE |
| ophthalmology, visual loss, loss of vision , blind | ||||
| EYE |
| ophthalmology, Central retinal vein occlusion, CRVO, branch retinal vein occlusion, BRVO | ||||
| EYE |
| ophthalmology, Central retinal artery occlusion, CRAO, cherry red spot, Branch retinal artery occlusion, BRAO | ||||
| EYE |
| ophthalmology, miosis, partial ptosis, anhidrosis, enophthalmos, horner | ||||
| EYE |
| ophthalmology, visual loss, Amaurosis fugax, TIA, transient ischemic attack | ||||
| EYE |
| ophthalmology, Pre-septal cellulitis, preseptal cellulitis, peri-orbital cellulitis, Post-septal cellulitis, post septal cellulitis, orbital cellulitis | ||||
| EYE |
| ophthalmology, AION, giant cell arteritis, GCA, Anterior ischemic optic neuropathy | ||||
| EYE |
| ophthalmology, Herpes simplex keratitis, dendritic ulcer | ||||
| EYE |
| ophthalmology, Conjunctival injection, conjunctivitis, keratoconjunctivitis, Adenovirus, trachoma, bacterial, viral, Parinaud oculoglandular conjunctivitis | ||||
| EYE |
| ophthalmology, Chemical injury, cement, alkali, burn, chemical conjunctivitis, colliquative necrosis, liquefactive | ||||
| EYE |
| ophthalmology, Ultraviolet keratitis, keratopathy, solar keratitis, photokeratitis, welder's flash, arc eye, bake eyes snow blindness. | ||||
| EYE |
| ophthalmology, Parinaud, adie, holmes, tabes dorsalis, neurosyphylis, argyll Robertson, small irregular | ||||
| EYE |
| ophthalmology, anterior Uveitis, HLA-B27, hypopyon | ||||
| EYE |
| ophthalmology, POCUS, ONSD, | ||||
| EYE |
| ophthalmology, Blowout fracture, infraorbital fracture | ||||
| EYE |
| ophthalmology, endophthalmitis, sympathetic ophthalmia, penetrating eye trauma | ||||
| EYE |
| ophthalmology, tobacco dust, Posterior vitreous detachment, vitreous debris, retinal tear, retinal break, Washer Machine Sign, Eales disease | ||||
| EYE |
| ophthalmology, Herpes zoster ophthalmicus, dendriform keratitis, Hutchinson sign | ||||
| EYE |
| ophthalmology, Siedel, FB, rust ring, Corneal foreign body, Seidel test | ||||
| EYE |
| ophthalmology, Papilloedema, Papilledema, pseudopapilloedema | ||||
| EYE |
| ophthalmology, optic disc, optic neuritis, Marcus-Gunn, papillitis, multiple sclerosis, funduscopy, optic atrophy, papilledema | ||||
| EYE |
| ophthalmology, retinal break, POCUS, retinoschisis, Retinal detachment | ||||
| EYE |
| ophthalmology, cupping, glaucoma, optic neuropathy, tonometry, intraocular pressure, open angle, closed angle, gonioplasty, Acute closed-angle glaucoma | ||||
| EYE |
| ophthalmology, Subconjunctival hemorrhage | ||||
| EYE |
| ophthalmology, Meibomitis, blepharitis, entropion, ectropion, canaliculitis, dacryocystitis | ||||
| EYE |
| ophthalmology, blepharospasm, blink, blinking | ||||
| EYE |
| Iritis, keratitis, acute angle-closure glaucoma, scleritis, orbital cellulitis, cavernous sinus thrombosis (CST) | ||||
| EYE |
| ophthalmology, fixed, dilated, pupil, holmes-adie, glass eye | ||||
| ECG |
| Wenckebach, AV block, SA, deliberate mistake, SA block | ||||
| ECG |
| dual chamber AV sequential pacemaker | ||||
| ECG |
| anterior AMI, De Winter T waves, LAD stenosis | ||||
| ECG |
| LMCA Stenosis, ST elevation in aVR, Left Main Coronary Artery | ||||
| ECG |
| LMCA, Left Main Coronary Artery Occlusion, ST elevation in aVR | ||||
| ECG |
| VT, BCT, WCT, Brugada criteria, Verekie | ||||
| ECG |
| severe hypokalaemia, spironalactone, rhabdomyolysis, ECG, u wave, diabetic ketoacidosis | ||||
| ECG |
| pacing, pacemaker, post-op, Mobitz I, Wenckebach, AV block | ||||
| ECG |
| bidirectional ventricular tachycardia, Catecholaminergic Polymorphic Ventricular Tachycardia, CPVT, digoxin toxicity | ||||
| ECG |
| congenital, short QT syndrome, SQTS, AF, Atrial fibrillation | ||||
| ECG |
| RVOT, broad complex tachycardia, BCT, Right Ventricular Outflow Tract Tachycardia, VF, Arrest, Arrhythmogenic Right Ventricular Cardiomyopathy, ARVC | ||||
| ECG |
| NSTEMI, inverted U wave, | ||||
| ECG |
| tricyclic antidepressant, TCA, Doxepin, QRS broadening, cardiotoxic | ||||
| ECG |
| AIVR, Accelerated idioventricular rhythm, Isorhythmic AV dissociation, Sinus arrhythmia, idioventricular | ||||
| ECG |
| LAD, LBBB, High left ventricular voltage, HLVV, WPW, Broad Complex Tachycardia | ||||
| ECG |
| tachy-brady, AVNRT, flutter, polymorphic VT, VF, torsades de pointes, R on T, Cardioversion | ||||
| ECG |
| LBBB, Wellens, ECG, proximal LAD, occlusion, rate-dependent, inferior ischaemia | ||||
| ECG |
| SI QIII TIII, PE, PTE pulmonary embolism, PEA arrest, RBBB, LAD | ||||
| Cardiology |
| HOCM, STE, aVR, LMCA, torsades des pointes. TDP | ||||
| Cardiology |
| aortic arch, right sided, diverticulum of Kommerell | ||||
| Cardiology |
| IABP, CABG, shock, circulatory collapse | ||||
| Cardiology |
| electrical alternans, ECG, pulsus paradoxus | ||||
| Cardiology |
| Intra-aortic Balloon Pump, Waveform, dicrotic notch | ||||
| Cardiology |
| DeBakey, TAA, aortic dissection, CTA | ||||
| Cardiology |
| Tetraology of Fallot, BT shunt, Blalock-Tausig, ToF | ||||
| Cardiology |
| PVP, cement, embolus, Percutaneous Vertebroplasty | ||||
| Cardiology |
| Pulmonary Embolism, PTE, PE, McConnell, thrombolysis, echo | ||||
| Bone and Joint |
| Missed posterior shoulder dislocation | ||||
| Paediatrics |
| rash, neck nodule, Kawasaki | ||||
| Paediatrics |
| rash, fever, scarlet, strawberry, Group A Beta Haemolytic Streptococci (GABHS) | ||||
| Tropical Travel |
| diphtheria, pseudomembrance, grey tonsils, pseudomembrane, tonsillitis, diphtheria, Corynebacterium diphtheriae, gram-positive bacillus | ||||
| Urinalysis |
| purple, urine, indican, indican | ||||
| Urinalysis |
| brown, urine, rhabdomyolysis | ||||
| Urinalysis |
| green, urine, propofol, PRIS | ||||
| Urinalysis |
| green, urine | ||||
| Urinalysis |
| orange, urine | ||||
| Bone and Joint |
| Nail, trauma, hematoma, subungual, haematoma, nail-bed | ||||
| Bone and Joint |
| Extensor tendon, hand injury, extensor digiti minimi, | ||||
| Bone and Joint |
| Thumb, fracture, base, phalanx, metacarpal, Edward Hallaran Bennett, bipartate | ||||
| Paediatrics |
| Food allergy, enterocolitis, | ||||
| Bone and Joint |
| FOOSH, wrist fracture, FOOSH - 'fall onto outstretched hand', Barton fracture, John Rhea Barton | ||||
| Paediatrics |
| pulled elbow, nursemaid, hyperpronation | ||||
| Cardiology |
| Phlegmasia, dolens | ||||
| Cardiology |
| ICC, intercostal, intra-cardiac, iatrogenic | ||||
| Bone and Joint |
| Compartment syndrome, Volkmann, fasciotomy | ||||
| Bone and Joint |
| Ankle, compound, fracture, dislocation, Six Hour Golden Rule, saline, iodine | ||||
| ENT |
| retropharngeal abscess, posterior pharynx, mediastinitis, Lemierre syndrome, Fusobacterium necrophorum | ||||
| ENT |
| enlarged tonsils, pharyngitis, tonsillitis | ||||
| Toxicology | Colgout, colchicine, label, fenofibrate | |||||
| Tropical Travel | Mary Mallon, Salmonella typhi, typhoid, typhoid mary | |||||
| Tropical Travel | Dengue Fever, single-stranded RNA virus, Aedes, mosquito, Dengue Shock Syndrome (DSS), Dengue Haemorrhagic Fever (DHF) | |||||
| Tropical Travel | AIDS, Human immunodeficiency virus, lentivirus, anti-retroviral, | |||||
| Tropical Travel | tuberculosis | |||||
| Tropical Travel | Falciparum, Vivax, Ovale, Malariae, Knowlesi, Plasmodium | |||||
| Tropical Travel | cholera, gram-negative comma-shaped bacillus, rice water stool, John Snow Pump, V. cholerae, vibrio | |||||
| Tropical Travel | Entamoeba histolytica, protozoan parasite, Amoebic dysentery, Flask Shaped amoebic trophozoite, Bloody stool, | |||||
| Tropical Travel | shigellosis, Shigella, Enterotoxin, dysentery, | |||||
| Tropical Travel | Tetanus, Tetanispasmin, Clostridium tetani, lock jaw, Opisthotonus, Autonomic dysfunction, toxoid | |||||
| Tropical Travel | Rabies Immunoglobulin | |||||
| Tropical Travel | Koplik, measles, rash, rubeola, Morbilivirus, | |||||
| Trauma | permissive hypotension, MBA, MVA, widened mediastinum, pleural effusion, ICC | |||||
| Trauma | knife, penetrating chest wound | |||||
| Trauma | knife, penetrating chest wound | |||||
| Trauma | TBSA %, Burns Wound Assessment, Total Body Surface Area | |||||
| Trauma | Arterial pressure index (API), DPI (Doppler Pressure Index), Arterial Brachial Index or Ankle Brachial Index (ABI) | |||||
| Trauma | crush injury, degloving, deglove, amputation | |||||
| Trauma | hip dislocation, Allis reduction, pelvic fracture | |||||
| Trauma | Pelvis fracture, stabilization, stabilisation, | |||||
| Trauma | pelvic stabilization, Pelvis fracture, stabilisation, Pre-peritoneal packing | |||||
| Trauma | massive transfusion protocol, Recombinant Factor VIIa, Thromboelastography (TEG) | |||||
| Trauma | Critical bleeding, hemorrhagic shock, haemorrhagic shock, lethal triad, acute coagulopathy of trauma | |||||
| Trauma | penetrating abdominal trauma | |||||
| Trauma | ||||||
| Trauma | penetrating chest trauma wound, stab, | |||||
| Trauma | Right Main Bronchus, RMB, Tracheostomy, Tooth, foreign Body | |||||
| Trauma | Lobar collapse, aspiration, blood clot | |||||
| Trauma | ||||||
| Trauma | Traumatic rupture of the diaphragm with strangulation of viscera | |||||
| Trauma | eschar, burns, full thickness, | |||||
| Trauma | supine hypotension syndrome | |||||
| Trauma | ||||||
| Trauma | iPhone | |||||
| Trauma | oleoma, lipogranuloma, | |||||
| Trauma | oral commissure, lingual artery hemorrhage, | |||||
| Trauma | polymer fume fever, dielectric heating, super-heating, thermal injury | |||||
| Trauma | DRE, Digital rectal exam examination trauma | |||||
| Trauma | Injury Severity score, ISS, golden hour, seatbelt sign | |||||
| Trauma | primary secondary survey | |||||
| Trauma | extradural hemorrhage, EDH, Monro-Kellie | |||||
| Trauma | ||||||
| Trauma | ||||||
| Trauma | ||||||
| Trauma | ||||||
| Trauma | ||||||
| Trauma | GU, trauma, penis, penile, urethra, bladder, rupture | |||||
| Pulmonary | swine flu, pneumomediastinum, CXR | |||||
| Pulmonary | Thrombocytopenia, antiphospholipid syndrome | |||||
| Pulmonary | Hermann Boerhaave, Boerhaave syndrome, esophagus rupture, oesophagus | |||||
| Pulmonary | ||||||
| Pulmonary | pneumococcal pneumonia, HIV, bronchoscope, anatomy, RMB | |||||
| Pulmonary | subcutaneous emphysema, FLAAARDS, | |||||
| Pulmonary | respiratory acidosis, hypercapnoea | |||||
| Pulmonary | hypersensivity pneumonitis, diffuse alveolar haemorrhage, alveolar infiltrates | |||||
| Pulmonary | Lung collapse, recruitment maneuver, bronchoscopy | |||||
| Pulmonary | Vocal cord dysfunction, VCD, paradoxical vocal cord motion, PVCM, posterior chinking | |||||
| Pulmonary | pneumococcus, Streptococcus pneumoniae, penicillin-resistant | |||||
| Pulmonary | DOPES, | |||||
| Pulmonary | asthma | |||||
| Pulmonary | dyssynchrony, mechanical ventilation, PEEP, Plateau pressure | |||||
| Pulmonary | pneumomediastinum, tracheostomy, trachy, complication | |||||
| Pulmonary | PERC rule, D-Dimer, Pulmonary Embolism Rule-out Criteria, HAD CLOTS, | |||||
| Pulmonary | AMS, acute mountain sickness, high altitude, High-altitude cerebral edema, HACE, HAPE, High-altitude pulmonary edema | |||||
| Pulmonary | ||||||
| Resus | Pulseless electrical activity, PEA | |||||
| Resus | intraosseous access, EZ-IO, | |||||
| Resus | ||||||
| Resus | Rocuronium, suxamethonium, succinylcholine, non-depolarising muscle relaxant, sugammadex, safe apnoea time | |||||
| Resus | FEAST, trial, research, pediatric, fluid resuscitation | |||||
| Resus | ||||||
| Resus | ||||||
| Resus | ||||||
| Resus | ICC, intercostal | |||||
| Resus | Mechanical ventilation | |||||
| Oncology | SVC obstruction | |||||
| Oncology | Tumour lysis syndrome, Tumor lysis syndrome | |||||
| Oncology | lung metastases braine mets testicular cancer BEP chemotherapy, Cannonball metastases | |||||
| Oncology | re-expansion pulmonary oedema edema | |||||
| Metabolic | abdominal aortic aneurysm, AAA, rupture, CT, rhabdomyolysis, creatine kinase | |||||
| Metabolic | hypokalemia, hypokalaemia, periodic paralysis, u wave | |||||
| Metabolic | CATMUDPILES, OGRE, NAGMA, HAGMA, USED CARP, hyperchloraemic metabolic acidosis | |||||
| Metabolic | anion gap, pyroglutamic acidemia, HAGMA, high-anion gap, high anion, 5-oxoprolinemia, γ-glutamyl cycle, staph aureus, sepsis | |||||
| Metabolic | HAGMA, high-anion gap, high anion, hypernatraemia, hypernatremia | |||||
| Metabolic | hypokalaemia, hypokalemia, potassium, systemic bromism, coke, pepsi, coca-cola | |||||
| Metabolic | CATMUDPILES, renal failure, HAGMA, LTKR | |||||
| Metabolic | ||||||
| Metabolic | acute hepatitis, arterial blood gas, fulminant hepatic failure, lactic acidosis, lactic acidosis with hypoglycaemia, metabolic acidosis, metabolic muddle | |||||
| Metabolic | hyperammonaemia, hyperammonemia | |||||
| Metabolic | Hyponatraemia, hypertonic saline, ultramarathon, runner, EAH, pontine myelinoysis | |||||
| Metabolic | Hyponatraemia, hypertonic saline, pontine myelinoysis, Osmolality, desmopressin, SIADH, syndrome of inappropriate anti-diuretic hormone secretion | |||||
| Gastrointestinal | Appendagitis, Epiploic, Abdominal pain, CT abdomen | |||||
| Gastrointestinal | CT abdomen, Small bowel obstruction, SBO | |||||
| Gastrointestinal | cathine, cathione, khat, hepatitis, cathionine | |||||
| Gastrointestinal | rectal foreign body, FB | |||||
| Gastrointestinal | abdominal compartment syndrome, intra-abdominal pressure, intra-abdominal hypertension, IAH, ACS | |||||
| Hematology | fibrinolytic, VTE, Wells, PERC | |||||
| Hematology | factor VIIa, rFVIIa, novoseven | |||||
| Hematology | Critical Bleeding, Massive Transfusion, Tranexamic Acid, TxA, MTP | |||||
| Hematology | Dyshemoglobinemia, Acute myeloid leukemia, AML | |||||
| Immunological | angiodema, angioedema, lip sweliing | |||||
| Immunological | frusemide, furosemide, lasix, sulfa, | |||||
| Immunological | wegener, GPA, granulomatosis, palpable purpura | |||||
| Obstetric | amniotic fluid embolism, DIC, obstetric complication, disseminated intravascular coagulation, schistocytes, | |||||
| Microbial | CSF, Meningococcal meningitis, | |||||
| Microbial | fulminant bacterial pneumonia, septic shock, Pneumococcus, Streptococcus pyogenes, urinary pneumococcal antigen, | |||||
| Microbial | Legionella, community acquired pneumonia | |||||
| Microbial | Staphylococcal toxic shock syndrome, Toxic-shock syndrome | |||||
| Microbial | ||||||
| Microbial | ||||||
| Microbial | Norovirus | |||||
| Toxicology | Coma, similie, metaphor, flashcard, toxidromes, anticholinergic, cholinergic, PHAILS, OTIS CAMPBELL, PACED, FAST, COOLS, CT SCAN | |||||
| Neurology | HIV, Mass effect, CNS lesion, Brain lesion | |||||
| Neurology | pancoast, argyll robertson, holmes-adie, coma, pinpoint, pin-point, horner syndrome | |||||
| Neurology | rule of 4, rules of four, brainstem, weber syndrome, wallenberg | |||||
| Neurology | rule of 4, rules of four, brainstem, Nothnagel syndrome, benedikt, claude, | |||||
| Neurology | ||||||
| Neurology | ||||||
| Neurology | ||||||
| Neurology | Unilateral internuclear ophthalmoplegia, medial longitudinal fasciculus, MLF, INO, one-and-a-half syndrome | |||||
| Neurology | GSW, gunshot wound, bullet, TBI, Codman ICP monitor, Trans-cranial doppler, Near-infrared spectroscopy, NIRS, cerebral microdialysis catheter | |||||
| Neurology | BPPV, Benign Paroxysmal Positional Vertigo, Dix-Hallpike test, semont, epley, dix hallpike, brandt-daroff | |||||
| Neurology | Anti-NMDA Receptor Encephalitis, teratoma | |||||
| To err is human | cognitive error, bias, entrapment | |||||
| To err is human | rule of thumb, heuristic, satisficing, cognitive bias, metacognition | |||||
| To err is human | | Anchoring Bias, confirmation, satisficing, clustering bias | ||||
| Cardiology | ||||||
| Paediatric | pediatric |
LITFL Top 100 Self Assessment Quizzes
- - Google Chrome
Intended for healthcare professionals
- My email alerts
- BMA member login
- Username * Password * Forgot your log in details? Need to activate BMA Member Log In Log in via OpenAthens Log in via your institution

Search form
- Advanced search
- Search responses
- Search blogs
- How to write a medical...
How to write a medical case report
- Related content
- Peer review
- Seema Biswas , editor-in-chief, BMJ Case Reports, London, UK ,
- Oliver Jones , student editor, BMJ Case Reports, London, UK
Two BMJ Case Reports journal editors take you through the process
This article contains...
- Choosing the right patient
- Choosing the right message
- Before you begin - patient consent
- How to write your case report
- How to get published
During medical school, students often come across patients with a unique presentation, an unfamiliar response to treatment, or even an obscure disease. Writing a case report is an excellent way of documenting these findings for the wider medical community—sharing new knowledge that will lead to better and safer patient care.
For many medical students and junior doctors, a case report may be their first attempt at medical writing. A published case report will look impressive on your curriculum vitae, particularly if it is on a topic of your chosen specialty. Publication will be an advantage when applying for foundation year posts and specialty training, and many job applications have points allocated exclusively for publications in peer reviewed journals, including case reports.
The writing of a case report rests on skills that medical students acquire in their medical training, which they use throughout their postgraduate careers: these include history taking, interpretation of clinical signs and symptoms, interpretation of laboratory and imaging results, researching disease aetiology, reviewing medical evidence, and writing in a manner that clearly and effectively communicates with the reader.
If you are considering writing a case report, try to find a senior doctor who can be a supervising coauthor and help you decide whether you have a message worth writing about, that you have chosen the correct journal to submit to (considering the format that the journal requires), that the process is transparent and ethical at all times, and that your patient is not compromised in your writing. Indeed, try to include your patient in the process from the …
Log in using your username and password
BMA Member Log In
If you have a subscription to The BMJ, log in:
- Need to activate
- Log in via institution
- Log in via OpenAthens
Log in through your institution
Subscribe from £184 *.
Subscribe and get access to all BMJ articles, and much more.
* For online subscription
Access this article for 1 day for: £33 / $40 / €36 ( excludes VAT )
You can download a PDF version for your personal record.
Buy this article
Log in using your username and password
- Search More Search for this keyword Advanced search
- Latest content
- Current issue
- Write for Us
- BMJ Journals
You are here
- Volume 21, Issue 1
- What is a case study?
- Article Text
- Article info
- Citation Tools
- Rapid Responses
- Article metrics
- Roberta Heale 1 ,
- Alison Twycross 2
- 1 School of Nursing , Laurentian University , Sudbury , Ontario , Canada
- 2 School of Health and Social Care , London South Bank University , London , UK
- Correspondence to Dr Roberta Heale, School of Nursing, Laurentian University, Sudbury, ON P3E2C6, Canada; rheale{at}laurentian.ca
https://doi.org/10.1136/eb-2017-102845
Statistics from Altmetric.com
Request permissions.
If you wish to reuse any or all of this article please use the link below which will take you to the Copyright Clearance Center’s RightsLink service. You will be able to get a quick price and instant permission to reuse the content in many different ways.
What is it?
Case study is a research methodology, typically seen in social and life sciences. There is no one definition of case study research. 1 However, very simply… ‘a case study can be defined as an intensive study about a person, a group of people or a unit, which is aimed to generalize over several units’. 1 A case study has also been described as an intensive, systematic investigation of a single individual, group, community or some other unit in which the researcher examines in-depth data relating to several variables. 2
Often there are several similar cases to consider such as educational or social service programmes that are delivered from a number of locations. Although similar, they are complex and have unique features. In these circumstances, the evaluation of several, similar cases will provide a better answer to a research question than if only one case is examined, hence the multiple-case study. Stake asserts that the cases are grouped and viewed as one entity, called the quintain . 6 ‘We study what is similar and different about the cases to understand the quintain better’. 6
The steps when using case study methodology are the same as for other types of research. 6 The first step is defining the single case or identifying a group of similar cases that can then be incorporated into a multiple-case study. A search to determine what is known about the case(s) is typically conducted. This may include a review of the literature, grey literature, media, reports and more, which serves to establish a basic understanding of the cases and informs the development of research questions. Data in case studies are often, but not exclusively, qualitative in nature. In multiple-case studies, analysis within cases and across cases is conducted. Themes arise from the analyses and assertions about the cases as a whole, or the quintain, emerge. 6
Benefits and limitations of case studies
If a researcher wants to study a specific phenomenon arising from a particular entity, then a single-case study is warranted and will allow for a in-depth understanding of the single phenomenon and, as discussed above, would involve collecting several different types of data. This is illustrated in example 1 below.
Using a multiple-case research study allows for a more in-depth understanding of the cases as a unit, through comparison of similarities and differences of the individual cases embedded within the quintain. Evidence arising from multiple-case studies is often stronger and more reliable than from single-case research. Multiple-case studies allow for more comprehensive exploration of research questions and theory development. 6
Despite the advantages of case studies, there are limitations. The sheer volume of data is difficult to organise and data analysis and integration strategies need to be carefully thought through. There is also sometimes a temptation to veer away from the research focus. 2 Reporting of findings from multiple-case research studies is also challenging at times, 1 particularly in relation to the word limits for some journal papers.
Examples of case studies
Example 1: nurses’ paediatric pain management practices.
One of the authors of this paper (AT) has used a case study approach to explore nurses’ paediatric pain management practices. This involved collecting several datasets:
Observational data to gain a picture about actual pain management practices.
Questionnaire data about nurses’ knowledge about paediatric pain management practices and how well they felt they managed pain in children.
Questionnaire data about how critical nurses perceived pain management tasks to be.
These datasets were analysed separately and then compared 7–9 and demonstrated that nurses’ level of theoretical did not impact on the quality of their pain management practices. 7 Nor did individual nurse’s perceptions of how critical a task was effect the likelihood of them carrying out this task in practice. 8 There was also a difference in self-reported and observed practices 9 ; actual (observed) practices did not confirm to best practice guidelines, whereas self-reported practices tended to.
Example 2: quality of care for complex patients at Nurse Practitioner-Led Clinics (NPLCs)
The other author of this paper (RH) has conducted a multiple-case study to determine the quality of care for patients with complex clinical presentations in NPLCs in Ontario, Canada. 10 Five NPLCs served as individual cases that, together, represented the quatrain. Three types of data were collected including:
Review of documentation related to the NPLC model (media, annual reports, research articles, grey literature and regulatory legislation).
Interviews with nurse practitioners (NPs) practising at the five NPLCs to determine their perceptions of the impact of the NPLC model on the quality of care provided to patients with multimorbidity.
Chart audits conducted at the five NPLCs to determine the extent to which evidence-based guidelines were followed for patients with diabetes and at least one other chronic condition.
The three sources of data collected from the five NPLCs were analysed and themes arose related to the quality of care for complex patients at NPLCs. The multiple-case study confirmed that nurse practitioners are the primary care providers at the NPLCs, and this positively impacts the quality of care for patients with multimorbidity. Healthcare policy, such as lack of an increase in salary for NPs for 10 years, has resulted in issues in recruitment and retention of NPs at NPLCs. This, along with insufficient resources in the communities where NPLCs are located and high patient vulnerability at NPLCs, have a negative impact on the quality of care. 10
These examples illustrate how collecting data about a single case or multiple cases helps us to better understand the phenomenon in question. Case study methodology serves to provide a framework for evaluation and analysis of complex issues. It shines a light on the holistic nature of nursing practice and offers a perspective that informs improved patient care.
- Gustafsson J
- Calanzaro M
- Sandelowski M
Competing interests None declared.
Provenance and peer review Commissioned; internally peer reviewed.
Read the full text or download the PDF:
We have a new app!
Take the Access library with you wherever you go—easy access to books, videos, images, podcasts, personalized features, and more.
Download the Access App here: iOS and Android . Learn more here!
- Remote Access
- Save figures into PowerPoint
- Download tables as PDFs
- Biochemistry
- Microbiology
- Neuroscience
- Pharmacology
- Anesthesiology
- Emergency Medicine
- Family Medicine
- Internal Medicine
- Clinical Neuroanatomy Cases
- Clinical Nutrition Cases
- Acid-Base Disturbance Cases
- Chest Imaging Cases
- Family Medicine Board Review
- Fluid/Electrolyte and Acid-Base Cases
- G&G Pharm Cases
- Harrison’s Visual Case Challenge
- Internal Medicine Cases
- Medical Microbiology Cases
- Neuroanatomy Cases
- Pathophysiology Cases
- Principles of Rehabilitation Medicine Case-Based Board Review
- Browse by System
- Internal Medicine Med-Peds Pediatrics
- Browse by Curriculum
| Below is a limited selection of Case Files content on AccessMedicine. Click here to browse the full set of Case Files on , which requires a separate institutional purchase. |
Basic Science

| Date | Medical Condition | Therapeutic Goals | Drug-Therapy Problem | Recommendations and Interventions | Monitoring Parameters, Desired Endpoints, and Frequency | Follow-up plan |
|---|
- Research Guides
- University Libraries
- Medical Sciences Guides
Medical Residents & Fellows--Library Resources
- Case Studies, Videos, Images
- General Resources
Multimedia/Teaching Resources
Case Studies
Multimedia resources - general, access medicine videos, specialty videos, power point presentations, audio resources.
- Departments/Specialties
- Drugs/Pharmacy
- Research/Scholarly/Citation Management
- Integrative Medicine
- Step 3 Review
- Physician Wellness Resources
- Author Profile
- Access Case Files
- Case Files Collection Offers Case Files content in an interactive format. Helps students learn and apply basic science and clinical medicine concepts in the context of realistic patient cases. Features the complete collection of basic science, clinical medicine, and post-graduate level cases from 23 Case Files series books, and the personalized functionality to let users mark their progress through cases.
- Cases on Diversity, Equity, and Inclusion for the Health Professions Educator, 1e, 2023
- Kumar & Clark's Cases in Clinical Medicine, 4e, 2021
- Lippincott Health Library Clerkship/Clinical Rotations Cases Case studies in family medicine, internal medicine, OB/GYN, pediatrics, psychiatry, and surgery. Can be broken down into any one of these topics.
- NEJM Clinical Cases Resource Center The Clinical Cases Resource Center on NEJM.org offers a wealth of interactive case studies and educational resources formulated specifically for teaching, continuous learning, and professional development.
- Bates' Visual Guide to Physical Examination, 2023 (videos) A companion website to Bates' Guide to Physical Examination and History Taking. Videos provide an evidence-based approach for physical examination techniques. Includes information on abnormalities and differential diagnoses. Categories of videos include Physical Examination, Communication & Interpersonal Skills, and OSCE Clinical Skills.
- ClinicalKey Hundreds of images and videos to select from.
- Instructor's Resources - Access Medicine
- JoVE Science Education videos Videos provide basic medical instruction.
- New England Journal of Medicine Multimedia
- Ovid Multimedia
- Procedural Videos - AccessMedicine
- Procedure Videos (formerly Procedures Consult) This link opens in a new window A procedure reference tool that offers access to complete details on how to prepare for, perform and follow up on the most common procedures. Many are demonstrated with videos. To set up a Procedure Video group to create and manage assignments, please contact the Medical Sciences Library ([email protected] or 979-845-7428).
- Access Medicine Contains a wide variety of medical videos, Including many Procedural Videos for Emergency Medicine/Intensive Care, Pediatrics, Gastroenterology/Hepatology, Gynecology, Neurology, Orthopedics, Dermatology, and Pulmonary Medicine
- Access Emergency Medicine These videos are part of a comprehensive set of study materials and review questions for the emergency medicine residency. Each module consists of 1 week of material. There are two rounds for each topic; Round 1 is designed for years 1-2 of residency, while Round 2 is designed for years 2-4 of residency. Completion of this curriculum is designed to give the residents a thorough multi-modality review of EM major topics.
- Access Neurology Contains videos on EEG, Movement Disorders, and Physical Exam. Also includes an interactive neuroanatomy atlas, lectures and animated tours of the brain and nervous system.
- Access Pediatrics Contains a comprehensive list of of videos for pediatrics practice.
- Access Pharmacy Contains videos and animations on pharmacology, arranged by organ systems. Harrison's Pathophysiology Animations are also available.
- Access Surgery Contains a large variety of procedural surgical videos arranged by organ system. Also contains videos from Zollinger's Video Atlas of Surgical Operations.
- Andreoli and Carpenter's Cecil Essentials of Medicine, 10e, 2022 Link to 20 videos by clicking on "Videos" button under "Table of Contents".
- Atlas of Cardiac Surgical Techniques, 2e, 2019 Link to 22 videos by clicking on "Videos" button under "Table of Contents".
- The Atlas of Emergency Medicine Videos, 5e, 2021 Virtual and ultrasound videos.
- Blumgart's Video Atlas : Liver, Biliary, & Pancreatic Surgery, 2e, 2021 Link to 121 videos by clicking on "Videos" button under "Table of Contents". Virtual surgical procedures are sometimes later in the videos.
- Counseling and Therapy in Video Over 500 videos allow students and scholars to see, experience, and study counseling. Many videos include supplementary materials to aid in classroom discussions and assignments.
- Green's Operative Hand Surgery, 8e, 2022 Link to 59 videos by clicking on "Videos" button under "Table of Contents".
- Hagberg and Benumof's Airway Management, 5e, 2023 Link to 30 videos by clicking on "Videos" button under "Table of Contents"
- Harrison's Principles of Internal Medicine, 21e, 2022 Virtual and ultrasound videos.
- Insall & Scott Surgery of the Knee, 6e, 2018 Link to 256 videos by clicking on "Videos" button under "Table of Contents".
- Morrey's The Elbow and Its Disorders, 5e, 2018 Link to 42 videos by clicking on "Videos" button under "Table of Contents".
- Roberts and Hedges' Clinical Procedures in Emergency Medicine and Acute Care, 7e, 2019 Over 200 videos on emergency and critical care.
- Textbook of Critical Care, 8e, 2024 Link to 51 videos by clicking on "Videos" button under "Table of Contents".
- Access Medicine Instructor's Resources
- Access Medicine Run the List Podcast Discussions on key principles of internal medicine, around board-style case vignettes from the acclaimed Harrison’s Self Assessment and Board Review.
- Access Medicine Symptom to Diagnosis Podcast A case-based discussion of signs, symptoms and diagnostics tests to improve clinical reasoning and evidence-based practice.
- Case Files Podcasts Features a series of conversational, informative podcasts correlated with the cases featured in Case Files Collection
- Harrison's Podcasts
- The Symptom to Diagnosis Podcasts A case-based discussion of signs, symptoms and diagnostics tests to improve clinical reasoning and evidence-based practice.
- USMLE Images for the Boards : a comprehensive image-based review, 1e, 2013
- VisualDX This link opens in a new window A Web and mobile enabled visual clinical decision support system used to build a differential by selecting symptoms and other parameters. View descriptions of possible matches, critical alerts, and images to support selection of the best diagnosis.
- Assessing Competence in Medicine and Other Health Professions, 1e, 2018
- Cinemeducation : a comprehensive guide to using film in medical education Paper copy available in MSL@RoundRock W 418 C5749 2005
- Curriculum Development for Medical Education : a six-step approach, 4e, 2022
- Evidence-based medicine : how to practice and teach EBM, 5e, 2019
- Healthy Presentations : how to craft exceptional lectures in medicine, the health professions, and the biomedical sciences, 1e, 2021
- The Master Adaptive Learner : from the AMA MedEd Innovation Series, 1e, 2020 Call Number: W 18 M4231 2020 Print copies available in MSL@Bryan, MSL@College Station, MSL@Dallas, and MSL@Houston
- A Primer for the Clinician Educator : supporting excellence and promoting change through storytelling, 1e, 2023
- Remediation Case Studies : helping struggling medical learners, 1e, 2021 Print copy available in MSL@College Station W 18 G934rc 2021
- Successful Doctoral Training in Nursing and Health Sciences : a guide for supervisors, students and advisors, 1e, 2022
- Survey Methods for Medical and Health Professions Education : a six-step approach, 1e, 2022 Print copy available in MSL@College Station W 20.5 S9631 2022
- Teaching Evidence-Based Medicine : a toolkit for educators, 1e, 2023
- Teaching Writing in the Health Professions : perspectives, problems, and practices, 1e, 2021
- Written Assessment in Medical Education, 1e, 2023
- << Previous: General Resources
- Next: Departments/Specialties >>
- Last Updated: Jun 17, 2024 8:43 AM
- URL: https://tamu.libguides.com/residents
- Open access
- Published: 20 June 2024
Circulating small extracellular vesicles in Alzheimer’s disease: a case–control study of neuro-inflammation and synaptic dysfunction
- Rishabh Singh 1 ,
- Sanskriti Rai 1 ,
- Prahalad Singh Bharti 1 ,
- Sadaqa Zehra 1 ,
- Priya Kumari Gorai 2 ,
- Gyan Prakash Modi 3 ,
- Neerja Rani 2 ,
- Kapil Dev 4 ,
- Krishna Kishore Inampudi 1 ,
- Vishnu V. Y. 5 ,
- Prasun Chatterjee 6 ,
- Fredrik Nikolajeff 7 &
- Saroj Kumar 1 , 7
BMC Medicine volume 22 , Article number: 254 ( 2024 ) Cite this article
194 Accesses
10 Altmetric
Metrics details
Alzheimer’s disease (AD) is a neurodegenerative disease characterized by Aβ plaques and neurofibrillary tangles. Chronic inflammation and synaptic dysfunction lead to disease progression and cognitive decline. Small extracellular vesicles (sEVs) are implicated in AD progression by facilitating the spread of pathological proteins and inflammatory cytokines. This study investigates synaptic dysfunction and neuroinflammation protein markers in plasma-derived sEVs (PsEVs), their association with Amyloid-β and tau pathologies, and their correlation with AD progression.
A total of 90 [AD = 35, mild cognitive impairment (MCI) = 25, and healthy age-matched controls (AMC) = 30] participants were recruited. PsEVs were isolated using a chemical precipitation method, and their morphology was characterized by transmission electron microscopy. Using nanoparticle tracking analysis, the size and concentration of PsEVs were determined. Antibody-based validation of PsEVs was done using CD63, CD81, TSG101, and L1CAM antibodies. Synaptic dysfunction and neuroinflammation were evaluated with synaptophysin, TNF-α, IL-1β, and GFAP antibodies. AD-specific markers, amyloid-β (1–42), and p-Tau were examined within PsEVs using Western blot and ELISA.
Our findings reveal higher concentrations of PsEVs in AD and MCI compared to AMC ( p < 0.0001). Amyloid-β (1–42) expression within PsEVs is significantly elevated in MCI and AD compared to AMC. We could also differentiate between the amyloid-β (1–42) expression in AD and MCI. Similarly, PsEVs-derived p-Tau exhibited elevated expression in MCI compared with AMC, which is further increased in AD. Synaptophysin exhibited downregulated expression in PsEVs from MCI to AD ( p = 0.047) compared to AMC, whereas IL-1β, TNF-α, and GFAP showed increased expression in MCI and AD compared to AMC. The correlation between the neuropsychological tests and PsEVs-derived proteins (which included markers for synaptic integrity, neuroinflammation, and disease pathology) was also performed in our study. The increased number of PsEVs correlates with disease pathological markers, synaptic dysfunction, and neuroinflammation.
Conclusions
Elevated PsEVs, upregulated amyloid-β (1–42), and p-Tau expression show high diagnostic accuracy in AD. The downregulated synaptophysin expression and upregulated neuroinflammatory markers in AD and MCI patients suggest potential synaptic degeneration and neuroinflammation. These findings support the potential of PsEV-associated biomarkers for AD diagnosis and highlight synaptic dysfunction and neuroinflammation in disease progression.
Peer Review reports
The progressive neurodegenerative condition known as Alzheimer’s disease (AD) is characterized by cognitive decline as a result of the formation of amyloid-β (Aβ) plaques, neurofibrillary tangles (NFTs), and chronic neuroinflammation that leads to neurodegeneration [ 1 , 2 , 3 ]. Synapse loss is a crucial pathophysiological event in disease progression, and synaptic proteins have been extensively studied due to earlier perturbations [ 4 , 5 ]. The pathological hallmark of AD, amyloid-β plaques, originates from the imprecise cleavage of the amyloid precursor protein (APP) by β-secretase (BACE1) and γ-secretase generating amyloid-β peptide forms [ 6 , 7 , 8 , 9 ]. Primary amyloid-β peptide forms are Aβ40 and Aβ42, where the majority of the amyloid-β plaques in AD brains are composed of Aβ42 [ 10 ]. Many point mutations in APP and γ-secretase cause familial early-onset AD, favoring Aβ42 formation, causing amyloid-β peptides prone to aggregate as fibrils and plaques [ 9 , 11 , 12 , 13 , 14 ]. Hyperphosphorylation of tau causes the formation of NFTs. The combined effect of accumulation of NFTs, amyloid-β fibrils, and plaques leads to neuronal function loss and cell death [ 15 , 16 ]. Aβ plaques activate immune receptors on microglia, thereby releasing pro-inflammatory cytokines and chemokines that mediate neuroinflammation, which, if it reaches a chronic level, causes damage to brain cells, including axonal demyelination and synaptic pruning [ 17 , 18 , 19 , 20 , 21 , 22 , 23 ]. In addition to these, other proteins, including the neurofilament light (NFL) protein, glial fibrillary acidic protein (GFAP), and synaptic proteins, have also been identified as AD biomarkers [ 24 , 25 , 26 , 27 , 28 ]. Understanding the intricate dynamics of AD in terms of its varied pathophysiological manifestations, such as neuroinflammation, synaptic loss, and proteinopathy, is essential for developing potential therapeutic interventions for AD and biomarker discovery. In clinical practice, cognitive assessment tools such as the Addenbrooke’s Cognitive Examination (ACE-III) and Mini-Mental State Examination (MMSE) are used to diagnose AD. These tools evaluate verbal fluency and temporal orientation, although results may be influenced by subject bias [ 29 , 30 , 31 ].
In recent years, small extracellular vesicles (sEVs) or exosomes have been acknowledged as crucial mediators of communication and signaling within the body, contributing significantly to the transmission of cellular cargo in various health and disease states. They also play a notable role in disseminating protein aggregates associated with neurodegenerative diseases [ 32 ]. sEVs are bi-layered membrane vesicles that have a heterogeneous group of (< 200 nm in diameter) that are found in different human body fluids, including blood, urine, saliva, and ascites, and that are actively released by all cell types [ 33 , 34 , 35 ]. For their functions in various physiological and pathological circumstances, sEVs are the most extensively researched type of EV [ 36 , 37 , 38 ]. sEVs exchange information between cells by transferring bioactive components (nucleic acids and proteins) [ 39 ]. As the sEVs’ composition bears the molecular signature of the secreting cell and bears an intrinsic property of transversing the blood–brain barrier (BBB) in both directions [ 40 , 41 ], they are a target of constant research in neurodegenerative disease. Furthermore, sEVs released by neuronal cells are crucial in transmitting signals to other nerve cells, influencing central nervous system (CNS) development, synaptic activity regulation, and nerve injury regeneration. Moreover, sEVs exhibit a dual function in neurodegenerative processes, as sEVs not only play an essential role in clearing misfolded proteins, thereby exerting detoxifying effects and providing neuroprotection [ 42 ]. On the other hand, they also have the potential to participate in the propagation and aggregation of misfolded proteins, particularly implicated in the pathological spread of Tau aggregates as indicated by both in vitro and in vivo studies [ 43 ]. As a protective mechanism, astrocytes (most abundant glial cells) accumulate at the locations where Aβ peptides are deposited, internalizing and breaking down aggregated peptides [ 44 ]. However, severe endosomal–lysosomal abnormalities arise in astrocytes when a significantly large amount of Aβ accumulates within astrocytes for a prolonged period without degradation [ 45 , 46 ]. Astrocytes then release engulfed amyloid-β (1-42) protofibrils through exosomes, leading to severe neurotoxicity to neighboring neurons [ 44 ]. Additionally, it has been found that the release of amyloid-β by microglia in association with large extracellular vesicles (Aβ-lEVs) damages synaptic plasticity and modifies the architecture of the dendritic spine [ 47 ]. Thus, sEVs can be a compelling subject for the investigation to understand AD’s inflammation and synaptic dysfunction [ 48 , 49 , 50 , 51 , 52 ].
In this study, we reported that protein levels are associated with AD pathology, neuroinflammation, and synaptic dysfunction in plasma-derived small extracellular vesicles (PsEVs). Our objective was to understand the pathophysiological process, neuroinflammation, synaptic dysfunction, and Aβ pathology through sEVs. Our study revealed a significant correlation between the concentration of cargo proteins derived from PsEVs and clinical diagnosis concerning ACE-III and MMSE scores. Furthermore, the levels of these studied proteins within PsEVs could differentiate between patients with MCI and AD. Thus, our study sheds light on the potential of PsEVs in understanding AD dynamics and offers insights into the underlying mechanisms of disease progression.
Subject recruitment
A total of n = 35 AD patients and n = 25 subjects with MCI were recruited from the Memory Clinic, Department of Geriatrics, All India Institute of Medical Sciences, New Delhi, India. Additionally, n = 30 healthy AMC (volunteers) were recruited. The inclusion criteria were as follows: a clinical diagnosis of MCI and AD patients using ACE-III and MMSE tests. The exclusion criteria encompass medical conditions such as cancer, autoimmune disorders, liver disease, hematological disorders, or stroke, as well as psychiatric conditions, substance abuse, or any impediment to participation. Controls were healthy, age-matched adults without neurological symptoms. AMC was 60–71, MCI was 65–79, and AD was 70–80 years of age range (Table 1 ). Neuropsychological scores, viz., ACE-III and MMSE, were recorded before subject selection.
Study ethical approval
The institutional ethics committee of All India Institute of Medical Sciences, New Delhi, India, granted the study ethical permission. The study has been granted the ethical approval number IECPG-670/25.08.2022. Following the acquisition of the written informed consent, all participants were enrolled.
Sample collection
One milliliter of blood was drawn from each participant using venipuncture, and blood collection vials were kept on ice during collection. The blood was centrifuged at 1700 g for 20 min at 4 °C to remove the cells, and the straw-colored plasma was collected. It was further clarified by centrifuging for 30 mi at 4 °C at 10,000 g. Finally, cleared plasma was stored at − 80 °C until further use. The samples were used for the downstream experiment after being thawed on ice and centrifuged at 10,000 g.
Isolation of PsEVs
The PsEVs were extracted by chemical-based precipitation from the plasma samples of AD patients, MCI patients, and AMC, as discussed previously [ 53 , 54 ]. In brief, 180 μL of plasma sample was used and filtered with 0.22 μm filter (SFNY25R, Axiva), followed by overnight incubation with the chemical precipitant (14% polyethylene glycol 6000) (807,491, Sigma). The samples underwent an hour-long, 13,000 g centrifugation at 4 °C the next day. Before being resuspended in 200 μL of 1X PBS (ML116-500ML, HiMedia), the pellet was first cleaned twice with 1X PBS. Before downstream experiments, the sEVs-enriched fraction was further filtered through a 100-kDa filter (UFC5100, Millipore).
Nanoparticle tracking analysis (NTA)
5000-fold dilution in 1X-PBS buffer was used for the NTA of PsEVs. In the ZetaView Twin system (Particle Metrix, Germany) sample chamber, 1 mL of diluted PsEVs sample was introduced. The following parameters were used throughout three cycles of scanning 11 cell locations each, and 60 frames per position were collected (video setting: high, focus: autofocus, shutter: 150, 488 nm internal laser, camera sensitivity: 80, cell temperature: 25 °C. CMOS cameras were used for recording, and the built-in ZetaView Software 8.05.12 (Particle Metrix, Germany) was used to analyze: 10 nm as minimum particle size, 1000 nm as maximum particle size, and 30 minimum particle brightness.
Transmission electron microscopy for morphological characterization
Transmission electron microscopy was employed to investigate PsEVs’ ultrastructural morphology. The resultant PsEVs pellet was diluted with PBS using 0.1 M phosphate buffer (pH 7.4). A carbon-coated copper grid of 300 mesh (01843, Ted Pella) was used to adsorb the separated PsEVs at room temperature for 30 min. After blot-drying, the adsorbed grids were dyed. For 10 s, 2% aqueous uranyl acetate solution (81,405, SRL Chem) as negative staining. After blotting the grids, they were inspected using a Talos S transmission electron microscope (ThermoScientific, USA).
Western blot
Based on the initial volume of biofluid input, all samples were normalized, i.e., 180 μL and the sample loading dye (2 × Laemmle Sample buffer) was mixed with PsEVs sample, and 20 μL equal volume was loaded to run on an 8–12% SDS PAGE [ 53 , 55 ]. After the completion of SDS-PAGE, protein from the gel was subjected to the Wet transfer onto the PVDF membrane of 0.22 μm (1,620,177, BioRad). The membrane-blocking with 3% bovine serum albumin (BSA) (D0024, BioBasic) in Tris (TB0194, BioBasic) base saline containing 0.1% of Tween 20 (65,296, SRL Chem) (TBST) using the BioRad Western blotting apparatus (BioRad, USA). Following this, overnight incubation of primary antibodies of CD63 (10628D, Invitrogen), CD81 (PA5-86,534, Invitrogen), TSG101 (MA1-23,296, Invitrogen), L1CAM (MA1-46,045, Invitrogen), synaptophysin (ADI-VAM-SV011-D, Enzo life sciences), GFAP (A19058, Abclonal), amyloid-β (1–42) oligomer (AHB0052, Invitrogen), phospho-Tau (s396) (35–5300, Invitrogen), interleukin 1β (IL-1β) (PA5-95,455, Invitrogen), tumor necrosis factor α (TNF-α) (E-AB-33121, Elabscience), and β-actin (AM4302, Invitrogen) were done at 4 °C. The membranes were washed with TBST buffer four times before at RT incubating with HRP-conjugated secondary antibodies, anti-rabbit (AB6721, Abcam), anti-mouse (31,430, Invitrogen). The Femto LUCENT™ PLUS-HRP kit (AD0023, GBiosciences) was used to develop the blot for visualizing the protein bands utilizing the method of enhanced chemiluminescence.
Enzyme-linked Immunosorbent Assay (ELISA)
According to the previous protocol, ELISA was carried out. [ 53 ]. PsEV samples were subjected to freeze–thaw cycles; next, PsEVs were ultrasonicated for two minutes, with a 30-s on-and-off cycle, at an amplitude of 25. Following this, they underwent a 10-min centrifugation at 10,000 g, at 4 °C, and the obtained supernatant was used. The samples were kept at 37 °C before loading into the ELISA plates. The bicinchoninic acid (BCA) protein assay kit (23,225, ThermoFisher Scientific) was used to quantify the total protein concentration using BSA (D0024, BioBasic) as a reference. The ELISA kit was used to detect the presence of protein in 100 μL of PsEV sample are as follows: amyloid-β (1–42) (E-EL-H0543, ELabsciences), p-Tau (s-396) (E-EL-H5314, ELabsciences), IL-1β (ITLK01270, GBiosciences), TNF-α (ITLK01190, GBiosciences), GFAP (E-EL-H6093, ELabsciences), and synaptophysin (E-EL-H2014, ELabsciences). The manufacturer’s instructions were followed for every step of the process. A 96-well microplate spectrophotometer (SpectraMax i3x Multi-Mode Microplate Reader, Molecular devices) was used to measure the absorbance at 450 nm.
Data and statistical analysis
The mean age values, ACE-III score, and MMSE score were ascertained using descriptive statistical analysis Table 1 . GraphPad Prism 8.0 was used for statistical data analysis, including NTA concentration, Western blotting densitometric analysis, and ELISA. Unpaired student t -test and ANOVA were used for group analysis, and statistical significance was determined. p < 0.05 was used to assess significance. The Image J software (NIH, USA) was used for the densitometry analysis. The receiver operating characteristic (ROC) curve was used to analyze the efficiency of distinguishing the case from controls. Correlation analysis was conducted between the concentration of PsEVs and the levels of ELISA proteins, including amyloid-β (1–42), p-Tau, IL-1β, TNF-α, GFAP, and synaptophysin, and additionally between the PsEVs-derived levels of amyloid-β (1–42) β1-42, p-Tau, IL-1β, TNF-α, GFAP, and synaptophysin with ACE-III and MMSE values. ROC curve is a probability curve utilized to assess the accuracy of a test. The test’s ability to distinguish between groups is indicated by the area under the curve (AUC), which acts as a quantitative measure of separability. An outstanding test typically exhibits an AUC close to 1, signifying a high level of separability. Conversely, a subpar test tends to have an AUC closer to 0, indicating a poor ability to distinguish between the two classes.
Characterization and validation of isolated sEVs
PsEVs were isolated, characterized, and validated following Minimal Information for Studies of Extracellular Vesicles (MISEV) 2018 guidelines, which suggest a protocol for documenting work specifically with extracellular vesicles [ 56 ]. PsEVs from AMC, MCI, and AD subjects were morphologically characterized by transmission electron microscopy, and spherical lipid bi-layered vesicles were observed in the size range of sEVs (Fig. 1 A–C). In Fig. 1 D–F, the size distribution and concentration of PsEVs were observed in the size range of 30–200 nm in diameter by NTA, which is within the sEVs’ size range. The mean concentration of PsEVs in AMC, MCI, and AD patients were 5.12E + 10, 2.6E + 11, and 3.13E + 11 particle/ml, respectively, with higher concentrations of PsEVs in MCI and AD than in AMC ( p < 0.0001) (Fig. 1 G). To differentiate AD from AMC, ROC and AUC analyses were performed where the AUC = 0.9748, with a sensitivity of 97.14% and specificity of 70.01% (Fig. 1 H), while in AMC versus MCI, AUC = 0.987, sensitivity of 96% and specificity of 86.67% (Fig. 1 I). Furthermore, we could also differentiate between MCI and AD, AUC = 0.629, sensitivity of 60%, and specificity of 56% (Fig. 1 J). Validation of PsEVs was done using immunoblot for sEVs-specific markers (CD63, CD81, and TSG101), which showed a significant increase in expressions in MCI and AD than in AMC (CD63, p = 0.0489, 0.0478 (Additional File 1 : Fig. S1); CD81, p = 0.0172, 0.0133 (Additional File 1 : Fig. S2); TSG101 p = 0.0240, 0.0329 (Additional File 1 : Fig. S3)) for AD and MCI respectively (Fig. 2 A–D). Additionally, higher L1CAM (neuron-associated marker) expression was observed in MCI ( p = 0.0100) and AD ( p = 0.0184) (Additional File 1 : Fig. S4) compared to AMC (Fig. 2 E). All densitometric values were normalized against β-actin, which was used as a loading control (Additional File 1 : Fig. S7).
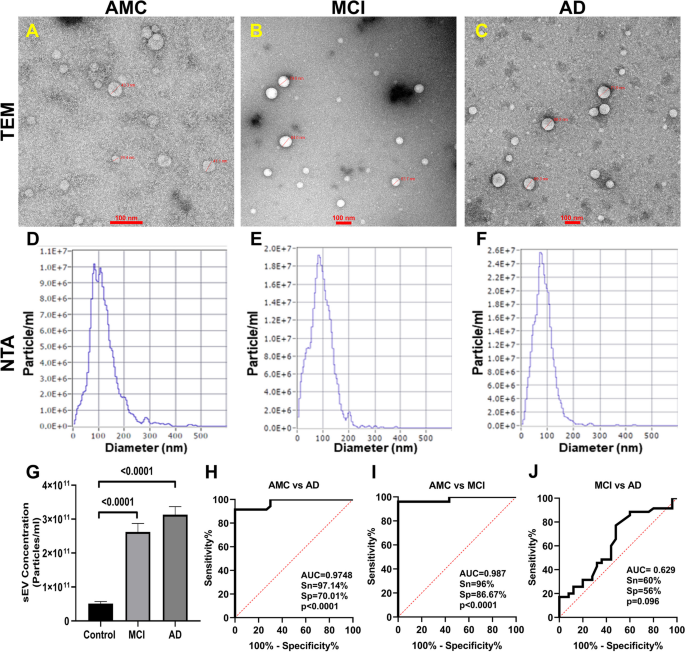
Isolation and analysis of PsEVs. The isolated PsEV morphology characterize by transmission electron microscopy from age-matched healthy controls (AMC) ( A ), mild-cognitive impairment (MCI) patients ( B ), and Alzheimer’s disease (AD) ( C ). The size distribution of PsEVs subpopulation (nm) versus the concentration (particle/ml) in AMC ( D ), individuals with MCI ( E ), and AD ( F ). Comparison of the sEVs concentration of AD, MCI, and AMC patients ( G ). Receiver operating characteristic (ROC) curve of PsEVs concentration in AMC v/s AD ( H ), AMC v/s MCI ( I ), and MCI v/s AD ( J ) (scale bar 100 nm)
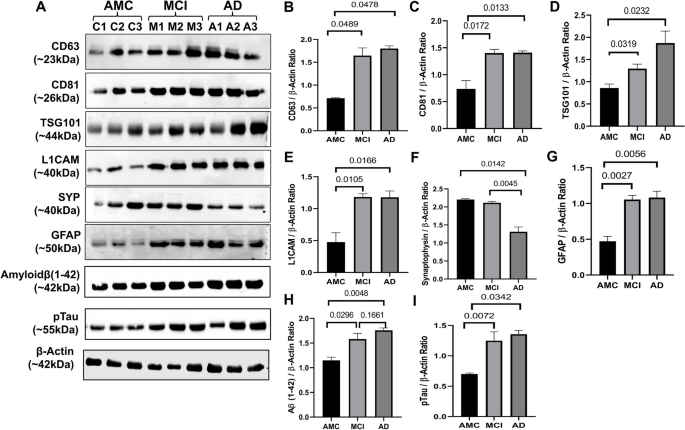
Validation of PsEVs expression analysis of different markers in PsEVs in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease patients (AD) ( A ). Densitometric analysis of CD63 ( B ), densitometric analysis of CD81 ( C ), densitometric analysis of TSG101 ( D ), densitometric analysis of L1CAM ( E ), densitometric analysis of synaptophysin ( F ), densitometric analysis of GFAP ( G ), and densitometric analysis of amyloid-β (1–42) oligomer ( H ). All densitometric values were normalized against β-actin
Differential expression of amyloid-β (1–42), p-Tau, synaptophysin, GFAP markers, and levels of IL-1β and TNF-α in PsEVs
Using ELISA, we measured levels of amyloid-β (1–42) and p-Tau in PsEVs from AMC, MCI, and AD patients. The significant increase of amyloid-β (1–42) and p-Tau among the groups (Fig. 3 A–H). Amyloid-β (1–42) levels were higher in MCI compared to AMC ( p < 0.0001) and more significant in AD than in MCI and AMC ( p < 0.0001) (Fig. 3 A). Similarly, in comparison to MCI and AMC, p-Tau levels were significantly higher in AD ( p < 0.0001) (Fig. 3 E). Similar levels of both markers were found in their Western blots (Fig. 2 ). We checked GFAP (astrocytic marker) and proinflammatory cytokines (TNF-α and IL-1β) to evaluate neuroinflammation. For proinflammatory markers, IL-1β and TNF-α levels showed a significant increase among the three groups ( p < 0.0001 for IL-1β and TNF-α) (Fig. 3 I, M). When comparing AD to MCI and AMC, the GFAP concentration in PsEVs was significantly higher ( p < 0.0001) (Fig. 3 Q). Similar trends were observed with Western blot analysis (Fig. 2 , Additional File 1 : Fig. S6, S9). Their elevated levels suggest prominent neuroinflammatory conditions contributing to potential neuronal damage. The elevated levels of these neuroinflammatory markers could be due to the activation of astrocytes and microglia and the subsequent increase in the secretion of PsEVs containing proinflammatory proteins, which suggests prominent neuroinflammatory conditions that may contribute to neuronal damage [ 57 ]. While synaptophysin concentration in PsEVs was downregulated in AD and MCI compared to AMC ( p < 0.0001) in ELISA (Fig. 3 U), it shows synaptic dysfunction. We also checked synaptophysin levels in PsEVs in Western blotting, finding it was downregulated in AD compared to MCI and AMC ( p = 0.0045, 0.0142), indicating synaptic degeneration in AD (Fig. 2 , Additional File 1 : Fig. S5). In MCI, synaptophysin levels did not significantly differ from AMC (Fig. 2 F). This aligns with synaptic loss in AD, reflected in lower neuropsychological test scores indicating more pronounced cognitive impairment compared to MCI and AMC.
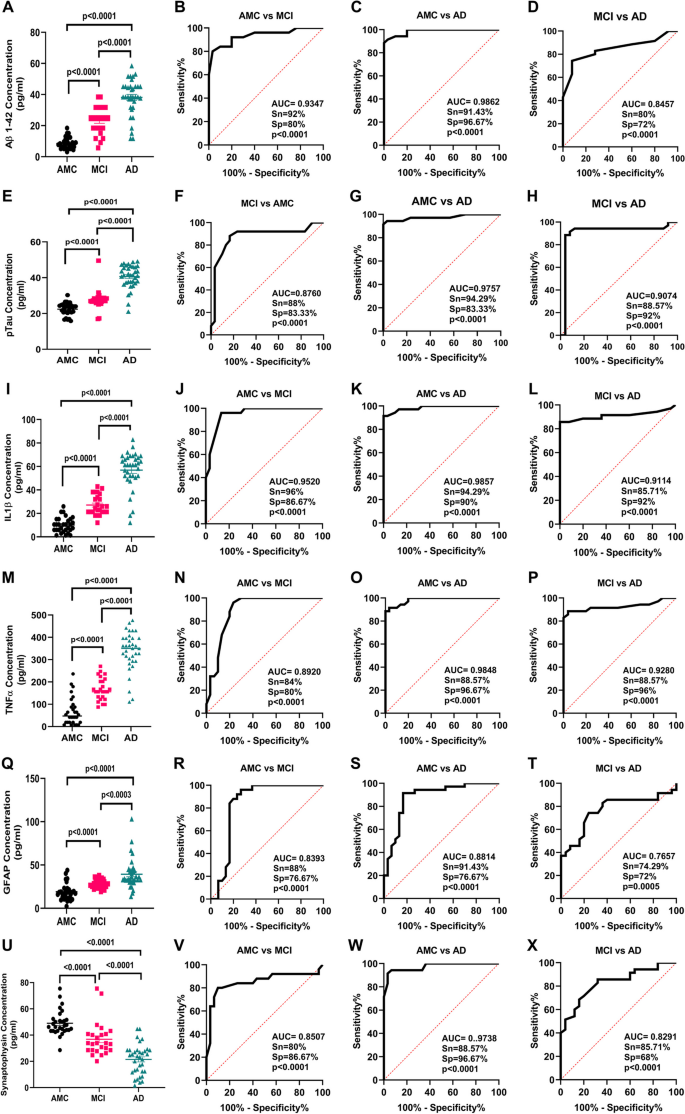
PsEVs derived amyloid-β (1–42), p-Tau, IL-1β, TNF-α, GFAP, and synaptophysin protein concentration was measured. ELISA results in A shows levels of PsEVs amyloid-β (1–42) in AMC, MCI, and AD and receiver operating characteristic (ROC) curve of PsEVs concentration in AMC v/s MCI ( B ), AMC v/s AD ( C ), and MCI v/s AD ( D ). Similarly, p-Tau concentration in AMC, MCI, and AD ( E ), ROC curve of PsEVs concentration in AMC v/s MCI ( F ), AMC v/s AD ( G ), and MCI v/s AD ( H ). PsEVs derived IL-1β concentration in AMC, MCI and AD ( I ), ROC curve of PsEVs concentration in AMC v/s MCI ( J ), AMC v/s AD ( K ), and MCI v/s AD ( L ). PsEVs derived TNF-α concentration in AMC, MCI and AD ( M ), ROC curve of PsEVs concentration in AMC v/s MCI ( N ), AMC v/s AD ( O ), and MCI v/s AD ( P ). Similarly, GFAP concentration in AMC, MCI, and AD ( Q ), ROC curve of PsEVs concentration in AMC v/s MCI ( R ), AMC v/s AD ( S ), and MCI v/s AD ( T ). For PsEVs-derived synaptophysin concentration in AMC, MCI, and AD ( U ), ROC curve of PsEVs concentration in AMC v/s MCI ( V ), AMC v/s AD ( W ), and MCI v/s AD ( X ). Abbreviations: AMC, age-matched control; MCI, mild-cognitive impairment patients; AD, Alzheimer’s disease patients; TNF-α, tumor necrosis factor-alpha; GFAP, glial fibrillary acidic protein
Determining the diagnostic potential of PsEVs-derived amyloid-β (1–42), p-Tau, IL-1β, TNF-α, GFAP and synaptophysin
We observed the levels of amyloid-β (1–42) and p-Tau in PsEVs, where the increase in amyloid-β (1–42) and p-Tau levels underscores their potential as biomarkers of MCI and AD. The diagnostic efficacy of amyloid-β (1–42) by ROC analysis was observed for AMC vs MCI [AUC = 0.9347, p < 0.0001, sensitivity (Sn) = 92%, specificity (Sp) = 80%] (Fig. 3 B), AMC vs AD (AUC = 0.9862, p < 0.0001, Sn = 91.43%, Sp = 96.67%) (Fig. 3 C), and MCI vs AD (AUC of 0.8457, p < 0.0001, Sn = 80%, and Sp = 72%) (Fig. 3 D). Similarly, diagnostic efficacy of p-Tau by ROC analysis was observed for AMC vs MCI (AUC = 0.8760, p < 0.0001, Sn = 88%, Sp = 83.33%) (Fig. 3 F), AMC vs AD (AUC = 0.9757, p < 0.0001, Sn = 94.29%, Sp = 83.33%) (Fig. 3 G), and MCI vs AD (AUC of 0.9074, p < 0.0001, Sn = 88.57%, and Sp = 92%) (Fig. 3 H). So, we observed that the pathological hallmarks of the disease, viz., amyloid-β (1–42) and p-Tau levels, are increased significantly in PsEVs cargo of AD and MCI groups.
Furthermore, we also checked GFAP, TNF-α, IL-1β, and synaptophysin in PsEVs from MCI and AD groups. The diagnostic efficacy of IL-1β by ROC analysis was observed for AMC vs MCI (AUC = 0.9520, p < 0.0001, Sn = 96%, Sp = 86.67%) (Fig. 3 J), AMC vs AD (AUC = 0.9857, p < 0.0001, Sn = 94.29%, Sp = 90%) compared to AMC (Fig. 3 K), MCI vs AD (AUC = 0.9114, p < 0.0001, Sn = 85.71%, Sp = 92%) (Fig. 3 L). Similarly, diagnostic efficacy of TNF-α by ROC analysis was observed for AMC vs MCI (AUC = 0.8920, p < 0.0001, Sn = 84%, Sp = 80%) (Fig. 3 N), AMC vs AD (AUC = 0.9848, p < 0.0001, Sn = 88.57%, Sp = 96.67%), and MCI vs AD (AUC = 0.9280, p < 0.0001, Sn = 88.57%, Sp = 96%) (Fig. 3 P). So, we observed an elevated expression of neuroinflammatory markers within the PsEVs isolated from the AD and MCI groups.
GFAP is an activation marker of astroglia, and in AD, this activation is associated with synaptic dysfunction [ 58 ]. In PsEVs, the diagnostic efficacy of GFAP by ROC analysis was observed as for AMC vs MCI (AUC = 0.8393, p < 0.0001, Sn = 88%, Sp = 76.67%) (Fig. 3 R), AMC vs. AD (AUC = 0.8814, p < 0.0001, Sn = 91.43%, Sp = 76.67%) compared to AMC (Fig. 3 S); MCI vs AD (AUC = 0.7657, p < 0.0001, Sn = 74.29%, Sp = 72%) (Fig. 3 T). In addition to this, we also checked the level of presynaptic protein, i.e., synaptophysin, within the PsEVs, as the level of synaptophysin correlates with cognitive decline in AD [ 59 ]. The diagnostic efficacy of synaptophysin by ROC analysis was observed as follows for AMC vs MCI (AUC = 0.8507, p < 0.0001, Sn = 80%, Sp = 86.67%) (Fig. 3 V), AMC vs AD (AUC = 0.9738, p < 0.0001, Sn = 88.57%, Sp = 96.67%) compared to AMC (Fig. 3 W); MCI vs AD (AUC = 0.8291, p < 0.0001, Sn = 85.71%, and Sp = 68%) (Fig. 3 X). Table 2 summarizes all the AUC, sensitivity, specificity, and p -value values for all the PsEVs-derived proteins.
Correlations of PsEVs concentration values with protein levels of amyloid-β (1–42), p-Tau, IL-1β, TNF-α, GFAP, and synaptophysin in PsEVs
As we found an elevated number of PsEVs in the diseased condition, we performed a correlation analysis between PsEVs concentration and the amyloid-β (1–42) level, p-Tau, IL-1β, and TNF-α within PsEV. We found that PsEV concentration was positively correlated with all the protein levels except synaptophysin, which showed a negative correlation (Fig. 4 ). In these correlations, amyloid-β (1–42) was positively correlated ( r = 0.7196, p < 0.0001) (Fig. 4 A); p-Tau positively correlates ( r = 0.7960, p < 0.0001) (Fig. 4 B); IL-1β also showed positive correlation ( r = 0.7220, p < 0.0001) (Fig. 4 C); and TNF-α also showed positive correlation ( r = 0.6473, p < 0.0001) (Fig. 4 D). GFAP showed a weak correlation with PsEVs concentration ( r = 0.5155, p < 0.0001) (Fig. 4 E), and synaptophysin showed a weak correlation ( r = 0.5752, p < 0.0001) (Fig. 4 F).
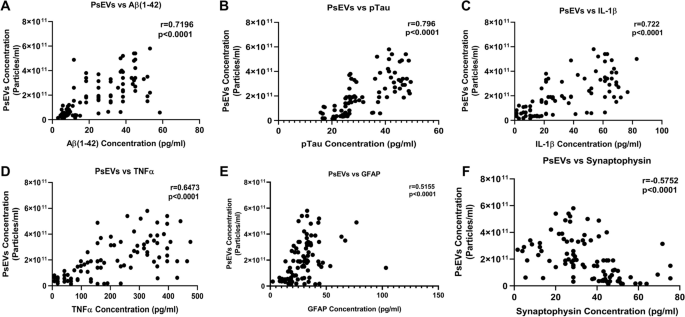
Correlation analysis between PsEVs concentration and PsEVs derived AD pathology markers. The correlation between PsEVs concentration with the amyloid-β (1–42) ( A ), p-Tau ( B ), IL-1β ( C ), TNF-α ( D ), GFAP ( E ), and synaptophysin ( F ). Abbreviations: p-Tau, Phospho-Tau, TNF-α, tumor necrosis factor-alpha; GFAP, glial fibrillary acidic protein. Spearman correlation was used for correlation analysis
Correlations of ACE-III and MMSE scores with protein levels of amyloid-β (1–42), p-Tau, IL-1β, and TNF-α in PsEVs
We performed a correlation analysis between ACE-III and MMSE values with the level of amyloid-β (1–42), p-Tau, IL-1β, TNF-α, GFAP, and synaptophysin (Fig. 5 ). We found that ACE-III and MMSE values were negatively correlated with all the protein levels except synaptophysin, which showed a positive value for the correlation coefficient. ACE-III values showed a negative correlation with amyloid-β (1–42) ( r = − 0.5107, p < 0.0001) (Fig. 5 A), p-Tau ( r = − 0.5055, p < 0.0001) (Fig. 5 B), IL-1β ( r = − 0.5684, p < 0.0001) (Fig. 5 C), and TNF-α ( r = − 0.6110, p < 0.0001) (Fig. 5 D). ACE-III values showed a negative correlation with GFAP ( r = − 0.5024, p < 0.0001) (Fig. 5 E), while synaptophysin showed a positive correlation ( r = 0.5036, p < 0.0001) (Fig. 5 F). In the case of MMSE, the values were as follows: for amyloid-β (1–42) ( r = − 0.5276, p < 0.0001) (Fig. 5 G), p-Tau ( r = − 0.6081, p < 0.0001) (Fig. 5 H), IL-1β ( r = − 0.5743, p < 0.0001) (Fig. 5 I), TNF-α ( r = − 0.5522, p < 0.0001) (Fig. 5 J), GFAP ( r = − 0.4596 p = 0.0002) (Fig. 5 K), and synaptophysin ( r = 0.5428, p < 0.0001) (Fig. 5 L). Table 3 summarizes all the values of Correlation coefficients for all the PsEVs-derived proteins.
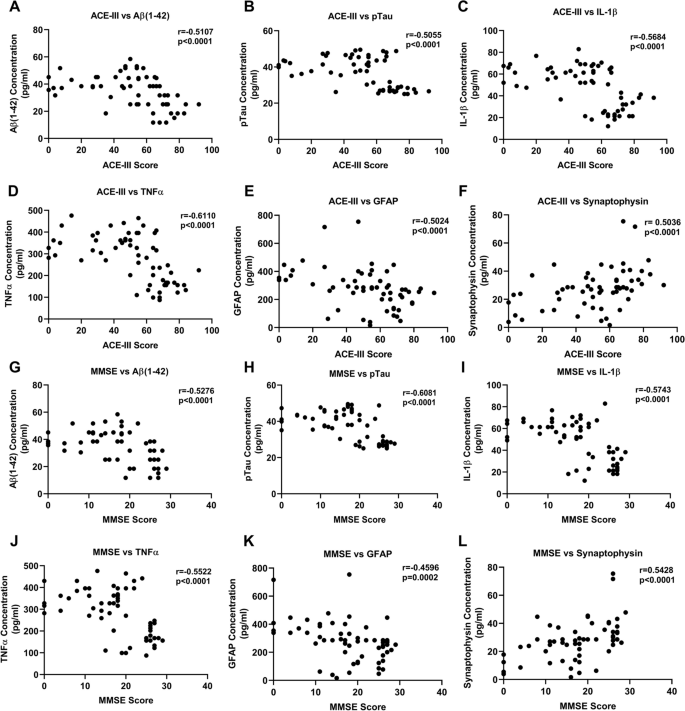
Correlation between neuropsychological test (ACE-III and MMSE) and PsEV-derived AD pathology markers. Amyloid-β (1–42) β, p-Tau, IL-1β, TNF-α, GFAP, and synaptophysin protein concentration. A – F Correlation between ACE-III scores and amyloid-β (1–42) ( A ), pTau ( B ), IL-1β ( C ), TNF-α ( D ), GFAP ( E ), and synaptophysin ( F ) protein concentration. G – L A correlation between MMSE Score and amyloid-β (1–42) ( G ), p-Tau (H), IL-1β ( I ), TNF-α ( J ), GFAP ( K ), and synaptophysin ( L ) protein concentration. Abbreviations: ACE-III, Addenbrooke Cognitive Examination; MMSE, Mini-Mental State Examination; p-Tau, Phospho-Tau; TNF-α, tumor necrosis factor-alpha; GFAP, glial fibrillary acidic protein. Spearman correlation was used for correlation analysis
In this study, we aimed to investigate the capacity of PsEVs to mirror pathological processes linked to AD and MCI. sEVs are extensively documented in the propagation of pathological processes associated with neurodegenerative and metabolic disorders [ 60 ]. The increased secretion of sEVs, coupled with the transmission of disease-related pathologies through sEVs-associated cargo, makes sEVs a viable candidate for understanding the physiological state of their originating cells, which is reflected in sEVs cargo [ 61 ]. To isolate the PsEVs, we employed a combined approach involving chemical precipitation followed by ultrafiltration, which effectively eliminates co-precipitants and minute protein contaminants such as albumin and LDL. We employed the neuronal protein L1CAM as a marker to ascertain the neuronal origin, although there is a debate surrounding its specificity for neuronal origin [ 62 ]. Nevertheless, in our study, the L1CAM marker is used to check for protein markers and not to confirm L1CAM affinity-based isolation. A two-step filtration procedure was used to accompany the sEV isolation method in our study to ensure high purity. Spherical lipid bi-layered vesicles within the typical size range of small extracellular vesicles (30–150 nm) were observed across AD, MCI, and AMC subjects (Fig. 1 A–C). NTA was employed to study the size distribution of sEVs in AD, MCI, and AMC. We observed that the isolated PsEVs come within the size range of < 200 nm, and there was a notable increase in the number of particles in diseased conditions compared to the control group. (Fig. 1 D–G).
Validation using sEVs-specific markers (CD63, CD81, and TSG101) demonstrated a noteworthy upregulation in MCI and AD, indicating PsEVs numbers are increased in disease conditions (Fig. 2 A–D). Levels of sEV-specific markers in AD and MCI are elevated because PsEV numbers are increased in the disease condition. As documented by various studies in MCI and AD, there is an increase in cross-talk between different pathophysiological processes, which leads to an increase in sEVs number and sEVs specific marker as a cellular response to heightened cellular stress aggravating neuronal damage and synaptic dysfunction [ 33 , 63 , 64 ]. Neuroinflammation, a characteristic feature of AD and MCI, may lead to the release of sEVs with inflammatory markers. Synaptic dysfunction, evidenced by synaptic degeneration, could contribute to the increased sEV-specific markers, reflecting vesicle release in response to altered synaptic activity [ 9 , 65 ]. Additionally, cells undergoing stress might activate compensatory mechanisms, and the elevated sEV-specific markers could signify communication for potential repair or damage mitigation. Therefore, the increase in sEV-specific markers may be linked to the progression of neurodegenerative processes, indicating ongoing pathological changes in the brain as the disease progresses. Additionally, the elevated expression of L1CAM, a neuron-associated marker, in MCI and AD further strengthens the association between PsEVs and neurodegenerative processes (Fig. 2 E). Furthermore, our observations extend beyond AD and MCI, showing increased concentrations of sEVs in other health conditions where higher levels of these vesicles correlate with elevated levels of disease markers [ 53 , 54 , 55 ]. The results of our research provide valuable insight into the characterization, validation, and functional implications of plasma-derived small extracellular vesicles (PsEVs) in the context of AD and MCI. Our comprehensive analysis encompassed morphological, biochemical, and functional aspects, shedding light on the potential role of PsEVs as biomarkers and contributors to neurodegenerative processes.
For this purpose, we performed the ELISA of amyloid-β (1–42) in PsEVs, where we observed higher protein concentrations of amyloid-β (1–42) in MCI. At the same time, in AD, the concentration also significantly increased (Fig. 3 A). In a similar study by A. Manolopoulos et al. [ 66 ], they studied levels of Aβ42, total Tau, and pro-brain-derived neurotrophic factor (BDNF) in both plasma neuron-derived extracellular vesicles (NDEVs) and plasma. The study reported a lack of correlation between the plasma and NDEVs, substantiating concerns about levels of the Aβ42 and total Tau measured in plasma originating from non-CNS sources. Multiple studies support the involvement of extracellular vesicles (EVs) in AD pathogenesis, where Aβ and Tau are released in association with EVs, influencing neuronal cell death and trans-synaptic spreading of the disease [ 7 , 15 , 54 , 67 ]. A progressive elevation in PsEV levels of p-Tau was observed in MCI, reaching a significantly higher AD concentration (Fig. 3 E). Previous research has revealed that p-tau alone effectively differentiates Frontotemporal Dementia (FTD) from AD with high specificity [ 68 , 69 ]. In our study, the alone analysis of p-Tau and amyloid-β (1–42) proved effective in distinguishing patients with MCI from AMC (Table 2 ). Consequently, studies have reported that the elevation of p-Tau suggests the future likelihood of AD development [ 70 ]. This dual elevation in amyloid-β (1–42) and p-Tau levels highlights their potential utility as concurrent biomarkers associated with MCI and AD diagnosis, as indicated by our ROC analysis. Therefore, the investigation into PsEV content revealed significant alterations in key markers associated with AD pathology, viz., amyloid-β (1–42) and p-Tau, which are a well-established marker of AD and exhibit an elevated level in PsEVs from AD and MCI patients compared to AMC in our study.
Synaptic dysfunction is considered a core feature of AD. It is suggested to precede other pathophysiological events of AD rather than neurodegeneration, which manifests during the later stages of the disease [ 71 ]. Synaptic dysfunction interacts with other core pathophysiology events of AD, such as the amyloid-β cascade, tau pathology, and neuroinflammation, eventually progressing to irreversible neurodegeneration and atrophy [ 72 , 73 ]. In this context, the synchronized exchange of proteins involved in these pathological processes between the CNS and neuronal-derived sEVs highlights the potential of sEVs as reliable carriers of pathophysiological cascade occurring at the pathological site [ 74 ]. In Fig. 3 U, we observed downregulated synaptophysin levels, a synaptic vesicle marker, in AD PsEVs compared to MCI and AMC. This suggests synaptic degeneration, which has also been discussed in several studies [ 59 , 63 , 64 ]. Synaptic damage induced by amyloid-β deposition triggers a response from the glia to eliminate impaired synapses. As amyloid-β accumulates, the severity of synaptic dysfunction intensifies, leading to tau hyperphosphorylation and the formation of tau tangles. Our study’s findings contradict J. Utz et al. (2021), which showed increased synaptophysin levels in microvesicles isolated from cerebrospinal fluid (CSF) in AD [ 28 ]. This discrepancy could be due to different biofluid sources, cellular origins, or clearance mechanisms for synaptophysin in these compartments. Our study also differs from Utz J et al. (2021) as we have studied PsEVs compared to microvesicles; both differ in biogenesis, structure, and functions. Moreover, our study aligns with existing studies that reported lower synaptophysin levels in plasma neuronal-derived EVs. Goetzl et al. [ 75 ] investigated the synaptic protein levels in neuronal-derived exosomes in plasma (NDEs) of patients with FTD and AD, where the authors found significantly lower levels of synaptopodin, neurogranin, synaptophysin, and synaptotagmin-2 in both conditions compared to controls. Furthermore, our results also align with the overall synaptic loss seen in AD patient’s brains, where lower levels of synaptophysin in the hippocampus have been reported to correlate with cognitive decline in AD [ 59 ]. Our study found that no significant difference in synaptophysin levels between MCI and AMC was observed, indicating that synapse dysfunction is more pronounced due to neuronal loss in the advanced disease stage, and its indication is reflected in PsEVs. Since the PsEVs pool also contains neuronal-derived EVs, we interpolate that the reduction in synaptic proteins in brain tissue is reflected in our results.
IL-1β, a potent immunomodulating cytokine, has previously been identified as a trigger for various inflammatory mediators in astrocytes and neurons [ 76 ]. Consistent evidence from post-mortem AD brain studies indicates the prevalent overexpression of IL-1β, with immunohistochemical analyses revealing its localization to microglia around plaques [ 77 ]. Moreover, pro-inflammatory markers (IL-1β and TNF-α) were significantly higher in PsEVs from AD and MCI subjects, as evidenced by ELISA and Western blot findings in our study (Fig. 3 I and M). Table 3 summarizes the correlation between PsEVs and neuroinflammatory markers. IL-1β plays a direct role in the pathophysiological changes associated with AD owing to its specific expression in the vicinity of plaques, and this localization suggests IL-1β as a mediator in the formation of plaques and tangles, thereby contributing to AD pathology [ 65 ]. TNF-α, another pro-inflammatory cytokine primarily secreted by activated macrophages and microglia, is recognized for its dual role in promoting cell survival and death in the central nervous system [ 78 , 79 ].
The cytoskeletal GFAP is found in astrocytic cells [ 80 ]. Increased plasma GFAP levels could result from “reactive astrogliosis,” another term for aberrant astrocytic function brought on by damage to neurons [ 81 ]. According to research on animal and cell models, reactive astrocytes encircle and penetrate amyloid-β plaques, contributing to the amyloid-β pathological process [ 82 , 83 ]. Research has demonstrated a correlation between amyloid-β burden, cognitive decline, and plasma GFAP [ 83 ]. PsEVs of GFAP were elevated in AD [ 27 ] and MCI (Fig. 3 Q). It is well known that sEVs play a pivotal role in the progression of disease pathologies in neurodegenerative and metabolic diseases [ 33 , 84 ]. The high levels of neuro-inflammatory markers (GFAP, TNF-α, and IL-1β) in PsEVs from MCI and AD subjects suggest a potential role of PsEVs in neuroinflammation. This activation of astrocytes and microglia precedes increased secretion of pro-inflammatory PsEVs and may contribute to neuronal damage and progressive cognitive impairment. Diseased conditions involve an increased secretion of sEVs and the cargo they carry, including pathological hallmark proteins or immunomodulatory cytokines [ 33 ].
Correlation analyses unveiled positive associations between PsEVs concentration and the protein levels of amyloid-β (1–42), p-Tau, IL-1β, TNF-α, GFAP, and synaptophysin (Fig. 4 ). Furthermore, our study also analyzed the correlation between cognitive examination scores (ACE-III and MMSE) and PsEV-associated protein levels (Fig. 5 ). The negative correlations observed imply that lower cognitive scores align with elevated levels of amyloid-β (1–42), p-Tau, IL-1β, and TNF-α in PsEVs Table 3 . This implies a strong connection between PsEV biomarkers and cognitive decline, reinforcing that PsEVs could serve as valuable diagnostic and prognostic tools. These findings underscore the potential of PsEVs as reliable disease progression and pathology indicators. The robust correlations further support the hypothesis that PsEVs may actively participate in disseminating neurodegenerative signals.
Our study extensively studied the multiple pathophysiological processes associated with AD by checking the protein levels involved in these processes within PsEVs, including amyloid-β (1–42), p-Tau, neuroinflammatory markers (IL-1β, TNF-α, GFAP), and synaptic protein levels. This comprehensive approach enhances diagnostic accuracy by considering the synergistic effects of these processes, providing valuable insights into disease progression from MCI to AD. We have also performed a systematic comparison with MCI, which was lacking in previous studies. We observed a significant correlation between these investigated protein levels within PsEVs and neuropsychological tests, thus filling a research gap addressing the clinical relevance of these dysregulated pathophysiological processes. The observed downregulated synaptophysin levels in AD PsEVs compared to MCI and control subjects shed light on the combined role of neuroinflammation and proteinopathy in the cognitive decline observed as the disease progresses. This finding suggests that PsEVs may reflect synaptic degeneration, opening avenues for further exploration into the role of PsEVs in synaptic damage and dysfunction in neurodegenerative diseases.
Our study provides a multifaceted examination of PsEVs, offering compelling evidence of their potential as biomarkers and functional contributors in AD. We have comprehensively discussed the synergistic role of synaptic dysfunction and neuroinflammation and their association with amyloid-β and tau pathologies within the PsEVs in AD progression. The pathophysiological conditions in the MCI and AD brain are reflected in PsEVs, as observed by the increased concentration of PsEVs containing disease-associated markers and markers for synaptic dysfunction and neuroinflammation. Therefore, the PsEVs can be exploited to understand the pathophysiological process involved in the progression and severity of MCI and AD.
Availability of data and materials
No datasets were generated or analysed during the current study.
Abbreviations
Addenbrooke Cognitive Examination
- Alzheimer’s disease
Age-matched controls
Glial fibrillary acidic protein
Interleukin-1β
- Mild cognitive impairment
Mini-Mental State Examination
Phospho-Tau
Tumor necrosis factor-alpha
Suescun J, Chandra S, Schiess MC. Chapter 13 - The role of neuroinflammation in neurodegenerative disorders. In: Actor JK, Smith KC, editors. Translational Inflammation. Academic Press; 2019. p. 241–67.
Chapter Google Scholar
DiSabato DJ, Quan N, Godbout JP. Neuroinflammation: the devil is in the details. J Neurochem. 2016;139(S2):136–53.
Article CAS PubMed PubMed Central Google Scholar
Lyman M, Lloyd DG, Ji X, Vizcaychipi MP, Ma D. Neuroinflammation: the role and consequences. Neurosci Res. 2014;79:1–12.
Article CAS PubMed Google Scholar
Mishra A, Kim HJ, Shin AH, Thayer SA. Synapse loss induced by interleukin-1β requires pre- and post-synaptic mechanisms. J Neuroimmune Pharmacol. 2012;7(3):571–8.
Article PubMed PubMed Central Google Scholar
Subramanian J, Savage JC, Tremblay MÈ. Synaptic loss in Alzheimer’s disease: mechanistic insights provided by two-photon in vivo imaging of transgenic mouse models. Front Cell Neurosci. 2020;14: 592607.
Chen GF, Xu TH, Yan Y, Zhou YR, Jiang Y, Melcher K, et al. Amyloid-β (1–42) beta: structure, biology and structure-based therapeutic development. Acta Pharmacol Sin. 2017;38(9):1205–35.
Sun X, Chen WD, Wang YD. β-Amyloid-β (1–42): the key peptide in the pathogenesis of Alzheimer’s DISEASE. Front Pharmacol. 2015;6:221.
Hampel H, Hardy J, Blennow K, Chen C, Perry G, Kim SH, et al. The amyloid-β (1–42)-β pathway in Alzheimer’s disease. Mol Psychiatry. 2021;26(10):5481–503.
Knopman DS, Amieva H, Petersen RC, Chételat G, Holtzman DM, Hyman BT, et al. Alzheimer disease. Nat Rev Dis Primers. 2021;7(1):1–21.
Article Google Scholar
Gu L, Guo Z. Alzheimer’s Aβ42 and Aβ40 peptides form interlaced Amyloid-β (1–42) fibrils. J Neurochem. 2013;126(3):305–11.
Bettens K, Sleegers K, Van Broeckhoven C. Current status on Alzheimer disease molecular genetics: from past, to present, to future. Hum Mol Genet. 2010;19(R1):R4–11.
George-Hyslop PHS. Molecular genetics of Alzheimer’s disease. Biol Psychiat. 2000;47(3):183–99.
Li NM, Liu KF, Qiu YJ, Zhang HH, Nakanishi H, Qing H. Mutations of beta-Amyloid-β (1–42) precursor protein alter the consequence of Alzheimer’s disease pathogenesis. Neural Regen Res. 2019;14(4):658–65.
Shen J, Kelleher RJ. The presenilin hypothesis of Alzheimer’s disease: evidence for a loss-of-function pathogenic mechanism. Proc Natl Acad Sci U S A. 2007;104(2):403–9.
Liu M, Sui D, Dexheimer T, Hovde S, Deng X, Wang KW, et al. Hyperphosphorylation renders Tau prone to aggregate and to cause cell death. Mol Neurobiol. 2020;57(11):4704–19.
Ferrer I, Andrés-Benito P, Ausín K, Pamplona R, del Rio JA, Fernández-Irigoyen J, et al. Dysregulated protein phosphorylation: a determining condition in the continuum of brain aging and Alzheimer’s disease. Brain Pathol. 2021;31(6): e12996.
Goel P, Chakrabarti S, Goel K, Bhutani K, Chopra T, Bali S. Neuronal cell death mechanisms in Alzheimer’s disease: an insight. Front Mol Neurosci. 2022;15: 937133.
Zhang W, Xiao D, Mao Q, Xia H. Role of neuroinflammation in neurodegeneration development. Sig Transduct Target Ther. 2023;8(1):1–32.
Choi SB, Kwon S, Kim JH, Ahn NH, Lee JH, Yang SH. The molecular mechanisms of neuroinflammation in Alzheimer’s disease, the consequence of neural cell death. Int J Mol Sci. 2023;24(14):11757.
Rajesh Y, Kanneganti TD. Innate immune cell death in neuroinflammation and Alzheimer’s disease. Cells. 2022;11(12):1885.
Skaper SD, Facci L, Zusso M, Giusti P. An inflammation-centric view of neurological disease: beyond the neuron. Front Cell Neurosci. 2018;10:00072.
Brucato DR, Benjamin DE. Synaptic pruning in Alzheimer’s disease: role of the complement system. Global J Med Res. 2020;20(F6):1–20.
Piccioni G, Mango D, Saidi A, Corbo M, Nisticò R. Targeting microglia-synapse interactions in Alzheimer’s disease. Int J Mol Sci. 2021;22(5):2342.
Mori Y, Tsuji M, Oguchi T, Kasuga K, Kimura A, Futamura A, et al. Serum BDNF as a potential biomarker of Alzheimer’s disease: verification through assessment of serum, cerebrospinal fluid, and medial temporal lobe atrophy. Front Neurol. 2021;12: 653267.
Giacomucci G, Mazzeo S, Bagnoli S, Ingannato A, Leccese D, Berti V, et al. Plasma neurofilament light chain as a biomarker of Alzheimer’s disease in subjective cognitive decline and mild cognitive impairment. J Neurol. 2022;269(8):4270–80.
Kim KY, Shin KY, Chang KA. GFAP as a potential biomarker for Alzheimer’s disease: a systematic review and meta-analysis. Cells. 2023;12(9):1309.
Ally M, Sugarman MA, Zetterberg H, Blennow K, Ashton NJ, Karikari TK, et al. Cross-sectional and longitudinal evaluation of plasma glial fibrillary acidic protein to detect and predict clinical syndromes of Alzheimer’s disease. Alzheimers Dement (Amst). 2023;15(4):e12492.
Article PubMed Google Scholar
Utz J, Berner J, Muñoz LE, Oberstein TJ, Kornhuber J, Herrmann M, et al. Cerebrospinal fluid of patients with Alzheimer’s disease contains increased percentages of synaptophysin-bearing microvesicles. Front Aging Neurosci. 2021;13: 682115.
Bruno D, Schurmann VS. Addenbrooke’s cognitive examination III in the diagnosis of dementia: a critical review. Neuropsychiatr Dis Treat. 2019;15:441–7.
Matías-Guiu JA, Valles-Salgado M, Rognoni T, Hamre-Gil F, Moreno-Ramos T, Matías-Guiu J. Comparative diagnostic accuracy of the ACE-III, MIS, MMSE, MoCA, and RUDAS for screening of Alzheimer disease. Dement Geriatr Cogn Disord. 2017;43(5–6):237–46.
Bajpai S, Upadhyay A, Sati H, Pandey RM, Chaterjee P, Dey AB. Hindi version of Addenbrook’s Cognitive Examination III: distinguishing cognitive impairment among older Indians at the lower cut-offs. Clin Interv Aging. 2020;15:329–39.
Howitt J, Hill AF. Exosomes in the pathology of neurodegenerative diseases. J Biol Chem. 2016;291(52):26589–97.
Rastogi S, Sharma V, Bharti PS, Rani K, Modi GP, Nikolajeff F, et al. The evolving landscape of exosomes in neurodegenerative diseases: exosomes characteristics and a promising role in early diagnosis. Int J Mol Sci. 2021;22(1):440.
Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977.
Gomes P, Tzouanou F, Skolariki K, Vamvaka-Iakovou A, Noguera-Ortiz C, Tsirtsaki K, et al. Extracellular vesicles and Alzheimer’s disease in the novel era of Precision Medicine: implications for disease progression, diagnosis and treatment. Exp Neurol. 2022;358: 114183.
Cocucci E, Meldolesi J. Ectosomes and exosomes: shedding the confusion between extracellular vesicles. Trends Cell Biol. 2015;25(6):364–72.
Pegtel DM, Gould SJ. Exosomes. Annu Rev Biochem. 2019Jun;20(88):487–514.
Gámez-Valero A, Campdelacreu J, Vilas D, Ispierto L, Reñé R, Álvarez R, et al. Exploratory study on microRNA profiles from plasma-derived extracellular vesicles in Alzheimer’s disease and dementia with Lewy bodies. Transl Neurodegener. 2019;8(1):31.
Lim WQ, Michelle Luk KH, Lee KY, Nurul N, Loh SJ, Yeow ZX, et al. Small extracellular vesicles’ miRNAs: biomarkers and therapeutics for neurodegenerative diseases. Pharmaceutics. 2023;15(4):1216.
Banks WA, Sharma P, Bullock KM, Hansen KM, Ludwig N, Whiteside TL. Transport of extracellular vesicles across the blood-brain barrier: brain pharmacokinetics and effects of inflammation. Int J Mol Sci. 2020;21(12):4407.
Zhou F, Ebea P, Mutai E, Wang H, Sukreet S, Navazesh S, et al. Small extracellular vesicles in milk cross the blood-brain barrier in murine cerebral cortex endothelial cells and promote dendritic complexity in the hippocampus and brain function in C57BL/6J mice. Front Nutr. 2022;10:9838543.
Google Scholar
Eren E, Leoutsakos JM, Troncoso J, Lyketsos CG, Oh ES, Kapogiannis D. Neuronal-derived EV biomarkers track cognitive decline in Alzheimer’s disease. Cells. 2022;11(3):436.
Asai H, Ikezu S, Tsunoda S, Medalla M, Luebke J, Haydar T, et al. Depletion of microglia and inhibition of exosome synthesis halt tau propagation. Nat Neurosci. 2015;18(11):1584–93.
Söllvander S, Nikitidou E, Brolin R, Söderberg L, Sehlin D, Lannfelt L, et al. Accumulation of amyloid-β (1–42)-β by astrocytes result in enlarged endosomes and microvesicle-induced apoptosis of neurons. Mol Neurodegener. 2016;11(1):38.
Sardar Sinha M, Ansell-Schultz A, Civitelli L, Hildesjö C, Larsson M, Lannfelt L, et al. Alzheimer’s disease pathology propagation by exosomes containing toxic amyloid-β (1–42)-beta oligomers. Acta Neuropathol. 2018;136(1):41–56.
Zyśk M, Beretta C, Naia L, Dakhel A, Påvénius L, Brismar H, et al. Amyloid-β (1–42)-β accumulation in human astrocytes induces mitochondrial disruption and changed energy metabolism. J Neuroinflammation. 2023;20(1):43.
Gabrielli M, Prada I, Joshi P, Falcicchia C, D’Arrigo G, Rutigliano G, et al. Microglial large extracellular vesicles propagate early synaptic dysfunction in Alzheimer’s disease. Brain. 2022;145(8):2849–68.
Beretta C, Nikitidou E, Streubel-Gallasch L, Ingelsson M, Sehlin D, Erlandsson A. Extracellular vesicles from amyloid-β (1–42)-β exposed cell cultures induce severe dysfunction in cortical neurons. Sci Rep. 2020;10(1):19656.
Chen Y, He Y, Han J, Wei W, Chen F. Blood-brain barrier dysfunction and Alzheimer’s disease: associations, pathogenic mechanisms, and therapeutic potential. Front Aging Neurosci. 2023;15:1258640.
Garcia-Contreras M, Thakor AS. Extracellular vesicles in Alzheimer’s disease: from pathology to therapeutic approaches. Neural Regen Res. 2022;18(1):18–22.
PubMed Central Google Scholar
Vella LJ, Hill AF, Cheng L. Focus on extracellular vesicles: exosomes and their role in protein trafficking and biomarker potential in Alzheimer’s and Parkinson’s disease. Int J Mol Sci. 2016;17(2):173.
Huo L, Du X, Li X, Liu S, Xu Y. The emerging role of neural cell-derived exosomes in intercellular communication in health and neurodegenerative diseases. Front Neurosci. 2021;15: 738442.
Rastogi S, Rani K, Rai S, Singh R, Bharti PS, Sharma V, et al. Fluorescence-tagged salivary small extracellular vesicles as a nanotool in early diagnosis of Parkinson’s disease. BMC Med. 2023;21(1):335.
Rani K, Rastogi S, Vishwakarma P, Bharti PS, Sharma V, Renu K, et al. A novel approach to correlate the salivary exosomes and their protein cargo in the progression of cognitive impairment into Alzheimer’s disease. J Neurosci Methods. 2021;347: 108980.
Rai S, Bharti PS, Singh R, Rastogi S, Rani K, Sharma V, et al. Circulating plasma miR-23b-3p as a biomarker target for idiopathic Parkinson’s disease: comparison with small extracellular vesicle miRNA. Front Neurosci. 2023;17:1174951.
Théry C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750.
Ng A, Tam WW, Zhang MW, Ho CS, Husain SF, McIntyre RS, et al. IL-1β, IL-6, TNF- α and CRP in elderly patients with depression or Alzheimer’s disease: systematic review and meta-analysis. Sci Rep. 2018;8(1):12050.
Hulshof LA, van Nuijs D, Hol EM, Middeldorp J. The role of astrocytes in synapse loss in Alzheimer’s disease: a systematic review. Front Cell Neurosci. 2022;16: 899251.
Sze CI, Troncoso JC, Kawas C, Mouton P, Price DL, Martin LJ. Loss of the presynaptic vesicle protein synaptophysin in hippocampus correlates with cognitive decline in Alzheimer disease. J Neuropathol Exp Neurol. 1997;56(8):933–44.
Yuyama K, Igarashi Y. Exosomes as carriers of Alzheimer’s amyloid-β (1–42)-ß. Front Neurosci. 2017;11:00229.
Watson LS, Hamlett ED, Stone TD, Sims-Robinson C. Neuronally derived extracellular vesicles: an emerging tool for understanding Alzheimer’s disease. Mol Neurodegener. 2019;14(1):22.
Gomes DE, Witwer KW. L1CAM-associated extracellular vesicles: a systematic review of nomenclature, sources, separation, and characterization. J Extracell Biol. 2022;1(3): e35.
Goetzl EJ, Kapogiannis D, Schwartz JB, Lobach IV, Goetzl L, Abner EL, et al. Decreased synaptic proteins in neuronal exosomes of frontotemporal dementia and Alzheimer’s disease. FASEB J. 2016;30(12):4141–8.
Goetzl EJ, Abner EL, Jicha GA, Kapogiannis D, Schwartz JB. Declining levels of functionally specialized synaptic proteins in plasma neuronal exosomes with progression of Alzheimer’s disease. FASEB J. 2018;32(2):888–93.
Jung YJ, Tweedie D, Scerba MT, Greig NH. Neuroinflammation as a factor of neurodegenerative disease: thalidomide analogs as treatments. Front Cell Develop Biol. 2019;7:00313.
Manolopoulos A, Delgado-Peraza F, Mustapic M, Pucha KA, Nogueras-Ortiz C, Daskalopoulos A, et al. Comparative assessment of Alzheimer’s disease-related biomarkers in plasma and neuron-derived extracellular vesicles: a nested case-control study. Front Mol Biosci. 2023;10:1254834.
He Z, Guo JL, McBride JD, Narasimhan S, Kim H, Changolkar L, et al. Amyloid-β (1–42)-β plaques enhance Alzheimer’s brain tau-seeded pathologies by facilitating neuritic plaque tau aggregation. Nat Med. 2018;24(1):29–38.
Pekeles H, Qureshi HY, Paudel HK, Schipper HM, Gornistky M, Chertkow H. Development and validation of a salivary tau biomarker in Alzheimer’s disease. Alzheimers Dement (Amst). 2018;11:53–60.
Silva MC, Ferguson FM, Cai Q, Donovan KA, Nandi G, Patnaik D, et al. Targeted degradation of aberrant tau in frontotemporal dementia patient-derived neuronal cell models. Elife. 2019;8:45457.
Article CAS Google Scholar
Holper S, Watson R, Yassi N. Tau as a biomarker of neurodegeneration. Int J Mol Sci. 2022;23(13):7307.
Lleó A, Núñez-Llaves R, Alcolea D, Chiva C, Balateu-Paños D, Colom-Cadena M, et al. Changes in synaptic proteins precede neurodegeneration markers in preclinical Alzheimer’s disease cerebrospinal fluid. Mol Cell Proteomics. 2019;18(3):546–60.
Guo T, Zhang D, Zeng Y, Huang TY, Xu H, Zhao Y. Molecular and cellular mechanisms underlying the pathogenesis of Alzheimer’s disease. Mol Neurodegener. 2020;15(1):40.
Breijyeh Z, Karaman R. Comprehensive review on Alzheimer’s disease: causes and treatment. Molecules. 2020;25(24):5789.
Snellman A, Ekblad LL, Koivumäki M, Lindgrén N, Tuisku J, Perälä M, et al. ASIC-E4: interplay of beta-amyloid-β (1–42), synaptic density and neuroinflammation in cognitively normal volunteers with three levels of genetic risk for late-onset Alzheimer’s disease – study protocol and baseline characteristics. Front Neurol. 2022;13: 826423.
Kinney JW, Bemiller SM, Murtishaw AS, Leisgang AM, Salazar AM, Lamb BT. Inflammation as a central mechanism in Alzheimer’s disease. Alzheimers Dement (N Y). 2018;4:575–90.
Mrak RE, Griffin WST. Interleukin-1 and the immunogenetics of Alzheimer disease. J Neuropathol Exp Neurol. 2000;59(6):471–6.
Frankola KA, Greig NH, Luo W, Tweedie D. Targeting TNF-alpha to elucidate and ameliorate neuroinflammation in neurodegenerative diseases. CNS Neurol Disord Drug Targets. 2011;10(3):391–403.
Jayaraman A, Htike TT, James R, Picon C, Reynolds R. TNF-mediated neuroinflammation is linked to neuronal necroptosis in Alzheimer’s disease hippocampus. Acta Neuropathol Commun. 2021;9(1):159.
Jain P, Wadhwa PK, Jadhav HR. Reactive astrogliosis: role in Alzheimer’s disease. CNS Neurol Disord Drug Targets. 2015;14(7):872–9.
Kamphuis W, Mamber C, Moeton M, Kooijman L, Sluijs JA, Jansen AHP, et al. GFAP isoforms in adult mouse brain with a focus on neurogenic astrocytes and reactive astrogliosis in mouse models of Alzheimer disease. PLoS ONE. 2012;7(8): e42823.
Chatterjee P, Pedrini S, Stoops E, Goozee K, Villemagne VL, Asih PR, et al. Plasma glial fibrillary acidic protein is elevated in cognitively normal older adults at risk of Alzheimer’s disease. Transl Psychiatry. 2021;11(1):1–10.
Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):6977.
Download references
Acknowledgements
We express our gratitude to the Electron Microscopy Facility, Sophisticated Analytical Instrumentation Facility (SAIF) at AIIMS, New Delhi.
Authors’ X handles
X (Formally Twitter) handle: Saroj Kumar (corresponding author)—@skumarlabaiims.
Open access funding provided by Lulea University of Technology. The Indian Council of Medical Research (ICMR, funding number 2020–1194), Council of Scientific and Industrial Research (CSIR, funding number 09/006(0533)/2021-EMR-I), and Department of Health Research (DHR, funding numbers GIA/2020/000595, YSS/2020/000158) provided funding for this research manuscript.

Author information
Authors and affiliations.
Department of Biophysics, All India Institute of Medical Sciences, New Delhi, 110029, India
Rishabh Singh, Sanskriti Rai, Prahalad Singh Bharti, Sadaqa Zehra, Krishna Kishore Inampudi & Saroj Kumar
Department of Anatomy, All India Institute of Medical Sciences, New Delhi, India
Priya Kumari Gorai & Neerja Rani
Department of Pharmaceutical Engineering and Technology, Indian Institute of Technology BHU, Varanasi, India
Gyan Prakash Modi
Department of Biotechnology, Jamia Millia Islamia, New Delhi, India
Department of Neurology, All India Institute of Medical Sciences, New Delhi, India
Vishnu V. Y.
Department of Geriatric Medicine, All India Institute of Medical Sciences, New Delhi, India
Prasun Chatterjee
Department of Health, Education, and Technology, Lulea University of Technology, Lulea, 97187, Sweden
Fredrik Nikolajeff & Saroj Kumar
You can also search for this author in PubMed Google Scholar
Contributions
S.K. conceptualized and designed the study. R.S., S.R., P.S.B., and S.Z. performed the acquisition and analysis of data. R.S., S.R., P.S.B., S.Z., and P.K.G. performed the drafting the text or preparing the figures. R.S., S.R., P.S.B., N.R., K.D., K.K.I., P.C., V.V.Y, G.P.M., F.N., and S.K. performed the initial revision and proofreading of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Saroj Kumar .
Ethics declarations
Ethics approval and consent to participate.
The ethical clearance for this study was obtained from the institutional ethics committee of All Institute of Medical Sciences, New Delhi, India. The ethical clearance number is IECPG-670/25.08.2022. All subjects were recruited for the study after obtainment of the written informed consent form. A detailed written participant information sheet and participant informed consent form were provided to the subjects to take part in this study and their signatures were obtained.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
12916_2024_3475_moesm1_esm.docx.
Additional file 1: Fig S1. [CD63 expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis]. Fig S2. [CD81 expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis]. Fig S3. [TSG101 expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis]. Fig S4. [L1CAM expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis]. Fig S5. [Synaptophysin (SYP) expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis]. Fig S6. [Glial Fibrillary Acidic Protein (GFAP) expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis]. Fig S7. [β-Actin expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis]. Fig S8. [Amyloidβ-42 Oligomer expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis]. Fig S9. [IL1β (A) and TNFα (B) expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis]. Fig S10. [p-Tau expression in age-matched controls (AMC), mild cognitive impairment (MCI), and Alzheimer’s disease (AD) and their densitometric analysis].
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Singh, R., Rai, S., Bharti, P.S. et al. Circulating small extracellular vesicles in Alzheimer’s disease: a case–control study of neuro-inflammation and synaptic dysfunction. BMC Med 22 , 254 (2024). https://doi.org/10.1186/s12916-024-03475-z
Download citation
Received : 27 February 2024
Accepted : 11 June 2024
Published : 20 June 2024
DOI : https://doi.org/10.1186/s12916-024-03475-z
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Small extracellular vesicles
- Synaptic dysfunction
- Neuroinflammation
BMC Medicine
ISSN: 1741-7015
- Submission enquiries: [email protected]
- General enquiries: [email protected]

- Our Latest News
Retrospective study based on electronic health records finds popular diabetes and weight-loss drugs associated with reduction in incidence and recurrence of alcohol-use disorder by at least half

A new study by researchers at the Case Western Reserve University School of Medicine reveals that the popular diabetes and weight-loss drugs Wegovy and Ozempic are linked to reduced incidence and recurrence of alcohol abuse or dependence.
The team’s findings, recently published in the journal Nature Communications, may suggest a possible new treatment for excessive alcohol use—including alcohol-use disorder (AUD), a health condition that causes about 178,000 deaths in the United States each year, according to the Centers for Disease Control.
To date, the U.S. Food and Drug Administration (FDA) has approved only three medications to treat AUD.
The active ingredient in Wegovy and Ozempic is semaglutide, which belongs to a class of medications known as glucagon-like peptide-1 receptor agonists (GLP-1). GLP-1 helps regulate blood sugar in type 2 diabetes and reduces appetite.
The researchers examined electronic health records of nearly 84,000 patients with obesity. They found those treated with semaglutide, compared to those treated with other anti-obesity medications, showed a 50% to 56% decrease for both the initiation and re-occurrence of alcohol-use disorder in the year following.

“This is very promising news in that we may have a new therapeutic method to treat AUD,” said Rong Xu, a professor of biomedical informatics at the School of Medicine and the study’s lead researcher.
Xu, also director of the medical school’s Center for AI in Drug Discovery , was joined by medical school co-authors Nathan Berger , the Hanna-Payne Professor of Experimental Medicine, and Pamela Davis , the Arline H. and Curtis F. Garvin Research Professor. Nora D. Volkow , director of the National Institute on Drug Abuse , also co-authored the study.
“We collected real-world evidence in a manner similar to our previous two studies reported earlier this year,” Berger said. “In January we showed that semaglutide is associated with a decrease in suicidal thoughts, and in March , we demonstrated that semaglutide is also associated with a reduction in both new diagnoses and recurrence of cannabis-use disorder.”
Similar findings were replicated when the team examined electronic health records for about 600,000 patients with type 2 diabetes. Again, they found consistent reductions in alcohol-use disorder diagnoses among those treated with semaglutide.
“While the findings are promising and provide preliminary evidence of the potential benefit of semaglutide in AUD in real-world populations,” Davis said, “further randomized clinical trials are needed to support its use clinically for AUD.”
Research reported in this press release was supported by the National Institute on Alcohol Abuse and Alcoholism, National Institute on Aging, and National Cancer Institute, all of the National Institutes of Health, under award numbers AA029831, AG057557, AG061388, AG062272, AG07664, CA221718, CA043703, and CA2332216. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
For more information please contact Patty Zamora at [email protected] .
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- BMC Res Notes

The clinical case report: a review of its merits and limitations
Trygve nissen.
1 Department of Clinical Medicine, University of Tromsø, N-9038 Tromsø, Norway
2 Division of General Psychiatry, University Hospital of North Norway, N-9291 Tromsø, Norway
3 Division of Addictions and Specialized Psychiatry, University Hospital of North Norway, N-9291 Tromsø, Norway
The clinical case report has a long-standing tradition in the medical literature. While its scientific significance has become smaller as more advanced research methods have gained ground, case reports are still presented in many medical journals. Some scholars point to its limited value for medical progress, while others assert that the genre is undervalued. We aimed to present the various points of view regarding the merits and limitations of the case report genre. We searched Google Scholar, PubMed and select textbooks on epidemiology and medical research for articles and book-chapters discussing the merits and limitations of clinical case reports and case series.
The major merits of case reporting were these: Detecting novelties, generating hypotheses, pharmacovigilance, high applicability when other research designs are not possible to carry out, allowing emphasis on the narrative aspect (in-depth understanding), and educational value. The major limitations were: Lack of ability to generalize, no possibility to establish cause-effect relationship, danger of over-interpretation, publication bias, retrospective design, and distraction of reader when focusing on the unusual.
Conclusions
Despite having lost its central role in medical literature in the 20th century, the genre still appears popular. It is a valuable part of the various research methods, especially since it complements other approaches. Furthermore, it also contributes in areas of medicine that are not specifically research-related, e.g. as an educational tool. Revision of the case report genre has been attempted in order to integrate the biomedical model with the narrative approach, but without significant success. The future prospects of the case report could possibly be in new applications of the genre, i.e. exclusive case report databases available online, and open access for clinicians and researchers.
Throughout history the clinical case report and case report series have been integral components of medical literature [ 1 ]. The case report genre held a strong position until it was sidelined in the second half of the 20 th century [ 2 , 3 ]. New methodologies for research articles paved the way for evidence-based medicine. Editors had to make space for these research articles and at the same time signaled less enthusiasm for publishing case reports [ 4 ]. This spurred some heated debates in medical journals as readers were worried that the traditional case report was in jeopardy [ 5 , 6 ]. Those who welcomed the new trend with fewer case reports being published pointed mainly to their low quality and inclination to emphasize mere curiosa [ 7 - 9 ]. Some of the proponents of the genre claimed that the case report had been and still was indispensible for furthering medical knowledge and that it was unique in taking care of the detailed study of the individual patient as opposed to the new research methods with their “…nomothetic approach [taking] precedence…” [ 5 ]. Still, the case report got a low ranking on the evidence hierarchy. After a decline in popularity a new interest for the case report emerged, probably beginning in the late 1990s [ 2 ]. A peer-reviewed ‘Case reports’ section was introduced in the Lancet in 1995 [ 10 ]. In 2007, the first international, Pubmed-listed medical journal publishing only case reports was established [ 11 , 12 ]. In the following years, several similar journals, for the most part online and open-access, have been launched.
The present debate is not so much focused on whether case reporting is obsolete or not. Some of the discussions after the turn of the century have been about adapting the case report genre to new challenges. One example is the suggestion of incorporating the narrative, i.e. “… stressing the patient’s story”, in the case report [ 13 ]. The authors termed their initiative “The storied case report”. Their endeavor was not met with success. In analyzing the causes for this, they wondered if “… junior trainees find it too hard to determine what is relevant and senior trainees find it too hard to change their habits” [ 13 ]. A similar attempt was done when the editors of the Journal of Medical Case Reports in 2012 encouraged authors to include the patients’ perspectives by letting patients describe their own experiences [ 14 ].
Notwithstanding, we feel there is much to be gained from having an ongoing discussion highlighting the indications and contraindications for producing case reports. This can to some degree be facilitated by getting an understanding of the merits and limitations of the genre. The objective of this article is to present the merits and limitations of case reports and case series reports.
We adopted Taber’s Cyclopedic Medical Dictionary’s definition of the case report : “A formal summary of a unique patient and his or her illness, including the presenting signs and symptoms, diagnostic studies, treatment course and outcome” [ 15 ]. A case report consists of one or two cases, most often only one. The case series or case series report usually consists of three to ten cases [ 16 ]. (In the following we use the term case report to denote both case reports and case series report). Case reports are most often naturalistic and descriptive. Sometimes, however, they can be prospective and experimental.
As literature specifically dealing with the case report genre seemed harder to elicit from the databases than the vast amount of particular case reports, we performed iterative searches. We searched Google Scholar and PubMed using the search terms ‘case report(s)’, ‘case series’, ‘case series report(s)’, ‘case reporting’ in various combinations with ‘clinical’, ‘medical’, ‘anecdotal’, ‘methodology’, ‘review’, ‘overview’, ‘strengths’, ‘weaknesses’, ‘merits’, and ‘limitations’. Further references were identified by examining the literature found in the electronic searches. We also consulted major textbooks on epidemiology [ 17 , 18 ], some scholars of medical genres [ 19 , 20 ] and a monograph on case reporting by the epidemiologist M. Jenicek [ 16 ]. We delimited our review to the retrospective, naturalistic, and descriptive case report, also labeled the “traditional” or “classic” case report, and case series including such reports. Thus we excluded other types, such as the planned, qualitative case study approach [ 21 ] and simulated cases [ 22 - 24 ]. Finally, we extracted the relevant data and grouped the merits and limitations items in rank order with the items we judged to be the most important first.
New observations
The major advantage of case reporting is probably its ability to detect novelties [ 16 ]. It is the only way to present unusual, uncontrolled observations regarding symptoms, clinical findings, course of illness, complications of interventions, associations of diseases, side effects of drugs, etc. In short, anything that is rare or has never been observed previously might be important for the medical community and ought to be published. A case report might sensitize readers and thus facilitate detection of similar or identical cases.
Generating hypotheses
From a single, or preferably several single case reports or a case series, new hypotheses could be formulated. These could then be tested with formal research methods that are designed to refute or confirm the hypotheses, i.e. comparative (observational and experimental) studies.
There are numerous examples of new discoveries or major advancements in medicine that started with a case report or, in some cases, as humbly as a letter to the editor. The first concern from the medical community about the devastating side effect of thalidomide, i.e. the congenital abnormalities, appeared as a letter to the editor in the Lancet in 1961 [ 25 ]. Soon thereafter, several case reports and case series reports were published in various journals. Case reporting is thus indispensable in drug safety surveillance (pharmacovigilance) [ 26 ].
Sometimes significant advancements in knowledge have come not from what researchers were pursuing, but from “accidental discoveries”, i.e. by serendipity. The story of Alexander Fleming’s discovery of penicillin in 1928 is well known in the medical field [ 27 ]. Psychiatry has profited to a large degree from this mode of advancing medical science as many of the drugs used for mental disorders have been discovered serendipitously [ 27 ]. One notable example is the discovery of the effect of lithium on manic episodes in patients with manic-depressive disorder [ 28 ]. A more recent discovery is the successful treatment of infantile hemangiomas with systemic propranolol. This discovery was published, as a case series report, in the correspondence section in New England Journal of Medicine [ 29 ]. However, the evidence for the effect of this treatment is still preliminary, and several randomized trials are under way [ 30 , 31 ].
Clear and operational entities are prerequisites for doing medical research. Descriptions must come before understanding. Clinical observations that lead to new disorders being described are well suited for case reporting. The medical literature is replete with case-based articles describing new diseases and syndromes. One notable example is the first description of neurasthenia by G. Beard in Boston Medical and Surgical Journal in 1869 [ 32 ].
Researching rare disorders
For rare disorders randomized controlled trials (RCTs) can be impossible to run due to lack of patients to be enrolled. Research on drug treatment and other kinds of interventions must therefore be based on less rigorous methodologies, among them case series and case reports. This would be in accordance with the European Commission’s recommendation to its members to improve health care for those with rare disorders [ 33 ].
Solving ethical constraints
Case reporting can be valuable when ethical constraints prohibit experimental research. Take as an example the challenge of how to manage the side effects of accidental extravasation of cytotoxic drugs. As RCTs on humans seem unethical in this clinical situation the current guidelines rest on small observational studies, case reports and animal studies [ 34 ]. Or another example: Physical restraint is sometimes associated with sudden, unexpected death. The cause or causes for this are to some degree enigmatic, and it is hard to conceive of a controlled study that could be ethical [ 35 , 36 ]. Case reports and case series being “natural experiments” might be the only evidence available for guiding clinical practice.
In-depth narrative case studies
Case reporting can be a way of presenting research with an idiographic emphasis. As contrasted to nomothetic research, an idiographic approach aims at in-depth understanding of human phenomena, especially in the field of psychology and psychiatry. The objective is not generalizable knowledge, but an understanding of meaning and intentionality for an individual or individuals. Sigmund Freud’s case studies are relevant examples. This usage of case reports borders on qualitative research. Qualitative studies, although developed in the social sciences, have become a welcome contribution within health sciences in the last two decades.
Educational value
Clinical medical learning is to a large degree case-based. Typical case histories and vignettes are often presented in textbooks, in lectures, etc. Unusual observations presented as published case reports are important as part of doctors’ continuing medical education, especially as they demonstrate the diversity of manifestations both within and between medical diseases and syndromes [ 37 , 38 ]. Among the various medical texts, the case report is the only one that presents day-to-day clinical practice, clinicians’ diagnostic reasoning, disease management, and follow-up. We believe that some case reports that are written with the aim of contributing to medical knowledge turn out to be of most value educationally because the phenomena have already been described elsewhere. Other case reports are clearly primarily written for educational value [ 37 ]. Some journals have regular sections dedicated to educational case reports, e.g. The Case Records of the Massachusetts General Hospital in the New England Journal of Medicine and the Clinical Case Conference found in the American Journal of Psychiatry.
The cost of doing a case report is low compared to planned, formal studies. Most often the necessary work is probably done in the clinical setting without specific funding. Larger studies, for instance RCTs, will usually need an academic setting.
Fast publication
The time span from observation to publication can be much shorter than for other kinds of studies. This is obviously a great advantage as a case report can be an important alert to the medical community about a serious event. The unexpected side effects of the sedative-antinauseant thalidomide on newborn babies is a telling story. The drug had been prescribed during pregnancy to the babies’ mothers. After the first published observation of severe abnormalities in babies appeared as a letter to the editor of the Lancet in December 16 th , 1961 [ 25 ], several case reports and series followed [ 39 , 40 ]. It should be mentioned though that the drug company had announced on December 2 nd , 1961, i.e. two weeks before the letter from McBride [ 25 ], that it would withdraw the drug form the market immediately [ 41 ].
Flexible structure
Riaz Agha, editor of the International Journal of Surgery Case Reports suggests that the case report, with its less rigid structure is useful as it “… allows the surgeon(s) to discuss their diagnostic approach, the context, background, decision-making, reasoning and outcomes” [ 42 ]. Although the editor is commenting on the surgical case report, the argument can be applied for the whole field of clinical medicine. It should be mentioned though, that other commentators have argued for a more standardized, in effect more rigid, structure [ 43 ].
Clinical practice can be changed
Case reporting can lead to or contribute to a change in clinical practice. A drug might be withdrawn from the market. Or a relabeling might change the attitude to and treatment of a condition. During Word War I the shell shock syndrome was labeled and described thoroughly in several articles in the Lancet , the first of them appearing in February 1915 [ 44 ]. The author was the British captain and military doctor Charles S. Myers. Before his efforts to bring good care and treatment to afflicted soldiers there had been a common misconception that many of these dysfunctional soldiers were malingerers or cowards.
Exercise for novice researchers
The case report format is well suited for young doctors not yet trained as researchers. It can be an opportunity for a first exercise in authoring an article and a preparation for a scientific career [ 37 , 45 , 46 ].
Communication between the clinical and academic fields
Articles authored by clinicians can promote communication between practicing clinicians and academic researchers. Observations published can generate ideas and be a trigger for further studies. For instance, a case series consisting of several similar cases in a short period can make up the case-group for a case–control study [ 47 ]. Clinicians could do the observation and publish the case series while the case–control study could be left to the academics.
Entertainment
Some commentators find reading case reports fun. Although a rather weak argument in favor of case reporting, the value of being entertained should not be dismissed altogether. It might inspire physicians to spend more time browsing and reading scientific literature [ 48 ].
Studying the history of medicine
Finally, we present a note on a different and unintended aspect of the genre. The accumulated case reports from past eras are a rich resource for researching and understanding medical history [ 49 , 50 ]. A close study of old case reports can provide valuable information about how medicine has been practiced through the centuries [ 50 , 51 ].
Limitations
No epidemiological quantities.
As case reports are not chosen from representative population samples they cannot generate information on rates, ratios, incidences or prevalences. The case or cases being the numerator in the equation, has no denominator. However, if a case series report consists of a cluster of cases, it can signal an important and possibly causal association, e.g. an epidemic or a side effect of a newly marketed drug.
Causal inference not possible
Causality cannot be inferred from an uncontrolled observation. An association does not imply a cause-effect relationship. The observation or event in question could be a mere coincidence. This is a limitation shared by all the descriptive studies [ 47 ]. Take the thalidomide tragedy already mentioned as an example; Unusual events such as congenital malformations in some of the children born to mothers having taken a specific drug during pregnancy does not prove that the drug is the culprit. It is a mere hypothesis until further studies have either rejected or confirmed it. Cause-effect relationships require planned studies including control groups that to the extent possible control for chance, bias and confounders [ 52 ].
Generalization not possible
From the argument above, it follows that findings from case reports cannot be generalized. In order to generalize we need both a cause-effect relationship and a representative population for which the findings are valid. A single case report has neither. A case series, on the other hand, e.g. many “thalidomide babies” in a short time period, could strengthen the suspicion of a causal relationship, demanding further surveillance and research.
Publication bias could be a limiting factor. Journals in general favor positive-outcome findings [ 53 ]. One group of investigators studying case reports published in the Lancet found that only 5% of case reports and 10% of case series reported treatment failures [ 54 ]. A study of 435 case reports from the field of dentistry found that in 99.1%, the reports “…clearly [had] a positive outcome and the intervention was considered and described as successful by the authors” [ 55 ].
Overinterpretation
Overinterpretation or misinterpretation is the tendency or temptation to generalize when there is no justification for it. It has also been labeled “the anecdotal fallacy” [ 56 ]. This is not a shortcoming intrinsic to the method itself. Overinterpretation may be due to the phenomenon of case reports often having an emotional appeal on readers. The story implicitly makes a claim to truth. The reader might conclude prematurely that there is a causal connection. The phenomenon might be more clearly illustrated by the impact of the clinician’s load of personal cases on his or her practice. Here exemplified by a young doctor’s confession: “I often tell residents and medical students, ‘The only thing that actually changes practice is adverse anecdote.’” [ 57 ].
Emphasis on the rare
As case reporting often deals with the rare and atypical, it might divert the readers’ attention from common diseases and problems [ 58 ].
Confidentiality
Journals today require written informed consent from patients before publishing case reports. Both authors and publishers are responsible for securing confidentiality. A guarantee for full confidentiality is not always possible. Despite all possible measures taken to preserve confidentiality, sometimes the patient will be recognized by someone. This information should be given to the patient. An adequately informed patient might not consent to publication. In 1995 in an Editorial in the British Journal of Psychiatry one commentator, Isaac Marks, feared that written consent would discourage case reports being written [ 59 ]. Fortunately, judged form the large number of reports being published today, it seems unlikely that the demand for consent has impeded their publication.
Other methodological limitations
Case reports and series are written after the relevant event, i.e. the observation. Thus, the reports are produced retrospectively. The medical record might not contain all relevant data. Recall bias might prevent us from getting the necessary information from the patient or other informants such as family members and health professionals.
It has also been held against case reporting that it is subjective. The observer’s subjectivity might bias the quality and interpretation of the observation (i.e. information bias).
Finally, the falsification criterion within science, which is tested by repeating an experiment, cannot be applied for case reports. We cannot design another identical and uncontrolled observation. However, unplanned similar “experiments” of nature can be repeated. Several such observations can constitute a case series that represents stronger indicative evidence than the single case report.
The major advantages of case reporting are the ability to make new observations, generate hypotheses, accumulate scientific data about rare disorders, do in-depth narrative studies, and serve as a major educational tool. The method is deficient mainly in being unable to deliver quantitative data. Nor can it prove cause-effect relationship or allow generalizations. Furthermore, there is a risk of overinterpretation and publication bias.
The traditional case report does not fit easily into the qualitative-quantitative dichotomy of research methods. It certainly shares some characteristics with qualitative research [ 16 ], especially with regard to the idiographic, narrative perspective – the patient’s “interior world” [ 60 ] – that sometimes is attended to. Apart from “The storied case report” mentioned in the Background-section, other innovative modifications of the traditional case report have been tried: the “evidence-based case report” [ 61 ], the “interactive case report” [ 62 ] and the “integrated narrative and evidence based case report” [ 63 ]. These modifications of the format have not made a lasting impact on the way case reports in general are written today.
The method of case reporting is briefly dealt with in some textbooks on epidemiology [ 17 , 18 ]. Journals that welcome case reports often put more emphasis on style and design than on content in their ‘instruction to authors’ section [ 64 ]. As a consequence, Sorinola and coworkers argue for more consensus and more consistent guidance on writing case reports [ 64 ]. We feel that a satisfactory amount of guidance concerning both style and content now exists [ 12 , 16 , 65 , 66 ]. The latest contribution, “The CARE guidelines”, is an ambitious endeavor to improve completeness and transparency of reports [ 66 ]. These guidelines have included the “Patient perspective” as an item, apparently a bit half-heartedly as this item is placed after the Discussion section, thus not allowing this perspective to influence the Discussion and/or Conclusion section. We assume this is symptomatic of medicine’s problem with integrating the biomedical model with “narrative-based medicine”.
In recent years the medical community has taken an increased interest in case reports [ 2 ], especially after the surge of online, exclusive case report journals started in 2007 with the Journal of Medical Case Reports (which was the first international, Pubmed-listed medical journal publishing only case reports) as the first of this new brand. The climate of skepticism has been replaced by enthusiasm and demand for more case reports. A registry for case reports, Cases Database, was founded in 2012 [ 67 ]. On the condition that it succeeds in becoming a large, international database it could serve as a register being useful for clinicians at work as well as for medical research on various clinical issues. Assuming Pamela P. Powell’s assertion that “[a]lmost all practicing physicians eventually will encounter a case worthy of being reported” [ 60 ] is valid, there should be no shortage of potential cases waiting to be reported and filed in various databases, preferably online and open access.
Limitations of this review
There are several limitations to this study. It is a weakness that we have not been able to review all the relevant literature. The number of publications in some way related to case reports and case report series is enormous, and although we have attempted to identify those publications relevant for our purpose (i.e. those that describe the merits and limitations of the case report genre), we might have missed some. It was difficult to find good search terms for our objective. Still, after repeated electronic searches supplemented with manual searches in reference lists, we had a corpus of literature where essentially no new merits or limitations emerged.
As we point out above, the ranking of merits and limitations represents our subjective opinion and we acknowledge that others might rank the importance of the items differently.
The perspective on merits and limitations of case reporting has been strictly medical. As a consequence we have not analyzed or discussed the various non-medical factors affecting the publication of case reports in different medical journals [ 2 ]. For instance, case reports are cited less often than other kinds of medical research articles [ 68 ]. Thus they can lower a journal’s impact factor, potentially making the journal less attractive. This might lead some high-impact journals to publish few or no case reports, while other journals have chosen to specialize in this genre.
Before deciding on producing a case report or case series based on a particular patient or patients at hand, the observant clinician has to determine if the case report method is the appropriate article type. This review could hopefully assist in that judgment and perhaps be a stimulus to the continuing debate in the medical community on the value of case reporting.
Competing interests
The authors declare that there are no competing interests.
Authors’ contributions
TN contributed to the conception, drafting, and revision of the article. RW contributed to the conception, drafting, and revision of the article. Both authors approved the final manuscript.
Acknowledgements
There was no specific funding for this study.
- Cabán-Martinez AJ, Beltrán WF. Advancing medicine one note at a time: the educational value in clinical case reports. BMC Res Notes. 2012; 5 :293. doi: 10.1186/1756-0500-5-293. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Nissen T, Wynn R. The recent history of the clinical case report: a narrative review. JRSM Sh Rep. 2012; 3 :87. doi: 10.1258/shorts.2012.012046. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Nissen T, Wynn R. The history of the case report: a selective review. JRSM Open. 2014; 5 :4. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Wilkinson G. Fare the well – the Editor’s last words. Br J Psychiatry. 2003; 182 :465–466. doi: 10.1192/bjp.182.6.465. [ CrossRef ] [ Google Scholar ]
- Williams DDR. In defence of case of the case report (Letter) Br J Psychiatry. 2004; 184 :84. doi: 10.1192/bjp.184.1.84. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Enoch MD. Case reports (Letter) Br J Psychiatry. 2005; 185 :169. [ PubMed ] [ Google Scholar ]
- Nahum AM. The clinical case report: “Pot boiler” or scientific literature? Head Neck Surg. 1979; 1 :291–292. doi: 10.1002/hed.2890010402. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Doherty M. What value case reports? (Editorial) Ann Rheum Dis. 1994; 53 :1–2. doi: 10.1136/ard.53.1.1. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Procopio M. Publication of case reports. Br J Psychiatry. 2005; 187 :91. [ PubMed ] [ Google Scholar ]
- Bignall J, Horton R. Learning from stories – Lancet’s case reports. Lancet. 1995; 346 :1256. [ PubMed ] [ Google Scholar ]
- Kidd M, Hubbard C. Introducing Journal of Medical Case Reports. J Med Case Rep. 2007; 1 :1. doi: 10.1186/1752-1947-1-1. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Rison RA. A guide to writing case reports for the Journal of Medical Case Reports and BioMed Central Research Notes. J Med Case Reports. 2013; 7 :239. doi: 10.1186/1752-1947-7-239. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Bayoumi AM, Kopplin PA. The storied case report. CMAJ. 2004; 171 :569–570. doi: 10.1503/cmaj.1031503. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Kidd MR, Saltman D. Case reports at the vanguard of the 21 st century medicine (Editorial) J Med Case Rep. 2012; 6 :156. doi: 10.1186/1752-1947-6-156. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Venes D. Taber’s Cyclopedic Medical Dictionary. 21. Philadelphia: F.A. Davis Company; 2009. [ Google Scholar ]
- Jenicek M. Clinical case reporting in evidence-based medicine. Oxford: Butterworth Heinemann; 1999. [ Google Scholar ]
- Fletcher RH, Fletcher SW. Clinical epidemiology: The essentials. 4. Philadelphia: Lippincott Williams & Wilkins; 2005. [ Google Scholar ]
- Sackett DL, Brian Haynes R, Tugwell P. Clinical epidemiology. Boston: Little, Brown and Company; 1985. [ Google Scholar ]
- Berkencotter C. Patient tales. Case histories and the uses of narrative in psychiatry. Columbia: University of South Carolina Press; 2008. [ Google Scholar ]
- Hunter KM. Doctors’ stories. The narrative structure of medical knowledge. Princeton: Princeton University Press; 1991. [ Google Scholar ]
- Crowe S, Cresswell K, Robertson A, Huby G, Avery A, Sheikh A. The case study approach. BMC Med Res Methodol. 2011; 11 :100. doi: 10.1186/1471-2288-11-100. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Wynn R, Kvalvik AM, Hynnekleiv T. Attitudes to coercion at two Norwegian psychiatric units. Nord J Psychiatry. 2011; 65 :133–137. doi: 10.3109/08039488.2010.513068. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Wynn R, Myklebust LH, Bratlid T. Psychologists and coercion: decisions regarding involuntary psychiatric admission and treatment in a group of Norwegian psychologists. Nord J Psychiatry. 2007; 61 :433–437. doi: 10.1080/08039480701773139. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Aaker E, Knudsen A, Wynn R, Lund A. General practitioners’ reactions to non-compliant patients. Scand J Prim Health Care. 2001; 19 :103–106. doi: 10.1080/028134301750235330. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- McBride WG. Thalidomide and congenital abnormalities. (Letter) Lancet. 1961; 278 :1358. [ Google Scholar ]
- Chakra CAN, Pariente A, Pinet M, Nkeng L, Moore N, Moride Y. Case series in drug safety. A review to determine characteristics and quality. Drug Saf. 2010; 33 :1081–1088. doi: 10.2165/11539300-000000000-00000. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Ban TA. The role of serendipity in drug discovery. Dialogues Clin Neurosci. 2006; 8 :335–344. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Cade JFJ. Lithium salts in the treatment of psychotic excitement. Med J Aust. 1949; 2 :349–352. [ PubMed ] [ Google Scholar ]
- Léauté-Labrèze C, de la Roque Dumas E, Hubiche T, Boralevi F. Propranolol for severe hemangiomas of infancy. (Letter to the editor) N Engl J Med. 2008; 358 :2649–2651. doi: 10.1056/NEJMc0708819. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Fretheim A. Godt nok dokumentert? (Sufficiently documented?) (Editorial) Tidsskr Nor Legeforen. 2010; 130 :1806. [ PubMed ] [ Google Scholar ]
- U.S. National Institutes of Health. Studies found for the search 'propanolol and hemangiomas' in the ClinicalTrials.gov database. Accessed on 22 April, 2014 at: http://www.clinicaltrials.gov/ct2/results?term=propanolol+and+hemangiomas&Search=Search .
- Beard G. Neurasthenia, or nervous exhaustion. Boston Med Surg J. 1869; 80 :217–221. doi: 10.1056/NEJM186904290801301. [ CrossRef ] [ Google Scholar ]
- Taylor DM. Developing a strategy for the management of rare disorders. BMJ. 2012; 344 :e2417. doi: 10.1136/bmj.e2417. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Bjånes TK. Lokale bivirkninger ved parenteral administrering av legemidler. (Localized side effects caused by parenteral administration of drugs) Tidsskr Nor Legeforen. 2011; 131 :472–474. [ PubMed ] [ Google Scholar ]
- Nissen T, Rørvik P, Haugslett L, Wynn R. Physical restraint and near death of a psychiatric patient. J Forensic Sci. 2013; 58 :259–262. doi: 10.1111/j.1556-4029.2012.02290.x. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Wynn R. Coercion in psychiatric care: clinical, legal, and ethical controversies. Int J Psychiatry Clin Pract. 2006; 10 :247–251. doi: 10.1080/13651500600650026. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Lundh A, Christensen M, Jørgensen AW. International or national publication of case reports. Dan Med Bul. 2011; 58 :A4242. [ PubMed ] [ Google Scholar ]
- Grønli O, Wynn R. Normocalcemic hyperparathyroidism and treatment resistant depression. Psychosomatics. 2013; 54 :493–497. doi: 10.1016/j.psym.2012.10.008. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Ward SP. Thalidomide and congenital abnormalities. BMJ. 1962; 2 (5305):646–647. doi: 10.1136/bmj.2.5305.646. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Coodin FJ, Uchida CM, Murphy CH. Phocomelia: report of three cases. CMAJ. 1962; 87 :735–739. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Heyman DJ. Managing director of the Destillers Company Ltd. (Letter) Lancet. 1961; 278 :1262. [ Google Scholar ]
- Agha R, Rosin RD. Time for a new approach to case reports. (Editorial) Int J Surg Case Rep. 2010; 1 :1–3. doi: 10.1016/j.ijscr.2010.04.001. doi:10.1016/j.ijscr.2010.04.001. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Aronson JA. Anecdotes as evidence. (Editorial) BMJ. 2003; 326 :1346. doi: 10.1136/bmj.326.7403.1346. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Myers CS. A contribution to the study of the shell shock. Being an account of three cases of loss of memory, vision, smell and taste, admitted into the Duchess of Westminster’s War Hospital, Le Touquet. Lancet. 1915; 185 :316–320. doi: 10.1016/S0140-6736(00)52916-X. [ CrossRef ] [ Google Scholar ]
- Morris BA. The importance of case reports. (Letter to the Editor) CMAJ. 1989; 141 :875–876. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Rison RA. Neurology case reports: a call for all. J Med Case Rep. 2011; 5 :113. doi: 10.1186/1752-1947-5-113. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Grimes DA, Schulz KF. Descriptive studies: what they can and cannot do. Lancet. 2002; 359 :145–149. doi: 10.1016/S0140-6736(02)07373-7. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Kang S. Anecdotes in medicine - 15 years of Lancet case reports. Lancet. 2010; 376 :1448–1449. doi: 10.1016/S0140-6736(10)60624-1. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Rose JC, Corn M. Dr. E. and other patients: new lessons from old case reports. J Hist Med Allied Sci. 1984; 39 :3–32. doi: 10.1093/jhmas/39.1.3. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Álvarez Millán C. Practice versus theory: tenth-century case histories from Islamic Middle East. Soc Hist Med. 2000; 13 :293–306. doi: 10.1093/shm/13.2.293. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Álvarez Millán C. The case history in Medieval Islamic medical literature: Tajarib and Mujarrabat as source. Med Hist. 2010; 54 :195–214. doi: 10.1017/S0025727300006712. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Hulley SB, Cummings SR, Browner WS, Grady DG, Newman TB. Designing clinical research. 3. Philadelphia: Lippincott Williams & Wilkins; 2007. [ Google Scholar ]
- Easterbrook PJ, Gopalan R, Berlin JA, Matthews DR. Publication bias in clinical research. Lancet. 1991; 337 :867–872. doi: 10.1016/0140-6736(91)90201-Y. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Albrecht J, Meves A, Bigby M. Case reports and case series from the Lancet had significant impact on medical literature. J Clin Epidemiol. 2005; 58 :1227–1232. doi: 10.1016/j.jclinepi.2005.04.003. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Oliveira GJ, Leles CR. Critical appraisal and positive outcome bias in case reports published in Brazilian dental journals. J Dent Educ. 2006; 70 :869–874. [ PubMed ] [ Google Scholar ]
- Charlton BG, Walston F. Individual case studies in clinical research. J Eval Clin Practice. 1998; 4 :147–155. doi: 10.1111/j.1365-2753.1998.tb00081.x. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Stuebe AM. Level IV evidence – adverse anecdote and clinical practice. N Engl J Med. 2011; 365 :8–9. doi: 10.1056/NEJMp1102632. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Hoffman JR. Rethinking case reports. West J Med. 1999; 170 :253–254. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Wilkinson G, Fahy T, Russell G, Healy D, Marks I, Tantam D, Dimond B. Case reports and confidentiality. (Editorial) Br J Psychiatry. 1995; 166 :555–558. doi: 10.1192/bjp.166.5.555. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Powell PP. The single-case report in medical literature: the ‘Elephant man’ serves as an exellent example. J Am Med Writers Assoc. 1990; 5 :9–12. [ Google Scholar ]
- Glasziou P. Evidence based case report: twenty year cough in a non-smoker. BMJ. 1998; 316 :1660–1661. doi: 10.1136/bmj.316.7145.1660. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Harker N, Montgomery A, Fahey T. Interactive case report. Treating nausea and vomiting during pregnancy: case outcome. BMJ. 2004; 328 :276. doi: 10.1136/bmj.328.7434.276. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Reis S, Hermoni D, Livingstone P, Borkan J. Integrated narrative and evidence based case report: case report of paroxysmal atrial fibrillation and anticoagulation. BMJ. 2002; 325 :1018–1020. doi: 10.1136/bmj.325.7371.1018. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Sorinola O, Olufowobi O, Coomarasamy A, Khan KS. Instructions to authors for case reporting are limited: a review of a core journal list. BMC Med Educ. 2004; 4 :4. doi: 10.1186/1472-6920-4-4. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Rossor MN. In: How to write a paper. 4. Hall GM, editor. London: Blackwell Publishing; 2008. How to write a case report; pp. 71–75. [ Google Scholar ]
- Gagnier JJ, Kienle G, Altman DG, Moher D, Sox H, Riley D. CARE group. The CARE guidelines: consensus-based clinical case reporting guideline development. J Med Case Rep. 2013; 7 :223. doi: 10.1186/1752-1947-7-223. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- BioMed Central. Cases Database. Accessed on 22 April, 2014 at: http://www.casesdatabase.com .
- Mason RA. The case report – an endangered species? (Editorial) Anesthesia. 2001; 56 :99–102. doi: 10.1046/j.1365-2044.2001.01919.x. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Election 2024
- Entertainment
- Newsletters
- Photography
- Personal Finance
- AP Investigations
- AP Buyline Personal Finance
- AP Buyline Shopping
- Press Releases
- Israel-Hamas War
- Russia-Ukraine War
- Global elections
- Asia Pacific
- Latin America
- Middle East
- Election Results
- Delegate Tracker
- AP & Elections
- Auto Racing
- 2024 Paris Olympic Games
- Movie reviews
- Book reviews
- Financial Markets
- Business Highlights
- Financial wellness
- Artificial Intelligence
- Social Media
Things to know about the gender-affirming care case as the Supreme Court prepares to weigh in
FILE - A flag supporting LGBTQ+ rights decorates a desk on the Democratic side of the Kansas House of Representatives during a debate, March 28, 2023, at the Statehouse in Topeka, Kan. The U.S. Supreme Court agreed Monday to consider whether a Tennessee ban on gender-affirming care for minors is constitutional. (AP Photo/John Hanna, File)
- Copy Link copied
The U.S. Supreme Court said Monday that it will hear arguments on the constitutionality of state bans on gender-affirming care for transgender minors.
The issue has emerged as a big one in the past few years. While transgender people have gained more visibility and acceptance in many respects, half the states have pushed back with laws banning certain health care services for transgender kids.
Things to know about the issue:
What is gender-affirming care?
Gender-affirming care includes a range of medical and mental health services to support a person’s gender identity, including when it’s different from the sex they were assigned at birth.
The services are offered to treat gender dysphoria, the unease a person may have because their assigned gender and gender identity don’t match. The condition has been linked to depression and suicidal thoughts.
Gender-affirming care encompasses counseling and treatment with medications that block puberty, and hormone therapy to produce physical changes. Those for transgender men cause periods to stop, increase facial and body hair, and deepen voices, among others. The hormones used by transgender women can have effects such as slowing growth of body and facial hair and increasing breast growth.
Gender-affirming care can also include surgery, including operations to transform genitals and chests. These surgeries are rarely offered to minors .
What laws are states passing?
Over the past three years, 26 Republican-controlled states have passed laws restricting gender-affirming care for minors. Most of the laws ban puberty blockers, hormone treatment and surgery for those under 18. Some include provisions that allow those already receiving treatment to continue.
The laws also make exceptions for gender-affirming treatments that are not part of a gender transition, such as medications to stop breast growth in boys and excessive facial hair in girls.
One of the laws — in Arkansas — was nixed by a federal court and is not being enforced.
Meanwhile, at least 14 Democratic-controlled states have adopted laws intended to protect access to gender-affirming care.
The gender-affirming care legislation is a major part of a broader set of laws and policies that has emerged in Republican-controlled states that rein in rights of transgender people. Other policies, adopted in the name of protecting women and girls, bar transgender people from school bathrooms and sports competitions that align with their gender.
What have courts said so far?
Most of the bans have faced court challenges, and most are not very far along in the legal pipeline yet.
The law in Arkansas is the only one to have been struck down entirely, but the state has asked a federal appeals court to reverse that ruling.
The 6th U.S. Circuit Court of Appeals, one step below the Supreme Court, last year ruled that Kentucky and Tennessee can continue to enforce their bans amid legal challenges. The high court has agreed to hear the Tennessee case in the term that starts later this year.
The U.S. Supreme Court in April ruled that Idaho can enforce its ban while litigation over it proceeds. A lower court had put it on hold.
What does the medical community think?
Every major U.S. medical group, including the American Academy of Pediatrics and the American Medical Association, has opposed the bans and said that gender-affirming treatments can be medically necessary and are supported by evidence.
But around the world, medical experts and government health officials are not in lockstep. Some European countries in recent years have warned about overdiagnosis of gender dysphoria.
In England, the state-funded National Health Service commissioned a review of gender identity services for children and adolescents, appointing retired pediatrician Dr. Hilary Cass to lead the effort. The final version of the Cass Review , published in April, found “no good evidence on the long-term outcomes of interventions to manage gender-related distress.”
England’s health service stopped prescribing puberty blockers to children with gender dysphoria outside of a research setting, following recommendations from Cass’ interim report.
The World Professional Association for Transgender Health and its U.S. affiliate issued a statement in May saying they’re deeply concerned about the process, content and consequences of the review, saying it “deprives young trans and gender diverse people of the high-quality care they deserve and causes immense distress and harm to both young patients and their families.”

DC's Care and Concern Matter to Judge
A patient suffered a stroke after experiencing neurological symptoms following a chiropractic treatment. What the DC did afterward made a significant impact on the outcome of his claim.
Posted in Case Studies on Wednesday, June 12, 2024
Carrie Nicholson, 35, first saw Dr. Schmidt on June 12, 2018, and complained of recent onset of pain in the posterior cervical region, left trapezius, upper thoracic and right posterior trapezius for about a week. Dr. Schmidt performed a maximum foramina compression bilaterally and a Jackson foraminal compression. The patient felt better after treatment. The cervical X-ray was normal.
The patient then returned to see Dr. Schmidt on June 14 and June 16, and the same treatment was administered. A few hours after the appointment on June 16, Ms. Nicholson called Dr. Schmidt and reported that her peripheral vision was fuzzy and she had a severe headache. She denied any other neurological symptoms. Dr. Schmidt informed her she should go to the ER for further evaluation. Dr. Schmidt called the patient later that evening to see how she was doing and she informed him that she decided not to go to the ER as she assumed it was a migraine.
She then returned to see Dr. Schmidt on June 18. At that visit she reported her neck and back pain had improved but had been experiencing a headache for a few days. The headache was located in the back of her head and top of her neck, or the occipital region. She believed it was this migraine that caused her blurry vision. Dr. Schmidt then administered the same treatment as he did on the previous visits. Immediately after the adjustment, the patient reported she felt dizzy, and this then worsened over the next several minutes. She attempted to keep her eyes open but was unable to and was also unable to respond to Dr. Schmidt’s questions. An ambulance was called, and Ms. Nicholson was taken to the emergency room. Dr. Schmidt contacted Mr. Nicholson to alert him to the situation. Dr. Schmidt went to the hospital after the ambulance left his office in order to provide any information that the ER physician may need in order to assist with the proper diagnosis and treatment.
Ms. Nicholson was diagnosed with a vertebral artery dissection and stroke, which was found to be remote infarctions in the medial occipital lobes bilaterally, and remote lacunar infarctions in the right and left cerebellar hemispheres. Ms. Nicholson suffered permanent vision and cognitive injuries. Several months after the stroke, Plaintiff underwent a neuropsychological examination that found significant memory and cognitive defects.
Ms. Nicholson filed a chiropractic malpractice lawsuit against Dr. Schmidt alleging that a proper history and physical was not performed on June 12 and June 18. Specifically, the Plaintiff asserted that Dr. Schmidt should have determined that Ms. Nicholson’s neck pain on June 12 was potentially a dissecting vertebral artery for which medical care was required. Plaintiff also asserted that her blurry vision and headache on June 16 and 18 were neurological symptoms which made chiropractic treatment a contraindication until she had been cleared medically.
On behalf of Dr. Schmidt, NCMIC retained a chiropractic and a neurology expert. The chiropractic expert saw no deviation from the standard of care on the part of Dr. Schmidt and noted that the patient improved from the treatments administered on June 12 and 14, so there was no reason to believe a different treatment should have been provided on June 18. The chiropractic expert also opined that visual disturbances are not a contraindication to cervical adjustment. The Defendant’s neurology expert believed that Ms. Nicholson was experiencing a dissecting vertebral artery prior to presenting to Dr. Schmidt on June 12, the dissection was healing and formed a clot, and that the cervical adjustment performed on July 18 dislodged a clot that resulted in her stroke. The neurologist also believed that, in hindsight, the patient’s symptoms of headache and visual disturbances on June 16 were potential warning signs of a stroke and she should have presented to the emergency room.
The primary issue with the case was the cervical adjustment on June 18. Ms. Nicholson’s symptoms of blurred vision and severe headache should have alerted Dr. Schmidt that he needed to do additional testing and possibly consider a medical referral, in light of his previous suggestion for Ms. Nicholson to go to the ER following the treatment two days earlier.
The Outcome
Because of the significant damages suffered by Ms. Nicholson, it was determined that the best course was to settle the case rather than taking a chance at a large verdict at trial. The initial settlement demand was $1 million, which was Dr. Schmidt’s policy limit. This amount included past medical bills of $200,000 as well as significant non-economic damages in the form of permanent vision loss, memory deficits, and general cognition impairment.
While the defense experts did not believe Dr. Schmidt caused the vertebral artery dissection, a jury could have found that the cervical adjustment caused the stroke by dislodging a clot or that Dr. Schmidt should have done additional testing and made a referral during the June 18th visit.
The lawsuit was resolved via settlement in the amount of $225,000. This favorable outcome was achieved due to standard of care expert support, but also because Dr. Schmidt’s actions, which were thoroughly charted, showed his genuine concern for his patient and that he did all he could to ensure she received prompt and proper care.
What Can We Learn
New symptoms or significant changes require new testing. Any new symptoms or significant changes in current symptoms require an additional evaluation and testing by the provider. If the additional testing and examinations are inconclusive, a possible medical referral should be discussed with the patient before additional treatment is considered.
Care about your patients. When a patient experiences a complication while receiving care, it may seem like the best route is to pretend like it did not happen. The best practice is to recognize the complication and immediately take action, which in rare situations requires calling EMS. The value of this case was decreased dramatically because Dr. Schmidt not only did the right thing by immediately addressing Ms. Nicholson’s medical emergency that occurred in his office, but actually went to the ER to provide any information that may have been beneficial for diagnosis and treatment. A jury understands that unexpected outcomes occur in medicine and favor physicians who go above and beyond to help their patients.
About the Author
Dede K. Zupanci focuses her practice on health care defense litigation and regulatory matters, including: chiropractic professional liability, hospital premises liability, and hospital regulations and compliance. She has represented clients in Illinois and Missouri, including metropolitan and rural hospitals, as well as chiropractors and physicians. She is a graduate of Saint Louis University (J.D.) and Southern Illinois University – Carbondale and is a member of the Illinois Association of Defense Trial Counsel, Madison County Bar Association and St. Louis Area Health Lawyers Association. Dede resides in Edwardsville, Illinois.
Resources by Topic
- Continuing Education
- Coronavirus
- Malpractice Insurance
- Business Insurance
- Personal Insurance
- Financial Products
- Chiropractic Resources
- Naturopath Resources
Knowledge to keep you sharp
Whether you're looking for articles, videos, resources, or tools on the business aspects of starting or growing a chiropractic practice, we're here for you.

Read more articles
- Skip to main content
- Keyboard shortcuts for audio player
Weekend Edition Sunday
- Latest Show
Sunday Puzzle
- Corrections
Listen to the lead story from this episode.
Politics chat: Trump and Biden's policy agendas will take center stage at the debate
by Ayesha Rascoe , Mara Liasson
Russia is finding new supporters after being isolated by the West
by Charles Maynes
State laws complicate the federal ban on gun possession for domestic abusers
by Paige Pfleger , Ayesha Rascoe
Middle East
Lebanon is home to the most refugees per capita. now it wants to send some of them back.
by Jane Arraf
Lebanon is home to the most number of refugees. Now it wants to send some of them back
Jonathan merritt's book reminds kids what makes them different makes them special.
by Jason DeRose
Republican lawmakers in Connecticut fight against a future ban on gas-powered vehicles
by Molly Ingram

Sunday Puzzle NPR hide caption
Sunday Puzzle: Summer movie blockbusters
by Will Shortz
Years before intimacy coordinators on Hollywood sets, there was the 1996 film Bound
by Isabella Gomez Sarmiento
Music Interviews
Agalisiga mackey reflects on writing songs in his native cherokee language.
by Ryan Benk , Ayesha Rascoe
Tsitsutsa Tsigesv reflects on writing songs in his native Cherokee language
How biden is getting ready for his high-stakes debate with former president trump.
by Asma Khalid
Young Black voters are feeling increasingly ambivalent about the Democratic Party
by Ayesha Rascoe
Hungary's far-right leader is set to take over rotating presidency of the EU
If ai provides false information, who takes the blame, the hottest new concert venue could be your local cemetery.
by Alison Cuddy
Health Care
'medicaid unwinding' can be dangerous for those who need opioid addiction medications.
by Stephanie Colombini
'Return to Rickwood' traces the history of the Major League's newest field
Author interviews, an incident at a grocery store set sadie dingfelder down the path of writing her book.
Searching for a song you heard between stories? We've retired music buttons on these pages. Learn more here.

IMAGES
VIDEO
COMMENTS
A 44-year-old woman presented with cough, dyspnea, and chest pain. On examination, she had tachycardia and hypotension. Evaluation revealed SARS-CoV-2 RNA in a nasopharyngeal swab, as well as eleva...
A journal publishing case reports in all medical disciplines, including general medicine, drug interaction and adverse reactions. The largest online collection of medical case reports. Validation period: 6/25/2024, 12:27:45 AM - 6/25/2024, 6:27:45 AM. Subscribe Login.
This case was presented at the Harvard Medical School postgraduate course, "Primary Care Internal Medicine 2018," directed by John D. Goodson, M.D. No potential conflict of interest relevant ...
Dr. Pavan K. Bendapudi: I was involved in the care of this patient and am aware of the diagnosis in this case. The patient presented with skin mottling and a rapidly progressive reticular rash ...
A case report is a detailed report of the symptoms, signs, diagnosis, treatment, and follow-up of an individual patient. Case reports usually describe an unusual or novel occurrence and as such, remain one of the cornerstones of medical progress and provide many new ideas in medicine. Some reports contain an extensive review of the relevant ...
Challenges Faced and Lessons Learned from Our Trial of VTE Prophylaxis. G. Le Gal and D. MottierNEJM Evid 2023;2 (9) In this Clinical Trials Case Study, the authors describe the challenges faced and lessons learned conducting a trial of venous thromboembolism prophylaxis among hospitalized older adults. Morning Report.
Journal of Medical Case Reports will consider any original case report that expands the field of general medical knowledge, and original research relating to case reports. Case reports should show one of the following: Unreported or unusual side effects or adverse interactions involving medications. Unexpected or unusual presentations of a disease.
First steps. Begin by sitting down with your medical team to discuss the interesting aspects of the case and the learning points to highlight. Ideally, a registrar or middle grade will mentor you and give you guidance. Another junior doctor or medical student may also be keen to be involved. Allocate jobs to split the workload, set a deadline ...
SAGE Open Medical Case Reports is a peer-reviewed, open access journal, which focusses on providing a publication home for short case reports and case series, which often do not find a place in traditional primary research journals, but provide key insights into real medical cases that are essential for physicians, and may ultimately help to improve patient outcomes.
Editorial. Introduction. Case reports and case series or case study research are descriptive studies to present patients in their natural clinical setting. Case reports, which generally consist of three or fewer patients, are prepared to illustrate features in the practice of medicine and potentially create new research questions that may contribute to the acquisition of additional knowledge ...
Origins of case study research. Case study is a research design that involves an intensive and holistic examination of a contemporary phenomenon in a real-life setting. 1-3 It uses a variety of methods and multiple data sources to explore, describe or explain a single case bounded in time and place (ie, an event, individual, group, organisation or programme).
The LITFL Clinical Case Collection includes over 250 Q&A style clinical cases to assist ' Just-in-Time Learning ' and ' Life-Long Learning '. Cases are categorized by specialty and can be interrogated by keyword from the Clinical Case searchable database. Search by keywords; disease process; condition; eponym or clinical features….
Writing a case report is an excellent way of documenting these findings for the wider medical community—sharing new knowledge that will lead to better and safer patient care. For many medical students and junior doctors, a case report may be their first attempt at medical writing. A published case report will look impressive on your ...
Case study is a research methodology, typically seen in social and life sciences. There is no one definition of case study research.1 However, very simply… 'a case study can be defined as an intensive study about a person, a group of people or a unit, which is aimed to generalize over several units'.1 A case study has also been described as an intensive, systematic investigation of a ...
Emergency medical services were called, and on their arrival, treatment with continuous positive airway pressure was initiated. ... a Chaplain case study on existential distress and ...
induced liver injury. In adults, 11% of cases the patient's platelet coun. anorexia, nausea, ab-Ischemic Hepatopathy dominal pain, and itching.10 Symptoms may first Although the patient had hypotension when the become evident during pregnancy, and postpar-EMS team arrived, the hemodynamic insult in tum e.
Case-control study including individual cases of calcium release deficiency syndrome (CRDS), 3 patient control groups, and genetic mouse models assesses the cardiac repolarization response on an electrocardiogram after brief tachycardia and a pause as a clinical diagnostic test for CRDS.
Harrison's Visual Case Challenge; Internal Medicine Cases; Medical Microbiology Cases; Neuroanatomy Cases; Pathophysiology Cases; Principles of Rehabilitation Medicine Case-Based Board Review; Vanderbilt IM/Peds Curriculum; Study Tools Clerkship Topics; Flashcards; Review Questions
Features the complete collection of basic science, clinical medicine, and post-graduate level cases from 23 Case Files series books, and the personalized functionality to let users mark their progress through cases. Case studies in family medicine, internal medicine, OB/GYN, pediatrics, psychiatry, and surgery.
Background Alzheimer's disease (AD) is a neurodegenerative disease characterized by Aβ plaques and neurofibrillary tangles. Chronic inflammation and synaptic dysfunction lead to disease progression and cognitive decline. Small extracellular vesicles (sEVs) are implicated in AD progression by facilitating the spread of pathological proteins and inflammatory cytokines. This study investigates ...
A 68-year-old man was admitted to the hospital with fever, shortness of breath, and acute kidney injury. Testing of a nasopharyngeal swab for SARS-CoV-2 RNA was positive. Respiratory failure and hy...
A new study by researchers at the Case Western Reserve University School of Medicine reveals that the popular diabetes and weight-loss drugs Wegovy and Ozempic are linked to reduced incidence and recurrence of alcohol abuse or dependence. The team's findings, recently published in the journal Nature Communications, may suggest a possible new treatment for excessive alcohol use—including ...
The study, published in The New England Journal of Medicine, represents the first evidence that having one copy of the Christchurch variant may confer some level of protection against autosomal ...
Background. Throughout history the clinical case report and case report series have been integral components of medical literature [].The case report genre held a strong position until it was sidelined in the second half of the 20 th century [2,3].New methodologies for research articles paved the way for evidence-based medicine.
Case Study Instruction. Please summarize the case of a patient (can be yourself) to whom you prescribed lifestyle change as the primary treatment for one or more chronic diseases. Provide: a brief history or background about the patient and the assessment. the types, intensity (length), and frequency of interventions offered (counseling, group ...
Five weeks earlier, nausea, vomiting, diarrhea, chills, and fever had developed. CT revealed bilateral pelvic masses; examination of a peripheral-blood smear revealed schistocytes, anisocytosis ...
The U.S. Supreme Court said Monday that it will hear arguments on the constitutionality of state bans on gender-affirming care for transgender minors.. The issue has emerged as a big one in the past few years. While transgender people have gained more visibility and acceptance in many respects, half the states have pushed back with laws banning certain health care services for transgender kids.
This amount included past medical bills of $200,000 as well as significant non-economic damages in the form of permanent vision loss, memory deficits, and general cognition impairment. While the defense experts did not believe Dr. Schmidt caused the vertebral artery dissection, a jury could have found that the cervical adjustment caused the ...
The HEALing Communities Study Consortium * Notes Dr. Samet can be contacted at [email protected] or at Boston Medical Center, 801 Massachusetts Ave., 2nd Fl., Boston, MA 02118.
Hear the Weekend Edition Sunday program for Jun 23, 2024